Abstract
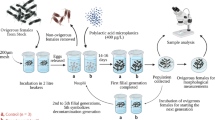
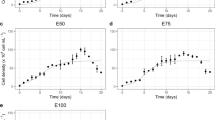
Brine shrimp Artemia is able to filter particulate substances non-selectively and continuously, and its filtration capacity is related to various environmental parameters as well as Artemia growth stage. In this study, the filter-feeding rate (FFR) of Artemia franciscana at different temperatures (20°C /25°C /30°C) and stocking densities (200/400/750/1000 ind./L at two earlier growth stages with the body length of 0.91 ± 0.05 mm and 1.53 ± 0.22 mm, respectively; 50/100/200 ind./L at two later growth stages with the body length of 4.72 ± 0.51 mm and 10.26 ± 0.46 mm, respectively) on three unicellular algae (Chlorella vulgaris, Porphyridium purpureum, Phaeodactylum tricornutum) and two sizes of polyethylene beads (30 μm and 50 μm) was determined at Artemia four growth stages. In total 144 combinations were tested. The results showed that the FFR was positively correlated with the ambient temperature and Artemia body length, while it was negatively correlated with the Artemia density and particle size. One way ANOVA analysis showed that ambient temperature, Artemia stocking density and particle size mostly had significant effects on FFR (P < 0.05). And the favorable filtration particle size of Artemia increased with its body length. The equation of FFR in function of temperature (T), Artemia body length (BL) and stocking density (SD), and particle size (PS) was obtained using multiple linear regression analysis: FFR = 0.487* BL + 0.067* T-0.01* SD-0.064* PS-1.508 (R2 = 0.513). Of these four variables, body length had the greatest effect on FFR, followed by ambient temperature, particle size and Artemia density. There were interactions among three factors (T, SD and PS) and the interactive degree varied with Artemia growth. The results of this study provide a valuable guidance for proper feeding in controlled Artemia production and standardization of feeding protocol for ecotoxicity and fundamental Artemia research.




Similar content being viewed by others
Data availability statement
All relevant data are within the paper.
References
Asil SM, Fereidouni AE, Ouraji H, Khalili KJ (2013) Effects of different light intensities on growth, survival, reproductive and life span characteristics of Artemia urmiana (Günther 1890). Aquac Res 44:554–566. https://doi.org/10.1111/j.1365-2109.2011.03058.x
Ben Naceur H, Jenhani ABR, Romdhane MS (2012) Impacts of salinity, temperature, and pH on the morphology of Artemia salina (Branchiopoda: Anostraca) from Tunisia. Zool Stud 51:453–462
Clarke A, Fraser KPP (2004) Why does metabolism scale with temperature? Funct Ecol 18:243–251. https://doi.org/10.1111/j.0269-8463.2004.00841.x
El-Bermawi N, Baxevanis AD, Abatzopoulos TJ, Van Stappen G, Sorgeloos P (2004) Salinity effects on survival, growth and morphometry of four Egyptian Artemia populations (International Study on Artemia. LXVII). Hydrobiologia 523:175–188. https://doi.org/10.1023/B:HYDR.0000033124.49676.5c
El-Gamal MM (2011) Respiration of Artemia franciscana cultured under different salinities. Anim Biol 61:413–425. https://doi.org/10.1163/157075511X596891
Evjemo JO, Olsen Y (1999) Effect of food concentration on the growth and production rate of Artemia franciscana feeding on algae (T. iso). J Exp Mar Biol Ecol 242:273–296. https://doi.org/10.1016/S0022-0981(99)00104-5
Fernández RG (2001) Artemia Bioencapsulation I. Effect of particle sizes on the filtering behavior of Artemia franciscana. J Crustac Biol 21:435–442. https://doi.org/10.1163/20021975-99990144
Gajardo G, Beardmore J (2012) The brine shrimp Artemia: Adapted to critical life conditions. Front Physiol 3:185. https://doi.org/10.3389/fphys.2012.00185
Gharibi MR, Noori A, Agh N, Atashbar B (2021) Rainbow trout farm effluent as a potential source of feed and medium for mass culture of Artemia parthenogenetica. Aquaculture 530:735714. https://doi.org/10.1016/j.aquaculture.2020.735714
