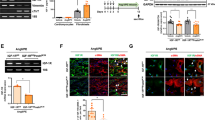
We studied the effects and mechanisms of action of conophylline in different concentrations in the original in vitro model of myocardial fibrosis (treatment of cardiac fibroblasts isolated form the hearts of newborn rats with angiotensin II). Viability, collagen content, and expression of related protein in cardiac fibroblasts were assessed using the MTT-test, Sircol assay, and Western blotting, respectively. Conophylline markedly protected the cultured cells against the development of angiotensin II-induced fibrosis, which was seen from reduced viability of fibroblasts, decreased collagen content, and down-regulation of the expression of α-smooth muscle actin (α-SMA). Conophylline did not affect the TGF-β pathway altered by angiotensin II, but markedly decreased the level of bone morphogenetic protein-4 (BMP4) enhanced by angiotensin II and BMP4 itself. Conophylline produced no effect on phosphorylation of α-SMA and Smad homologue-1/5/8, the classic BMP4 downstream pathway elements, but reduced the level of c-Jun N-terminal kinase (JNK) elevated by BMP4. Conophylline did not inhibit the development of myocardial fibrosis in the presence of JNK activator anisomycin. Thus, conophylline inhibited angiotensin II-provoked myocardial fibrosis via the BMP4/JNK pathway.
Similar content being viewed by others
References
Crawford JR, Haudek SB, Cieslik KA, Trial J, Entman ML. Origin of developmental precursors dictates the pathophysiologic role of cardiac fibroblasts. J. Cardiovasc. Transl. Res. 2012;5(6):749-759. doi: https://doi.org/10.1007/s12265-012-9402-7
Creemers EE, van Rooij E. Function and Therapeutic Potential of Noncoding RNAs in Cardiac Fibrosis. Circ. Res. 2016;118(1):108-118. doi: https://doi.org/10.1161/CIRCRESAHA.115.305242
Dobaczewski M, Chen W, Frangogiannis NG. Transforming growth factor (TGF)-beta signaling in cardiac remodeling. J. Mol. Cell Cardiol. 2011;51(4):600-606. doi: https://doi.org/10.1016/j.yjmcc.2010.10.033
Guo WT, Dong DL. Bone morphogenetic protein-4: a novel therapeutic target for pathological cardiac hypertrophy/heart failure. Heart Fail Rev. 2014;19(6):781-788. doi: https://doi.org/10.1007/s10741-014-9429-8
Guo X, Yan F, Shan X, Li J, Yang Y, Zhang J, Yan X, Bu P. SIRT3 inhibits Ang II-induced transdifferentiation of cardiac fibroblasts through β-catenin/PPAR-γ signaling. Life Sci. 2017;186:111-117. doi: https://doi.org/10.1016/j.lfs.2017.07.030
Hughes SE. The pathology of hypertrophic cardiomyopathy. Histopathology. 2004;44(5):412-427. doi: https://doi.org/10.1111/j.1365-2559.2004.01835.x
Kubo N, Saito R, Hamano K, Nagasawa M, Aoki F, Takei I, Umezawa K, Kuwano H, Kojima I. Conophylline suppresses hepatic stellate cells and attenuates thioacetamide-induced liver fibrosis in rats. Liver Int. 2014;34(7):1057-1067. doi: https://doi.org/10.1111/liv.12328
Nakade Y, Sakamoto K, Yamauchi T, Inoue T, Kobayashi Y, Yamamoto T, Ishii N, Ohashi T, Sumida Y, Ito K, Nakao H, Fukuzawa Y, Umezawa K, Yoneda M. Conophylline inhibits non-alcoholic steatohepatitis in mice. PLoS One. 2017;12(6);e0178436. doi: https://doi.org/10.1371/journal.pone.0178436
Piccoli MT, Gupta SK, Viereck J, Foinquinos A, Samolovac S, Kramer FL, Garg A, Remke J, Zimmer K, Batkai S, Thum T. Inhibition of the Cardiac Fibroblast-Enriched lncRNA Meg3 Prevents Cardiac Fibrosis and Diastolic Dysfunction. Circ. Res. 2017;121(5):575-583. doi: https://doi.org/10.1161/CIRCRESAHA.117.310624
Saito R, Yamada S, Yamamoto Y, Kodera T, Hara A, Tanaka Y, Kimura F, Takei I, Umezawa K, Kojima I. Conophylline suppresses pancreatic stellate cells and improves islet fibrosis in Goto-Kakizaki rats. Endocrinology. 2012;153(2):621-630. doi: https://doi.org/10.1210/en.2011-1767
Sun B, Huo R, Sheng Y, Li Y, Xie X, Chen C, Liu HB, Li N, Li CB, Guo WT, Zhu JX, Yang BF, Dong DL. Bone morphogenetic protein-4 mediates cardiac hypertrophy, apoptosis, and fibrosis in experimentally pathological cardiac hypertrophy. Hypertension. 2013;61(2):352-360. doi: https://doi.org/10.1161/HYPERTENSIONAHA.111.00562
Tezuka T, Ota A, Karnan S, Matsuura K, Yokoo K, Hosokawa Y, Vigetti D, Passi A, Hatano S, Umezawa K, Watanabe H. The plant alkaloid conophylline inhibits matrix formation of fibroblasts. J. Biol. Chem. 2018;293(52):20214-20226. doi: https://doi.org/10.1074/jbc.RA118.005783
Umezawa K, Ohse T, Yamamoto T, Koyano T, Takahashi Y. Isolation of a new vinca alkaloid from the leaves of Ervatamia microphylla as an inhibitor of ras functions. Anticancer Res. 1994;14(6B):2413-2417.
Wang YC, Xiao XL, Li N, Yang D, Xing Y, Huo R, Liu MY, Zhang YQ, Dong DL. Oestrogen inhibits BMP4-induced BMP4 expression in cardiomyocytes: a potential mechanism of oestrogen-mediated protection against cardiac hypertrophy. Br. J. Pharmacol. 2015;172(23):5586-5595. doi: https://doi.org/10.1111/bph.12983
Weber KT, Sun Y, Bhattacharya SK, Ahokas RA, Gerling IC. Myofibroblast-mediated mechanisms of pathological remodelling of the heart. Nat. Rev. Cardiol. 2013;10(1):15-26. doi: https://doi.org/10.1038/nrcardio.2012.158
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 171, No. 3, pp. 282-289, March, 2021
Rights and permissions
About this article
Cite this article
Zhang, S.Q., Bao, Y.N., Lv, L.Y. et al. Conophylline Suppresses Angiotensin II-Induced Myocardial Fibrosis In Vitro via the BMP4/JNK Pathway. Bull Exp Biol Med 171, 305–311 (2021). https://doi.org/10.1007/s10517-021-05217-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05217-0