Abstract
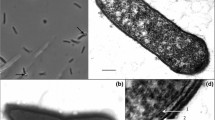
The rod-shaped and Gram-stain-negative bacterial strain 16FT, isolated from an air sample collected at King George Island, maritime Antarctica, was investigated to determine its taxonomic status. Strain 16FT is strictly aerobic, catalase positive, oxidase positive and non-motile. Strain 16FT hydrolyses casein, lecithin, Tween 20, 60 and 80, but not aesculin, gelatin and starch. Growth of strain 16FT is observed at 0–20 °C (optimum 10 °C), pH 5.0–8.0 (optimum pH 6.0), and in the presence of 0–2.0% NaCl (optimum 0.5%). The predominant menaquinone is MK-6, and the major fatty acids comprise anteiso-C15:0 and iso-C15:0. The major polar lipids are phosphatidylethanolamine, ornithine lipid OL2, unidentified phospholipid PL1 and the unidentified lipids L3 and L6 lacking functional groups. The DNA G + C content based on the draft genome sequence is 32.3 mol%. Sequence analysis of the 16S rRNA gene indicates the highest similarity to Kaistella palustris 3A10T (95.4%), Kaistella chaponensis Sa 1147-06 T (95.2%), Kaistella antarctica AT1013T (95.1%), Kaistella carnis NCTC 13525 T (95.1%) and below 95.0% to other species with validly published names. Phylogenetic analysis based on 16S rRNA gene and whole-genome sequences places strain 16FT in a distinct branch, indicating a separate lineage within the family Weeksellaceae. Based on the data from our polyphasic approach, 16FT represents a novel species of a new genus, for which the name Frigoriflavimonas asaccharolytica gen. nov, sp. nov. is proposed. The type strain is 16FT (= CCM 8975 T = CGMCC No.1.16844 T).


Similar content being viewed by others
References
Altenburger P, Kämpfer P, Makristathis A, Lubitz W, Busse H-J (1996) Classification of bacteria isolated from a medieval wall painting. J Biotechnol 47:39–52. https://doi.org/10.1016/0168-1656(96)01376-4
Arkin AP, Cottingham RW, Henry CS, Harris NL, Stevens RL et al (2018) KBase: The United States department of energy systems biology knowledgebase. Nat Biotechnol 36:566–569. https://doi.org/10.1038/nbt.4163
Aziz RK, Bartels D, Best A, DeJongh M, Disz T et al (2008) The RAST Server: rapid annotations using subsystems technology. BMC Genom 9:75. https://doi.org/10.1186/1471-2164-9-75
Bajerski F, Ganzert L, Mangelsdorf K, Padur L, Lipski A et al (2013) Chryseobacterium frigidisoli sp. nov., a psychrotolerant species of the family Flavobacteriaceae isolated from sandy permafrost from a glacier forefield. Int J Syst Evol Microbiol 63:2666–2671. https://doi.org/10.1099/ijs.0.046904-0
Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M et al (2012) SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19:455–477. https://doi.org/10.1089/cmb.2012.0021
Barrow GI, Feltham RKA (2003) Cowan and Steel’s Manual for Identification of Medical Bacteria. Cambridge University Press, Cambridge
Bennett S (2004) Solexa Ltd. Pharmacogenomics 5:433–438. https://doi.org/10.1517/14622416.5.4.433
Busse H-J, Auling G (1988) Polyamine pattern as a chemotaxonomic marker within the Proteobacteria. Syst Appl Microbiol 11:1–8. https://doi.org/10.1016/S0723-2020(88)80040-7
Busse H-J, Bunka S, Hensel A, Lubitz W (1997) Discrimination of members of the family Pasteurellaceae based on polyamine patterns. Int J Syst Evol Microbiol 47:698–708. https://doi.org/10.1099/00207713-47-3-698
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J et al (2009) BLAST+: architecture and applications. BMC Bioinformatics 10:421. https://doi.org/10.1186/1471-2105-10-421
Cavicchioli R, Charlton T, Ertan H, Mohd Omar S et al (2011) Biotechnological uses of enzymes from psychrophiles. Microb Biotechnol 4:449–460. https://doi.org/10.1111/j.1751-7915.2011.00258.x
Chen IMA, Chu K, Palaniappan K, Pillay M, Ratner A et al (2019) IMG/M vol 5.0: an integrated data management and comparative analysis system for microbial genomes and microbiomes. Nucleic Acids Res 47:D666–D677. https://doi.org/10.1093/nar/gky901