Green AJ, Sánchez MI, Amat F, Figuerola J, Hontoria F, Ruiz O, Hortas F (2005) Dispersal of invasive and native brine shrimps Artemia (Anostraca) via waterbirds. Limnol Oceanogr 50(2):737–742. https://doi.org/10.4319/lo.2005.50.2.0737
Gui L, Xu L, Liu Z, Zhou Z, Sun Z (2022) Carotenoid-rich microalgae promote growth and health conditions of Artemia nauplii. Aquaculture 546:737289. https://doi.org/10.1016/j.aquaculture.2021.737289
Guzmán-Agüero JE, Nieves-Soto M, Hurtado MÁ, Piña-Valdez P, del Garza-Aguirre MC (2013) Feeding physiology and scope for growth of the oyster Crassostrea corteziensis (Hertlein, 1951) acclimated to different conditions of temperature and salinity. Aquac Int 21:283–297. https://doi.org/10.1007/s10499-012-9550-4
Han KN, Lee SW, Wang SY (2008) The effect of temperature on the energy budget of the Manila clam, Ruditapes philippinarum. Aquac Int 16:143–152. https://doi.org/10.1007/s10499-007-9133-y
Huang L, Xi Y, Xu X, Wen X (2012) Responses in population growth and reproduction of the freshwater rotifer Brachionus calyciflorus to microcystin-LR at different temperatures. Ann Limnol Int J Limnol 48:383–390. https://doi.org/10.1051/limn/2012029
Jeyavani J, Sibiya A, Bhavaniramya S, Mahboob S, Al-Ghanim KA, Nisa Z, Riaz MN, Nicoletti M, Govindarajan M, Vaseeharan B (2022) Toxicity evaluation of polypropylene microplastic on marine microcrustacean Artemia salina: An analysis of implications and vulnerability. Chemosphere 296:133990. https://doi.org/10.1016/j.chemosphere.2022.133990
Kundu S, Dasgupta N, Chakraborty B, Paul A, Ray S, Bhattacharya S (2021) Growth acceleration is the key for identifying the most favorable food concentration of Artemia sp. Ecol Model 455:109639. https://doi.org/10.1016/j.ecolmodel.2021.109639
Lavens P, Sorgeloos P (1996) Manual on the production and use of live food for aquaculture. FAO, Rome
Lenormand T, Nougué O, Jabbour-Zahab R, Arnaud F, Dezileau L, Chevin L-M, Sánchez MI (2018) Resurrection ecology in Artemia. Evol Appl 11:76–87. https://doi.org/10.1111/eva.12522
Libralato G, Prato E, Migliore L, Cicero AM, Manfra L (2016) A review of toxicity testing protocols and endpoints with Artemia spp. Ecol Indic 69:35–49. https://doi.org/10.1016/j.ecolind.2016.04.017
Lopes-dos-Santos RMA, De Troch M, Bossier P, Van Stappen G (2019) Archivory in hypersaline aquatic environments: Haloarchaea as a dietary source for the brine shrimp Artemia. FEMS Microbiol Ecol 95:fiz178. https://doi.org/10.1093/femsec/fiz178
Lopes-dos-Santos RMA, Groot R, Liying S, Bossier P, Van Stappen G (2019) Halophilic bacteria as a food source for the brine shrimp Artemia. Aquaculture 500:631–639. https://doi.org/10.1016/j.aquaculture.2018.10.068
Makridis P (1999) Food size selectivity of Artemia franciscana at three developmental stages. J Plankton Res 21:2191–2201. https://doi.org/10.1093/plankt/21.11.2191
Marinho-Soriano E, Azevedo CAA, Trigueiro TG, Pereira DC, Carneiro MAA, Camara MR (2011) Bioremediation of aquaculture wastewater using macroalgae and Artemia. Int Biodeterior Biodegrad 65:253–257. https://doi.org/10.1016/j.ibiod.2010.10.001
Muñoz J, Amat F, Green AJ, Figuerola J, Gomez A (2013) Bird migratory flyways influence the phylogeography of the invasive brine shrimp Artemia franciscana in its native American range. PeerJ 1:e200. https://doi.org/10.7717/peerj.200
Nevejan N, De Schryver P, Wille M, Dierckens K, Baruah K, Van Stappen G (2018) Bacteria as food in aquaculture: do they make a difference? Rev Aquac 10:180–212. https://doi.org/10.1111/raq.12155
Ogburn NJ, Duan L, Subashchandrabose SR, Sorgeloos P, O’Connor W, Megharaj M, Naidu R (2023) Agricultural wastes for brine shrimp Artemia production: A review. Rev Aquac 15:1159–1178. https://doi.org/10.1111/raq.12784
