Abstract
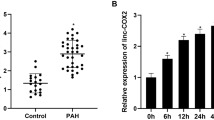
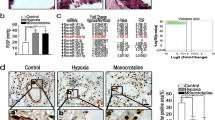
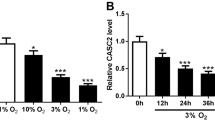
Hypoxic pulmonary hypertension (HPH) is a pathophysiological syndrome in which pulmonary vascular pressure increases under hypoxic stimulation and there is an urgent need to develop emerging therapies for the treatment of HPH. LncRNA MIR210HG is a long non-coding RNA closely related to hypoxia and has been widely reported in a variety of tumor diseases. But its mechanism in hypoxic pulmonary hypertension is not clear. In this study, we identified for the first time the potential effect of MIR210HG on disease progression in HPH. Furthermore, we investigated the underlying mechanism through which elevated levels of MIR210HG promotes the transition from a contractile phenotype to a synthetic phenotype in PASMCs under hypoxia via activation of autophagy-dependent ferroptosis pathway. While overexpression of HIF-2α in PASMCs under hypoxia significantly reversed the phenotypic changes induced by MIR210HG knockdown. We further investigated the potential positive regulatory relationship between STAT3 and the transcription of MIR210HG in PASMCs under hypoxic conditions. In addition, we established both in vivo and in vitro models of HPH to validate the differential expression of specific markers associated with hypoxia. Our findings suggest a potential mechanism of LncRNA MIR210HG in the progression of HPH and offer potential targets for disease intervention and treatment.








Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Humbert M, Kovacs G, Hoeper MM et al (2023) 2022 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Respiratory J Jan 61(1):2200879. https://doi.org/10.1183/13993003.00879-2022
Hoeper MM, Humbert M, Souza R et al (2016) A global view of pulmonary hypertension. Lancet Respiratory Med Apr 4(4):306–322. https://doi.org/10.1016/s2213-2600(15)00543-3
Ruffenach G, Hong J, Vaillancourt M, Medzikovic L, Eghbali M (2020) Pulmonary hypertension secondary to pulmonary fibrosis: clinical data, histopathology and molecular insights. Respir Res Nov 18(1):303. https://doi.org/10.1186/s12931-020-01570-2
Owens GK, Kumar MS, Wamhoff BR (2004) Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol Rev Jul 84(3):767–801. https://doi.org/10.1152/physrev.00041.2003
Ma B, Cao Y, Qin J, Chen Z, Hu G, Li Q (2023) Pulmonary artery smooth muscle cell phenotypic switching: a key event in the early stage of pulmonary artery hypertension. Drug Discov Today May 28(5):103559. https://doi.org/10.1016/j.drudis.2023.103559
Wang G, Jacquet L, Karamariti E, Xu Q (2015) Origin and differentiation of vascular smooth muscle cells. J Physiol Jul 15(14):3013–3030. https://doi.org/10.1113/jp270033
Frid MG, Aldashev AA, Dempsey EC, Stenmark KR (1997) Smooth muscle cells isolated from discrete compartments of the mature vascular media exhibit unique phenotypes and distinct growth capabilities. Circ Res Dec 81(6):940–952. https://doi.org/10.1161/01.res.81.6.940
Zhang H, Zhou S, Sun M et al (2022) Ferroptosis of endothelial cells in Vascular diseases. Nutrients Oct 26(21):4506. https://doi.org/10.3390/nu14214506
Dixon SJ, Stockwell BR (2014) The role of iron and reactive oxygen species in cell death. Nat Chem Biol Jan 10(1):9–17. https://doi.org/10.1038/nchembio.1416
Xie SS, Deng Y, Guo SL et al (2022) Endothelial cell ferroptosis mediates monocrotaline-induced pulmonary hypertension in rats by modulating NLRP3 inflammasome activation. Sci Rep Feb 23(1):3056. https://doi.org/10.1038/s41598-022-06848-7
