Abstract
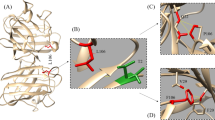
One of the recognized motor neuron degenerative disorders is amyotrophic lateral sclerosis (ALS). By now, several mutations have been reported and linked to ALS patients, some of which are induced by mutations in the human superoxide dismutase (hSOD1) gene. The ALS-provoking mutations are located throughout the structure of hSOD1 and promote the propensity to aggregate. Despite numerous investigations, the underlying mechanism related to the toxicity of mutant hSOD1 through the gain of a toxic function is still vague. We surveyed two mutant forms of hSOD1 by removing and adding cysteine at positions 146 and 72, respectively, to investigate the biochemical characterization and amyloid formation. Our findings predicted the harmful and destabilizing impact of two SOD1 mutants using multiple programs. The specific activity of the wild-type form was about 1.42- and 1.92-fold higher than that of C146R and G72C mutants, respectively. Comparative structural studies using CD spectropolarimetry, and intrinsic and ANS fluorescence showed alterations in secondary structure content, exposure of hydrophobic patches, and structural compactness of WT-hSOD1 vs. mutants. We demonstrated that two mutants were able to promote amyloid-like aggregates under amyloid induction circumstances (50-mM Tris–HCl pH 7.4, 0.2-M KSCN, 50-mM DTT, 37 °C, 190 rpm). Monitoring aggregates were done using an enhancement in thioflavin T fluorescence and alterations in Congo red absorption. The mutants accelerated fibrillation with subsequently greater fluorescence amplitude and a shorter lag time compared to WT-SOD1. These findings support the aggregation of ALS-associated SOD1 mutants as an integral part of ALS pathology.







Similar content being viewed by others
References
Abbasabadi AO, Javanian A, Nikkhah M, Meratan AA, Ghiasi P, Nemat-Gorgani M (2013) Disruption of mitochondrial membrane integrity induced by amyloid aggregates arising from variants of SOD1. Int J Biol Macromol 61:212–217
Alonso Villela SM, Kraiem H, Bouhaouala-Zahar B, Bideaux C, Aceves Lara CA, Fillaudeau L (2020) A protocol for recombinant protein quantification by densitometry. Microbiol Open 9(6):1175–1182
Andersen PM (2006) Amyotrophic lateral sclerosis associated with mutations in the CuZn superoxide dismutase gene. Curr Neurol Neurosci Rep 6(1):37–46
Andersen PM, Sims KB, Xin WW, Kiely R, O’Neill G, Ravits J et al (2003) Sixteen novel mutations in the Cu/Zn superoxide dismutase gene in amyotrophic lateral sclerosis: a decade of discoveries, defects and disputes. Amyotroph Lateral Scler Other Motor Neuron Disord 4(2):62–73
Banci L, Bertini I, Durazo A, Girotto S, Gralla EB, Martinelli M et al (2007) Metal-free superoxide dismutase forms soluble oligomers under physiological conditions: a possible general mechanism for familial ALS. Proc Natl Acad Sci USA 104(27):11263–11267
Basith S, Manavalan B, Lee G (2022) Amyotrophic lateral sclerosis disease-related mutations disrupt the dimerization of superoxide dismutase 1-A comparative molecular dynamics simulation study. Comput Biol Med 151:106319
Baziyar P, Seyedalipour B, Hosseinkhani S (2022a) Zinc binding loop mutations of hSOD1 promote amyloid fibrils under physiological conditions: Implications for initiation of amyotrophic lateral sclerosis. Biochimie 199:170–181
Baziyar P, Seyedalipour B, Hosseinkhani S, Nazifi E (2022b) Development of in silico analysis and molecular dynamics simulation on L67P and D76Y mutants of the human superoxide dismutase 1 (hSOD1) related to amyotrophic lateral sclerosis. Iran J Biotechnol 20(4):26–37