Chandrakar S, Gupta AK (2017) Antibiotic potential of endophytic actinomycetes of medicinal herbs against human pathogenic bacteria. Proc Natl Acad Sci India Biol Sci 87:905–915. https://doi.org/10.1007/s40011-015-0668-9
Cong B, Yin X, Deng A, Shen J, Yang H (2020) Diversity of cultivable microbes from soil of the Fildes Peninsula, Antarctica, and their potential application. Front Microbiol 11:570836. https://doi.org/10.3389/fmicb.2020.570836
Danilovich ME, Sánchez LA, Acosta F, Delgado OD (2018) Antarctic bioprospecting: in pursuit of microorganisms producing new antimicrobials and enzymes. Polar Biol 41:1417–1433. https://doi.org/10.1007/s00300-018-2295-4
Donegan NP, Cheung AL (2009) Regulation of the mazEF toxin-antitoxin module in Staphylococcus aureus and its impact on sigB expression. J Bacteriol 191:2795–2805. https://doi.org/10.1128/JB.01713-08
Farris JS (1972) Estimating phylogenetic trees from distance matrices. Am Nat 106:645–667. https://doi.org/10.1086/282802
Felsenstein J (1981) Evolutionary trees from DNA sequences: A maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Zool 20:406–416. https://doi.org/10.1093/sysbio/20.4.406
Garcia-Lopez M, Meier-Kolthoff JP, Tindall BJ, Gronow S, Woyke T et al (2019) Analysis of 1,000 type-strain genomes improves taxonomic classification of Bacteroidetes. Front Microbiol. https://doi.org/10.3389/fmicb.2019.02083
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P et al (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Hahnke RL, Meier-Kolthoff JP, García-López M, Mukherjee S, Huntemann M et al (2016) Genome-based taxonomic classification of Bacteroidetes. Front Microbiol 7:2003. https://doi.org/10.3389/fmicb.2016.02003
Huntemann M, Ivanova NN, Mavromatis K, James Tripp H, Paez-Espino D et al (2015) The standard operating procedure of the DOE-JGI Microbial Genome Annotation Pipeline (MGAP vol 4). Stand Genomic Sci 10:4–9. https://doi.org/10.1186/s40793-015-0077-y
Kämpfer P, Kroppenstedt RM (1996) Numerical analysis of fatty acid patterns of coryneform bacteria and related taxa. Can J Microbiol 42:989–1005. https://doi.org/10.1139/m96-128
Kämpfer P, Vaneechoutte M, Lodders B, De Baere T, Avesani V, et al. (2009) Description of Chryseobacterium anthropic sp. nov. to accommodate clinical isolates biochemically similar to Kaistella koreensis and Chryseobacterium haifense, proposal to reclassify Kaistella koreensis as Chryseobacterium koreense comb. nov. and emended description of the genus Chryseobacterium. Int J Syst Evol Microbiol, 59:2421–2428.https://doi.org/10.1099/ijs.0.008250-0
Kämpfer P, Poppel MT, Wilharm G, Busse H-J, McInroy JA, Glaeser SP (2014) Chryseobacterium gallinarum sp. nov., isolated from a chicken, and Chryseobacterium contaminans sp. nov., isolated as a contaminant from a rhizosphere sample. Int J Syst Evol Microbiol 64:1419–1427. https://doi.org/10.1099/ijs.0.058933-0
Kämpfer P, Busse H-J, McInroy JA, Glaeser SP (2015a) Chryseobacterium sediminis sp. nov., isolated from a river sediment. Int J Syst Evol Microbiol. 65:4019–4024. https://doi.org/10.1099/ijsem.0.000527
Kämpfer P, Busse H-J, McInroy JA, Glaeser SP (2015b) Chryseobacterium arachidiradicis sp. nov., isolated from the geocarposphere (soil around the peanut) of very immature peanuts (Arachis hypogaea). Int J Syst Evol Microbiol 65:2179–2186. https://doi.org/10.1099/ijs.0.000237
Kim KK, Kim MK, Lim JH, Park HY, Lee ST (2005) Transfer of Chryseobacterium meningosepticum and Chryseobacterium miricola to Elizabethkingia gen. nov. as Elizabethkingia meningoseptica comb. nov. and Elizabethkingia miricola comb. nov. Int J Syst Evol Microbiol 55:1287–1293. https://doi.org/10.1099/ijs.0.63541-0