Wong CM, Preston IR, Hill NS, Suzuki YJ (2012) Iron chelation inhibits the development of pulmonary vascular remodeling. Free Radic Biol Med Nov 1(9):1738–1747. https://doi.org/10.1016/j.freeradbiomed.2012.08.576
Wang E, Zhou S, Zeng D, Wang R (2023) Molecular regulation and therapeutic implications of cell death in pulmonary hypertension. Cell Death Discov Jul 12(1):239. https://doi.org/10.1038/s41420-023-01535-6
Chen YB (2019) Autophagy and its role in pulmonary hypertension. Aging Clin Exp Res Aug 31(8):1027–1033. https://doi.org/10.1007/s40520-018-1063-1
Antonioli M, Di Rienzo M, Piacentini M, Fimia GM (2017) Emerging mechanisms in initiating and terminating Autophagy. Trends Biochem Sci Jan 42(1):28–41. https://doi.org/10.1016/j.tibs.2016.09.008
Zou F, Zhang ZH, Zou SS et al (2023) LncRNA MIR210HG promotes the proliferation, migration, and invasion of lung cancer cells by inhibiting the transcription of SH3GL3. Kaohsiung J Med Sci Dec 39(12):1166–1177. https://doi.org/10.1002/kjm2.12775
Saigusa H, Mimura I, Kurata Y, Tanaka T, Nangaku M (2023) Hypoxia-inducible lncRNA MIR210HG promotes HIF1alpha expression by inhibiting mir-93-5p in renal tubular cells. FEBS J Aug 290(16):4040–4056. https://doi.org/10.1111/febs.16794
Hu XL, Huang XT, Zhang JN et al (2022) Long noncoding RNA MIR210HG is induced by hypoxia-inducible factor 1alpha and promotes cervical cancer progression. Am J Cancer Res 12(6):2783–2797
Darnell JE Jr., Kerr IM, Stark GR (1994) Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. J Article Rev Sci Jun 3(5164):1415–1421. https://doi.org/10.1126/science.8197455
Hu WP, Xie L, Hao SY et al (2022) Protective effects of progesterone on pulmonary artery smooth muscle cells stimulated with interleukin 6 via blocking the shuttling and transcriptional function of STAT3. Article. Int Immunopharmacol Jan 102:108379. https://doi.org/10.1016/j.intimp.2021.108379
Liu YY, Zhang WY, Zhang ML et al (2022) DNA-PKcs participated in hypoxic pulmonary hypertension. Respir Res Sep 16(1):246. https://doi.org/10.1186/s12931-022-02171-x
Ding X, Zhou S, Li M et al (2017) Upregulation of SRF is Associated with Hypoxic Pulmonary hypertension by promoting viability of smooth muscle cells via increasing expression of Bcl-2. J Cell Biochem Sep 118(9):2731–2738. https://doi.org/10.1002/jcb.25922
Xia X, Huang L, Zhou S et al (2023) Hypoxia-induced long non-coding RNA plasmacytoma variant translocation 1 upregulation aggravates pulmonary arterial smooth muscle cell proliferation by regulating autophagy via miR-186/Srf/Ctgf and miR-26b/Ctgf signaling pathways. Int J Cardiol Jan 1 370:368–377. https://doi.org/10.1016/j.ijcard.2022.09.060
Zhou S, Sun L, Cao C et al (2018) Hypoxia-induced microRNA-26b inhibition contributes to hypoxic pulmonary hypertension via CTGF. J Cell Biochem Feb 119(2):1942–1952. https://doi.org/10.1002/jcb.26355
Zhou S, Liu Y, Li M et al (2018) Combined effects of PVT1 and MiR-146a single nucleotide polymorphism on the lung function of smokers with chronic obstructive Pulmonary Disease. Int J Biol Sci 14(10):1153–1162. https://doi.org/10.7150/ijbs.25420
Sun L, Xu A, Li M et al (2021) Effect of methylation status of lncRNA-MALAT1 and MicroRNA-146a on pulmonary function and expression level of COX2 in patients with Chronic Obstructive Pulmonary Disease. Front Cell Dev Biol 9:667624. https://doi.org/10.3389/fcell.2021.667624
Zhu K, Zhou S, Xu A et al (2020) Microbiota Imbalance contributes to COPD Deterioration by enhancing IL-17a production via miR-122 and miR-30a. Mol Ther Nucleic Acids Dec 4:22:520–529. https://doi.org/10.1016/j.omtn.2020.09.017