Bendl J, Stourac J, Salanda O, Pavelka A, Wieben ED, Zendulka J et al (2014) PredictSNP: robust and accurate consensus classifier for prediction of disease-related mutations. PLoS Comput Biol 10(1):e1003440
Beyer WF Jr, Fridovich I, Mullenbach GT, Hallewell R (1987) Examination of the role of arginine-143 in the human copper and zinc superoxide dismutase by site-specific mutagenesis. J Biol Chem 262(23):11182–11187
Bhatia NK, Srivastava A, Katyal N, Jain N, Khan MAI, Kundu B, Deep S (2015) Curcumin binds to the pre-fibrillar aggregates of Cu/Zn superoxide dismutase (SOD1) and alters its amyloidogenic pathway resulting in reduced cytotoxicity. Biochim Biophys Acta Proteins Proteomics 1854(5):426–436
Biancalana M, Koide S (2010) Molecular mechanism of Thioflavin-T binding to amyloid fibrils. Biochim Biophys Acta Proteins Proteomics 1804(7):1405–1412
Bourassa MW, Miller LM (2012) Metal imaging in neurodegenerative diseases. Metallomics 4(8):721–738
Brown NM, Torres AS, Doan PE, O’Halloran TV (2004) Oxygen and the copper chaperone CCS regulate posttranslational activation of Cu, Zn superoxide dismutase. Proc Natl Acad Sci USA 101(15):5518–5523
Byström R, Andersen PM, Gröbner G, Oliveberg M (2010) SOD1 mutations targeting surface hydrogen bonds promote amyotrophic lateral sclerosis without reducing apo-state stability. J Biol Chem 285(25):19544–19552
Capriotti E, Fariselli P, Casadio R (2005) I-Mutant2.0: predicting stability changes upon mutation from the protein sequence or structure. Nucleic Acids Res 33(Suppl_2):W306–W310
Chattopadhyay M, Nwadibia E, Strong CD, Gralla EB, Valentine JS, Whitelegge JP (2015) The disulfide bond, but not zinc or dimerization, controls initiation and seeded growth in amyotrophic lateral sclerosis-linked Cu, Zn superoxide dismutase (SOD1) fibrillation. J Biol Chem 290(51):30624–30636
Chia R, Chiò A, Traynor BJ (2018) Novel genes associated with amyotrophic lateral sclerosis: diagnostic and clinical implications. Lancet Neurol 17(1):94–102
Chiti F, Taddei N, Baroni F, Capanni C, Stefani M, Ramponi G, Dobson CM (2002) Kinetic partitioning of protein folding and aggregation. Nat Struct Biol 9(2):137–143
Choi ES, Dokholyan NV (2021) SOD1 oligomers in amyotrophic lateral sclerosis. Curr Opin Struct Biol 66:225–230
Choi I, Song HD, Lee S, Yang YI, Nam JH, Kim SJ et al (2011) Direct observation of defects and increased ion permeability of a membrane induced by structurally disordered Cu/Zn-superoxide dismutase aggregates. PLoS ONE 6(12):e28982
Esmaeili S, Ghobadi N, Akbari V, Moradi S, Shahlaie M, Ghobadi S et al (2020) Pyridine-2, 3-dicarboxylate, quinolinic acid, induces 1N4R Tau amyloid aggregation in vitro: another evidence for the detrimental effect of the inescapable endogenous neurotoxin. Chemico-Biol Interact 315:108884
Ferraroni M, Rypniewski W, Wilson K, Viezzoli M, Banci L, Bertini I, Mangani S (1999) The crystal structure of the monomeric human SOD mutant F50E/G51E/E133Q at atomic resolution. The enzyme mechanism revisited. J Mol Biol 288(3):413–426
Fetherolf MM, Boyd SD, Taylor AB, Kim HJ, Wohlschlegel JA, Blackburn NJ et al (2017) Copper–zinc superoxide dismutase is activated through a sulfenic acid intermediate at a copper ion entry site. J Biol Chem 292(29):12025–12040
Frid P, Anisimov SV, Popovic N (2007) Congo red and protein aggregation in neurodegenerative diseases. Brain Res Rev 53(1):135–160
Furukawa Y (2021) Good and bad of Cu/Zn-superoxide dismutase controlled by metal ions and disulfide bonds. Chem Lett 50(2):331–341