Pinto PM, Bio A, Hontoria F, Almeida V, Vieira N (2013) Portuguese native Artemia parthenogenetica and Artemia franciscana survival under different abiotic conditions. J Exp Mar Biol Ecol 440:81–89. https://doi.org/10.1016/j.jembe.2012.11.016
Redón S, Amat F, Hontoria F, Vasileva GP, Nikolov PN, Georgiev BB (2011) Participation of metanauplii and juvenile individuals of Artemia parthenogenetica (Branchiopoda) in the circulation of avian cestodes. Parasitol Res 108:905–912. https://doi.org/10.1007/s00436-010-2132-3
Reeve MR (1963) The filter-feeding of Artemia: I. In Pure Cultures of Plant Cells. J Exp Biol 40:195–205. https://doi.org/10.1242/jeb.40.1.195
Riisgård HU, Zalacáin D, Jeune N, Wiersma JB, Lüskow F, Pleissner D (2015) Adaptation of the brine shrimp Artemia salina (Branchiopoda: Anostraca) to filter-feeding: Effects of body size and temperature on filtration and respiration rates. J Crustac Biol 35:650–658. https://doi.org/10.1163/1937240X-00002362
Snell TW, Dingmann BJ, Serra M (2001) Density-dependent regulation of natural and laboratory rotifer populations. Hydrobiologia 446:39–44. https://doi.org/10.1023/A:1017564804089
Sorgeloos P, Dhert P, Candreva P (2001) Use of the brine shrimp, Artemia spp., in marine fish larviculture. Aquaculture 200:147–159. https://doi.org/10.1016/S0044-8486(01)00698-6
Sui L, Deng Y, Wang J, Sorgeloos P, Van Stappen G (2014) Impact of brine acidification on hatchability, survival and reproduction of Artemia parthenogenetica and Artemia franciscana in salt ponds, Bohai Bay. China J Oceanol Limnol 32:81–87. https://doi.org/10.1007/s00343-014-3107-5
Tantanasarit C, Babel S, Englande AJ, Meksumpun S (2013) Influence of size and density on filtration rate modeling and nutrient uptake by green mussel (Perna viridis). Mar Pollut Bull 68:38–45. https://doi.org/10.1016/j.marpolbul.2012.12.027
Tzima CS, Banti CN, Hadjikakou SK (2022) Assessment of the biological effect of metal ions and their complexes using Allium cepa and Artemia salina assays: a possible environmental implementation of biological inorganic chemistry. JBIC J Biol Inorg Chem 27:611–629. https://doi.org/10.1007/s00775-022-01963-2
Wang T, Li Q (2020) Effects of temperature, salinity and body size on the physiological responses of the Iwagaki oyster Crassostrea nippona. Aquac Res 51:728–737. https://doi.org/10.1111/are.14423
Xiang XL, Chen YY, Xu QL, Zhu LY, Wen XL, Xi YL (2017) Combined Effects of Temperature and the Microcystin MC-LR on the Feeding Behavior of the Rotifer Brachionus calyciflorus. Bull Environ Contam Toxicol 99:493–499. https://doi.org/10.1007/s00128-017-2172-9
Yampolsky LY, Zeng E, Lopez J, Williams PJ, Dick KB, Colbourne JK, Pfrender ME (2014) Functional genomics of acclimation and adaptation in response to thermal stress in Daphnia. BMC Genom 15:859. https://doi.org/10.1186/1471-2164-15-859
Funding
National Key Research and Development Plan Blue Granary Technology Innovation Project (2020YFD0900705): Demonstration of Integration of Engineering Aquaculture, Fishery and Agriculture Based on Purification and Utilization of Tidal Flat Water Resources in Bohai Bay (2020YFD0900705-2).
Author information
Authors and Affiliations
Contributions
Ke Li: Conducting the experiment, data analysis and manuscript preparation. Yudie Wang: Conducting the experiment and data analysis. Guoru Du and Xueliang Yao: microalgae preparation. Hanyan Bao and Xuekai Han: providing the constructive discussions. Liying Sui: Experimental design, data analysis and manuscript revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, K., Wang, Y., Du, G. et al. Effects of temperature and particle size on the filter-feeding rate of brine shrimp Artemia franciscana at different growth stages and stocking densities. Aquacult Int (2024). https://doi.org/10.1007/s10499-024-01518-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-024-01518-z