Zhou S, Zhu K, Du Y et al (2020) Estrogen administration reduces the risk of pulmonary arterial hypertension by modulating the miR-133a signaling pathways in rats. Gene Ther Apr 27(3–4):113–126. https://doi.org/10.1038/s41434-019-0103-6
Wang C, Liu Y, Zhang W et al (2022) circ-BPTF serves as a mir-486-5p sponge to regulate CEMIP and promotes hypoxic pulmonary arterial smooth muscle cell proliferation in COPD. Acta Biochim Biophys Sin (Shanghai) Dec 25(3):438–448. https://doi.org/10.3724/abbs.2022178
Wang E, Wang Y, Zhou S et al (2022) Identification of three hub genes related to the prognosis of idiopathic pulmonary fibrosis using bioinformatics analysis. Int J Med Sci 19(9):1417–1429. https://doi.org/10.7150/ijms.73305
Hu P, Xu Y, Jiang Y et al (2022) The mechanism of the imbalance between proliferation and ferroptosis in pulmonary artery smooth muscle cells based on the activation of SLC7A11. Article. Eur J Pharmacol Aug 5:928:175093. https://doi.org/10.1016/j.ejphar.2022.175093
Hoeper MM, Barberà JA, Channick RN et al (2009) Diagnosis, assessment, and treatment of non-pulmonary arterial hypertension pulmonary hypertension. J Am Coll Cardiol Jun 30(1 Suppl):85–96. https://doi.org/10.1016/j.jacc.2009.04.008
Ahmad A, Ahmad S, Malcolm KC et al (2013) Differential regulation of pulmonary vascular cell growth by hypoxia-inducible transcription factor-1alpha and hypoxia-inducible transcription factor-2alpha. Article. Am J Respir Cell Mol Biol Jul 49(1):78–85. https://doi.org/10.1165/rcmb.2012-0107OC
Yan L, Gao H, Li C, Han X, Qi X (2017) Effect of miR-23a on anoxia-induced phenotypic transformation of smooth muscle cells of rat pulmonary arteries and regulatory mechanism. Article. Oncol Lett Jan 13(1):89–98. https://doi.org/10.3892/ol.2016.5440
Wang GS, Qian GS, Zhou DS, Zhao JQ (2005) JAK-STAT signaling pathway in pulmonary arterial smooth muscle cells is activated by hypoxia. Article. Cell Biol Int Jul 29(7):598–603. https://doi.org/10.1016/j.cellbi.2005.03.014
Nowak WN, Deng J, Ruan XZ, Xu Q (2017) Reactive Oxygen species Generation and Atherosclerosis. Article. Arterioscler Thromb Vasc Biol May 37(5):41–52. https://doi.org/10.1161/ATVBAHA.117.309228
Zhang S, Bei Y, Huang Y et al (2022) Induction of ferroptosis promotes vascular smooth muscle cell phenotypic switching and aggravates neointimal hyperplasia in mice. Article. Mol Med Oct 3(1):121. https://doi.org/10.1186/s10020-022-00549-7
Song J, Chen Y, Chen Y et al (2023) Ferrostatin-1 blunts right ventricular hypertrophy and dysfunction in pulmonary arterial hypertension by suppressing the HMOX1/GSH signaling. J Cardiovasc Transl Res Aug 21. https://doi.org/10.1007/s12265-023-10423-4
Liao J, Xie SS, Deng Y et al (2023) PRDX6-mediated pulmonary artery endothelial cell ferroptosis contributes to monocrotaline-induced pulmonary hypertension. Article. Microvasc Res Mar 146:104471. https://doi.org/10.1016/j.mvr.2022.104471
Vogel NT, Annis J, Prisco S et al (2023) Ferroptosis promotes pulmonary hypertension. Preprint bioRxiv Dec 2. https://doi.org/10.1101/2023.01.19.524721
Lee S, Hwang N, Seok BG, Lee S, Lee SJ, Chung SW (2023) Autophagy mediates an amplification loop during ferroptosis. Rev Cell Death Disease Jul 25(7):464. https://doi.org/10.1038/s41419-023-05978-8
Kopp F, Mendell JT (2018) Functional Classification and Experimental Dissection of Long Noncoding RNAs. Cell. Jan 25. ;172(3):393–407. https://doi.org/10.1016/j.cell.2018.01.011