Furukawa Y, Torres AS, O’Halloran TV (2004) Oxygen-induced maturation of SOD1: a key role for disulfide formation by the copper chaperone CCS. EMBO J 23(14):2872–2881
Glenner G, Page D, Eanes E (1972) The relation of the properties of Congo red-stained amyloid fibrils to the β-conformation. J Histochem Cytochem 20(10):821–826
Goutman SA, Chen KS, Paez-Colasante X, Feldman EL (2018) Emerging understanding of the genotype–phenotype relationship in amyotrophic lateral sclerosis. Handb Clin Neurol 148:603–623
Hawe A, Sutter M, Jiskoot W (2008) Extrinsic fluorescent dyes as tools for protein characterization. Pharm Res 25:1487–1499
Hough MA, Strange RW, Hasnain SS (2000) Conformational variability of the Cu site in one subunit of bovine CuZn superoxide dismutase: the importance of mobility in the Glu119-Leu142 loop region for catalytic function. J Mol Biol 304(2):231–241
Huai J, Zhang Z (2019) Structural properties and interaction partners of familial ALS-associated SOD1 mutants. Front Neurol 10:527
Huo J, Shi H, Yao Q, Chen H, Wang L, Chen K (2010) Cloning and purification of recombinant silkworm dihydrolipoamide dehydrogenase expressed in Escherichia coli. Protein Expr Purif 72(1):95–100
Hwang Y-M, Stathopulos PB, Dimmick K, Yang H, Badiei HR, Tong MS et al (2010) Nonamyloid aggregates arising from mature copper/zinc superoxide dismutases resemble those observed in amyotrophic lateral sclerosis. J Biol Chem 285(53):41701–41711
Jahan I, Nayeem SM (2020) Conformational dynamics of superoxide dismutase (SOD1) in osmolytes: a molecular dynamics simulation study. RSC Adv 10(46):27598–27614
Jang J-Y, Cho H, Park H-Y, Rhim H, Kang S (2017) ALS-linked mutant SOD1 proteins promote Aβ aggregates in ALS through direct interaction with Aβ. Biochem Biophys Res Commun 493(1):697–707
Kang J, Lim L, Song J (2023) ATP and Zn2+ induce folding of ALS-associated hSOD1 with fundamentally distinctive mechanisms. bioRxiv 2023.2004. 2029.538825
Karch CM, Prudencio M, Winkler DD, Hart PJ, Borchelt DR (2009) Role of mutant SOD1 disulfide oxidation and aggregation in the pathogenesis of familial ALS. Proc Natl Acad Sci USA 106(19):7774–7779
Keerthana S, Kolandaivel P (2015) Structural investigation on the electrostatic loop of native and mutated SOD1 and their interaction with therapeutic compounds. RSC Adv 5(44):34630–34644
Khare SD, Caplow M, Dokholyan NV (2006) FALS mutations in Cu, Zn superoxide dismutase destabilize the dimer and increase dimer dissociation propensity: a large-scale thermodynamic analysis. Amyloid 13(4):226–235
Klunk WE, Jacob RF, Mason RP (1999) [19] Quantifying amyloid by Congo red spectral shift assay. In: Methods in enzymology, vol 309. Elsevier, Amsterdam, pp 285–305
Kozlowski H, Luczkowski M, Remelli M, Valensin D (2012) Copper, zinc and iron in neurodegenerative diseases (Alzheimer’s, Parkinson’s and prion diseases). Coord Chem Rev 256(19–20):2129–2141
Lakowicz JR (2006) Principles of fluorescence spectroscopy. Springer, Berlin
Li H-F, Wu Z-Y (2016) Genotype–phenotype correlations of amyotrophic lateral sclerosis. Transl Neurodegener 5(1):1–10
Lin F, Yan D, Chen Y, E, F., Shi, H., Han, B., & Zhou, Y. (2018) Cloning, purification and enzymatic characterization of recombinant human superoxide dismutase 1 (hSOD1) expressed in Escherichia coli. Acta Biochim Pol 65(2):235–240
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47(3):469–474
Mavadat E, Seyedalipour B, Hosseinkhani S (2023a) A double point mutation of SOD1 targeting net charge promotes aggregation under destabilizing conditions: Correlation of charge distribution and ALS-provoking mutation. Biochim Biophys Acta Gen Subj 1867(5):130325