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Kreft L, Botzki A, Coppens F, Vandepoele K, Van Bel M (2017) PhyD3: a phylogenetic tree viewer with extended phyloXML support for functional genomics data visualization. Bioinformatics 33:2946–2947. https://doi.org/10.1093/bioinformatics/btx324
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kyrpides NC, Hugenholtz P, Eisen JA, Woyke T, Göker M et al (2014) Genomic encyclopedia of bacteria and archaea: sequencing a myriad of type strains. PLoS Biol 12(8):e1001920
Lagesen K, Hallin EP, Rødland A, Stærfeldt HH et al (2007) RNAmmer: consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res 35:3100–3108. https://doi.org/10.1093/nar/gkm160
Lambrechts S, Willems A, Tahon G (2019) Uncovering the uncultivated majority in Antarctic soils: toward a synergistic approach. Front Microbiol 10:242. https://doi.org/10.3389/fmicb.2019.00242
Lamilla C, Pavez M, Santos A, Hermosilla A, Llanquinao V, Barrientos L (2017) Bioprospecting for extracellular enzymes from culturable Actinobacteria from the South Shetland Islands, Antarctica. Polar Biol 40:719–726. https://doi.org/10.1007/s00300-016-1977-z
Lefort V, Desper R, Gascuel O (2015) FastME 2.0: A comprehensive, accurate, and fast distance-based phylogeny inference program. Mol Biol Evol 32:2798–2800. https://doi.org/10.1093/molbev/msv150
Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14:60. https://doi.org/10.1186/1471-2105-14-60
Meier-Kolthoff JP, Göker M (2019) TYGS is an automated high-throughput platform for state-of-the-art genomebased taxonomy. Nat Commun 10:2182. https://doi.org/10.1038/s41467-019-10210-3
Montero-Calasanz M, Göker M, Rohde M, Spröer C, Schumann P et al (2013) Chryseobacterium hispalense sp. nov., a plant-growth-promoting bacterium isolated from a rainwater pond in an olive plant nursery, and emended descriptions of Chryseobacterium defluvii, Chryseobacterium indologenes, Chryseobacterium wanjuense and Chryseobacterium gregarium. Int J Syst Evol Microbiol 63:4386–4395. https://doi.org/10.1099/ijs.0.052456-0
Montero-Calasanz M, Göker M, Rohde M, Spröer C, Schumann P et al (2014) Chryseobacterium oleae sp. nov., an efficient plant growth promoting bacterium in the rooting induction of olive tree (Olea europaea L.) cuttings and emended descriptions of the genus Chryseobacterium, C. daecheongense, C. gambrini, C. gleum, C. joostei, C. jejuense, C. luteum, C. shigense, C. taiwanense, C. ureilyticum and C. vrystaatense. Syst Appl Microbiol 37:342–350. https://doi.org/10.1016/j.syapm.2014.04.004
Nicholson AC, Gulvik CA, Whitney AM, Humrighouse BW, Bell ME et al (2019) Division of the genus Chryseobacterium: Observation of discontinuities in amino acid identity values, a possible consequence of major extinction events, guides transfer of nine species to the genus Epilithonimonas, eleven species to the genus Kaistella. Int J Syst Evol Microbiol 70:4432–4450. https://doi.org/10.1099/ijsem.0.003935
Núñez-Montero K, Barrientos L (2018) Advances in Antarctic research for antimicrobial discovery: a comprehensive narrative review of bacteria from Antarctic environments as potential sources of novel antibiotic compounds against human pathogens and microorganisms of industrial importance. Antibiotics 7:90. https://doi.org/10.3390/antibiotics7040090
Parks DH, Imelfort M, Skennerton CT, Hugenholtz P, Tyson GW (2015) CheckM assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res 25:1043–1055. https://doi.org/10.1101/gr.186072.114
Ondov BD, Treangen TJ, Melsted P, Mallonee AB, Bergman NH et al (2016) Mash: fast genome and metagenome distance estimation using MinHash. Genome Biol 17:1–14. https://doi.org/10.1186/s13059-016-0997-x
Pires C, Carvalho MF, De Marco P, Magan N, Castro PML (2010) Chryseobacterium palustre sp. nov. and Chryseobacterium humi sp. nov., isolated from industrially contaminated sediments. Int J Syst Evol Microbiol 60:402–407. https://doi.org/10.1099/ijs.0.010348-0
Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T et al (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:D590–D596. https://doi.org/10.1093/nar/gks1219
Rizzo C, Lo Giudice A (2020) The Variety and inscrutability of polar environments as a resource of biotechnologically relevant molecules. Microorganisms 8:1422. https://doi.org/10.3390/microorganisms8091422
Rodriguez-R L, Konstantinidis K (2016) The enveomics collection: a toolbox for specialized analyses of microbial genomes and metagenomes. Peer J Prepr PeerJ. https://doi.org/10.7287/peerj.preprints.1900v1
Roldán DM, Kyrpides N, Woyke T, Shapiro N, Whitman WB et al (2020) Hymenobacter artigasi sp. nov., isolated from air sampling in maritime Antarctica. Int J Syst Evol Microbiol 70:4935–4941. https://doi.org/10.1099/ijsem.0.004362
Saitou N, Nei M (1987) The Neighbor-joining Method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Stolz A, Busse H-J, Kämpfer P (2007) Pseudomonas knackmussii sp. nov. Int J Syst Evol Microbiol 57:572–576. https://doi.org/10.1099/ijs.0.64761-0
Tindall BJ (1990) Lipid composition of Halobacterium lacusprofundi. FEMS Microbiol Lett 66:199–202. https://doi.org/10.1111/j.1574-6968.1990.tb03996.x
Tindall BJ (1990) A comparative study of the lipid composition of Halobacterium saccharovorum from various sources. Syst Appl Microbiol 413:128–130. https://doi.org/10.1016/S0723-2020(11)80158-X
Tindall BJ, Sikorski J, Smibert RA, Krieg NR (2014) Phenotypic characterization and the principles of comparative systematics. In: Reddy CA, Beveridge TJ, Breznak JA, Marzluf GA, Schmidt TM et al (eds) Methods for General and Molecular Microbiology, 3rd edn. ASM, Washington DC, pp 330–393
Yamanaka K, Fang L, Inouye M (1998) The CspA family in Escherichia coli: multiple gene duplication for stress adaptation. Mol Microbiol 27:247–255. https://doi.org/10.1046/j.1365-2958.1998.00683.x
Yassin AF, Hupfer H, Siering C, Busse HJ (2010) Chryseobacterium treverense sp. nov., isolated from a human clinical source. Int J Syst Evol Microbiol 60:1993–1998. https://doi.org/10.1099/ijs.0.017327-0
Yoon SH, Ha SM, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Zhang R-G, Tan X, Liang Y, Meng T-Y, Liang H-Z et al (2014) Description of Chishuiella changwenlii gen. nov., sp. nov., isolated from freshwater, and transfer of Wautersiella falsenii to the genus Empedobacter as Empedobacter falsenii comb. nov. Int J Syst Evol Microbiol 64:2723–2728. https://doi.org/10.1099/ijs.0.063115-0
Acknowledgements
To the Uruguayan Antarctic Institute and the staff of the Artigas Antarctic Scientific Base during the summer 2018 campaign. RJM is PEDECIBA and ANII researcher.We are grateful to Professor Aharon Oren (The Hebrew University of Jerusalem, Israel) for the Latin construction of the taxa names.
Funding
The work conducted by the U.S. Department of Energy Joint Genome Institute, a DOE Office of Science User Facility, was supported by the Office of Science of the U.S. Department of Energy under contract number DE-AC02-05CH11231.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Availability of data and material
The GenBank/EMBL/DDBJ accession number for the 16S rRNA gene sequence for strain 16FT is MN007167. The Whole Genome Shotgun project has been deposited at DDBJ/ENA/GenBank under the accession JABSNO000000000. The version described in this paper is version JABSNO000000000.1.
Consent for publication
All authors consent to the publication of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Menes, R.J., Machin, E.V., Roldán, D.M. et al. Frigoriflavimonas asaccharolytica gen. nov., sp. nov., a novel psychrophilic esterase and protease producing bacterium isolated from Antarctica. Antonie van Leeuwenhoek 114, 1991–2002 (2021). https://doi.org/10.1007/s10482-021-01656-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-021-01656-x