Liu P, Huang H, Qi X et al (2021) Hypoxia-Induced LncRNA-MIR210HG promotes Cancer Progression by inhibiting HIF-1α degradation in Ovarian Cancer. Front Oncol 11:701488. https://doi.org/10.3389/fonc.2021.701488
Hu XL, Huang XT, Zhang JN et al (2022) Long noncoding RNA MIR210HG is induced by hypoxia-inducible factor 1α and promotes cervical cancer progression. Am J Cancer Res 12(6):2783–2797
Tai YY, Yu Q, Tang Y et al (2024) Allele-specific control of rodent and human lncRNA KMT2E-AS1 promotes hypoxic endothelial pathology in pulmonary hypertension. Sci Transl Med Jan 10(729):eadd2029. https://doi.org/10.1126/scitranslmed.add2029
Zhou X, Zheng Y, Sun W et al (2021) D-mannose alleviates osteoarthritis progression by inhibiting chondrocyte ferroptosis in a HIF-2α-dependent manner. Cell Prolif Nov 54(11):e13134. https://doi.org/10.1111/cpr.13134
Chan XY, Volkova E, Eoh J et al (2021) HIF2A gain-of-function mutation modulates the stiffness of smooth muscle cells and compromises vascular mechanics. iScience Apr 23(4):102246. https://doi.org/10.1016/j.isci.2021.102246
Yerabolu D, Weiss A, Kojonazarov B et al (2021) Targeting Jak-Stat Signaling in Experimental Pulmonary Hypertension. Am J Respir Cell Mol Biol Jan 64(1):100–114. https://doi.org/10.1165/rcmb.2019-0431OC
Fu M, Luo F, Wang E et al (2021) Magnolol attenuates right ventricular hypertrophy and fibrosis in Hypoxia-Induced Pulmonary arterial hypertensive rats through inhibition of the JAK2/STAT3 signaling pathway. Front Pharmacol 12:755077. https://doi.org/10.3389/fphar.2021.755077
Huang H, Lin D, Hu L et al (2023) RNA binding protein quaking promotes hypoxia-induced smooth muscle reprogramming in Pulmonary Hypertension. Article. Am J Respir Cell Mol Biol Aug 69(2):159–171. https://doi.org/10.1165/rcmb.2022-0349OC
Acknowledgements
We sincerely thank Dr. Haijie Wang for her assistance during the preparation of this manuscript.
Funding
This research was supported by the fund for Natural Science Foundation of China (No.81970051), A sub-project of the Anhui Medical University National first-class undergraduate specialty construction program (clinical medicine), Young Jianghuai famous medical training project, Research Fund project of Anhui Medical University (2023xkj144), Excellent physician Training program of Anhui Medical University, Research Fund of Anhui Institute of translational medicine (2023zhyx-C40), Construction projects of key disciplines in Hefei (Occupational medicine), Health Research Project of Anhui Province (AHWJ2023A30009) and the Applied Medical Research Project of Hefei Health Commission (Hwk2021zd008, Hwk2022zd013).
Author information
Authors and Affiliations
Contributions
EW and BZ analyzed the data and wrote the first draft. LH and SZ designed the study, DZ and RW revised the submission. PL and RH directed the statistical analyses of the data. All authors have read and agreed to the submitted version of the manuscript.
Corresponding authors
Ethics declarations
Ethics committee approval
All animal experiments in this study were approved by the Animal Experimental Ethics Committee of Anhui Medical University and were performed in strict accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health.
Competing interests
The authors have declared that no competing interest exists.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, E., Zhang, B., Huang, L. et al. LncRNA MIR210HG promotes phenotype switching of pulmonary arterial smooth muscle cells through autophagy-dependent ferroptosis pathway. Apoptosis (2024). https://doi.org/10.1007/s10495-024-01963-4
Accepted:
Published:
DOI: https://doi.org/10.1007/s10495-024-01963-4