Mavadat E, Seyedalipour B, Hosseinkhani S, Colagar AH (2023b) Role of charged residues of the “electrostatic loop” of hSOD1 in promotion of aggregation: Implications for the mechanism of ALS-associated mutations under amyloidogenic conditions. Int J Biol Macromol 244:125289
McAlary L, Shephard V, Wright G, Yerbury J (2022) A copper chaperone-mimetic polytherapy for SOD1-associated amyotrophic lateral sclerosis. J Biol Chem 298(3):101612
Menzies FM, Fleming A, Rubinsztein DC (2015) Compromised autophagy and neurodegenerative diseases. Nat Rev Neurosci 16(6):345–357
Mulligan VK, Chakrabartty A (2013) Protein misfolding in the late-onset neurodegenerative diseases: common themes and the unique case of amyotrophic lateral sclerosis. Proteins Struct Funct Bioinform 81(8):1285–1303
Münch C, Bertolotti A (2010) Exposure of hydrophobic surfaces initiates aggregation of diverse ALS-causing superoxide dismutase-1 mutants. J Mol Biol 399(3):512–525
Namadyan N, Seyedalipour B, Hosseinkhani S, Baziyar P (2023) Biochemical and biophysical properties of the novel ALS-linked hSOD1 mutants: an experimental study accompanied by in silico analysis. J Iran Chem Soc 20(1):125–138
Nilsson MR (2004) Techniques to study amyloid fibril formation in vitro. Methods 34(1):151–160
Oztug Durer ZA, Cohlberg JA, Dinh P, Padua S, Ehrenclou K, Downes S et al (2009) Loss of metal ions, disulfide reduction and mutations related to familial ALS promote formation of amyloid-like aggregates from superoxide dismutase. PLoS ONE 4(3):e5004
Pereira GRC, Vieira B, d. A. A., & De Mesquita, J. F. (2021) Comprehensive in silico analysis and molecular dynamics of the superoxide dismutase 1 (SOD1) variants related to amyotrophic lateral sclerosis. PLoS ONE 16(2):e0247841
Phillips S, Wilson L, Borkman R (1986) Acrylamide and iodide fluorescence quenching as a structural probe of tryptophan microenvironment in bovine lens crystallins. Curr Eye Res 5(8):611–620
Pires DE, Ascher DB, Blundell TL (2014) DUET: a server for predicting effects of mutations on protein stability using an integrated computational approach. Nucleic Acids Res 42(W1):W314–W319
Rae TD, Torres AS, Pufahl RA, O’Halloran TV (2001) Mechanism of Cu, Zn-superoxide dismutase activation by the human metallochaperone hCCS. J Biol Chem 276(7):5166–5176
Rodríguez-Sureda V, Vilches Á, Sánchez O, Audí L, Domínguez C (2015) Intracellular oxidant activity, antioxidant enzyme defense system, and cell senescence in fibroblasts with trisomy 21. Oxid Med Cell Longev 2015:509241
Sala FA, Wright GS, Antonyuk SV, Garratt RC, Hasnain SS (2019) Molecular recognition and maturation of SOD1 by its evolutionarily destabilised cognate chaperone hCCS. PLoS Biol 17(2):e3000141
Salehi M, Nikkhah M, Ghasemi A, Arab SS (2015) Mitochondrial membrane disruption by aggregation products of ALS-causing superoxide dismutase-1 mutants. Int J Biol Macromol 75:290–297
Sandelin E, Nordlund A, Andersen PM, Marklund SS, Oliveberg M (2007) Amyotrophic lateral sclerosis-associated copper/zinc superoxide dismutase mutations preferentially reduce the repulsive charge of the proteins. J Biol Chem 282(29):21230–21236
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682
Shaw BF, Valentine JS (2007) How do ALS-associated mutations in superoxide dismutase 1 promote aggregation of the protein? Trends Biochem Sci 32(2):78–85
Sheng Y, Abreu IA, Cabelli DE, Maroney MJ, Miller A-F, Teixeira M, Valentine JS (2014) Superoxide dismutases and superoxide reductases. Chem Rev 114(7):3854–3918
Sirangelo I, Iannuzzi C (2017) The role of metal binding in the amyotrophic lateral sclerosis-related aggregation of copper–zinc superoxide dismutase. Molecules 22(9):1429
Stewart H, Mackenzie I, Eisen A, Brännström T, Marklund S, Andersen P (2006) Clinicopathological phenotype of ALS with a novel G72C SOD1 gene mutation mimicking a myopathy. Muscle Nerve off J Am Assoc Electrodiagn Med 33(5):701–706
Tiwari MK, Hägglund PM, Møller IM, Davies MJ, Bjerrum MJ (2019) Copper ion/H2O2 oxidation of Cu/Zn-Superoxide dismutase: implications for enzymatic activity and antioxidant action. Redox Biol 26:101262
Tompa DR, Kadhirvel S (2020) Changes in hydrophobicity mainly promotes the aggregation tendency of ALS associated SOD1 mutants. Int J Biol Macromol 145:904–913
Valentine JS, Hart PJ (2003) Misfolded CuZnSOD and amyotrophic lateral sclerosis. Proc Natl Acad Sci USA 100(7):3617–3622
Valentine JS, Doucette PA, Zittin Potter S (2005) Copper–zinc superoxide dismutase and amyotrophic lateral sclerosis. Annu Rev Biochem 74:563–593
Vassall KA, Stubbs HR, Primmer HA, Tong MS, Sullivan SM, Sobering R et al (2011) Decreased stability and increased formation of soluble aggregates by immature superoxide dismutase do not account for disease severity in ALS. Proc Natl Acad Sci USA 108(6):2210–2215
Wahiduzzaman KV, Anjum F, Shafie A, Elasbali AM, Islam A et al (2021) Delineating the aggregation-prone hotspot regions (peptides) in the human Cu/Zn superoxide dismutase 1. ACS Omega 6(49):33985–33994
Wang W, Ma T, Zhang B, Yao N, Li M, Cui L et al (2014) A novel mechanism of protein thermostability: a unique N-terminal domain confers heat resistance to Fe/Mn-SODs. Sci Rep 4(1):1–9
Wright GS, Antonyuk SV, Hasnain SS (2019) The biophysics of superoxide dismutase-1 and amyotrophic lateral sclerosis. Q Rev Biophys 52:e12
Zaji HD, Seyedalipour B, Hanun HM, Baziyar P, Hosseinkhani S, Akhlaghi M (2023) Computational insight into in silico analysis and molecular dynamics simulation of the dimer interface residues of ALS-linked hSOD1 forms in apo/holo states: a combined experimental and bioinformatic perspective. 3 BIotech 13(3):92
Zhao N, Feng Z, Shao M, Cao J, Wang F, Liu C (2017) Stability profiles and therapeutic effect of Cu/Zn superoxide dismutase chemically coupled to o-quaternary chitosan derivatives against dextran sodium sulfate-induced colitis. Int J Mol Sci 18(6):1121
Zhao B, Zhuang X, Pi Z, Liu S, Liu Z, Song F (2018) Determining the effect of catechins on SOD1 conformation and aggregation by ion mobility mass spectrometry combined with optical spectroscopy. J Am Soc Mass Spectrom 29(4):734–741
Acknowledgements
The authors would like to express their gratitude to the Research Council of Mazandaran University for financial support during the course of this project.
Author information
Authors and Affiliations
Contributions
BS contributed to conceptualization, supervision, methodology, validation, data curation, formal analysis, and writing, reviewing, and editing of the manuscript; SM contributed to methodology, data curation, validation, and writing and original draft preparation; SZH contributed to methodology, data curation, validation, and writing and original draft preparation; SH contributed to conceptualization, validation, data curation, and writing, reviewing, and editing of the manuscript; MM contributed to methodology and writing, reviewing, and editing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohammadi, S., Seyedalipour, B., Hashemi, S.Z. et al. Implications of ALS-Associated Mutations on Biochemical and Biophysical Features of hSOD1 and Aggregation Formation. Biochem Genet (2024). https://doi.org/10.1007/s10528-023-10619-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10528-023-10619-y