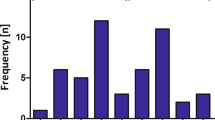
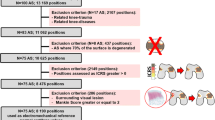
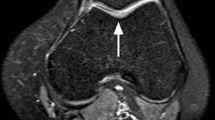
Abstract
Many arthroscopic tools developed for knee joint assessment are contact-based, which is challenging for in vivo application in narrow joint spaces. Second harmonic generation (SHG) laser imaging is a non-invasive and non-contact method, thus presenting an attractive alternative. However, the association between SHG-based measures and cartilage quality has not been established systematically. Here, we investigated the feasibility of using image-based measures derived from SHG microscopy for objective evaluation of cartilage quality as assessed by mechanical testing. Human tibial plateaus harvested from nine patients were used. Cartilage mechanical properties were determined using indentation stiffness (Einst) and streaming potential-based quantitative parameters (QP). The correspondence of the cartilage electromechanical properties (Einst and QP) and the image-based measures derived from SHG imaging, tissue thickness and cell viability were evaluated using correlation and logistic regression analyses. The SHG-related parameters included the newly developed volumetric fraction of organised collagenous network (Φcol) and the coefficient of variation of the SHG intensity (CVSHG). We found that Φcol correlated strongly with Einst and QP (ρ = 0.97 and − 0.89, respectively). CVSHG also correlated, albeit weakly, with QP and Einst, (|ρ| = 0.52–0.58). Einst and Φcol were the most sensitive predictors of cartilage quality whereas CVSHG only showed moderate sensitivity. Cell viability and tissue thickness, often used as measures of cartilage health, predicted the cartilage quality poorly. We present a simple, objective, yet effective image-based approach for assessment of cartilage quality. Φcol correlated strongly with electromechanical properties of cartilage and could fuel the continuous development of SHG-based arthroscopy.





Similar content being viewed by others
References
Abusara, Z., S. H. J. Andrews, M. V. Kossel, and W. Herzog. Menisci protect chondrocytes from load-induced injury. Sci. Rep. 8:14150, 2018.
Abusara, Z., I. Haider, E. K. Moo, S. Miller, S. Timmermann, and W. Herzog. Chondrocyte morphology as an indicator of collagen network integrity. Connect Tissue Res. 63:319–328, 2022.
Anderson, A. S., and R. F. Loeser. Why is osteoarthritis an age-related disease? Best Pract. Res. Clin. Rheumatol. 24:15, 2010.
Bae, W. C., M. M. Temple, D. Amiel, R. D. Coutts, G. G. Niederauer, and R. L. Sah. Indentation testing of human cartilage: sensitivity to articular surface degeneration. Arthritis Rheumatism. 48:3382–3394, 2003.
Baskey, S. J., M. Andreana, E. Lanteigne, A. Ridsdale, A. Stolow, and M. E. Schweitzer. Pre-clinical translation of second harmonic microscopy of meniscal and articular cartilage using a prototype nonlinear microendoscope. IEEE J. Transl. Eng. Health Med. 7:1–11, 2019.
Brama, P. A., A. Barneveld, D. Karssenberg, G. P. Van Kampen, and P. R. Van Weeren. The application of an indenter system to measure structural properties of articular cartilage in the horse. Suitability of the instrument and correlation with biochemical data. J. Vet. Med. Ser. A. 48(4):213–21, 2001.
Brockbank, K. G. M., W. R. MacLellan, J. Xie, S. F. Hamm-Alvarez, Z. Z. Chen, and K. Schenke-Layland. Quantitative second harmonic generation imaging of cartilage damage. Cell Tissue Bank. 9:299–307, 2008.
Bugbee, W. D., A. L. Pallante-Kichura, S. Görtz, D. Amiel, and R. Sah. Osteochondral allograft transplantation in cartilage repair: graft storage paradigm, translational models, and clinical applications. J. Orthop. Res. 34:31–38, 2016.
Changoor, A., J. P. Coutu, M. Garon, E. Quenneville, M. B. Hurtig, and M. D. Buschmann. Streaming potential-based arthroscopic device is sensitive to cartilage changes immediately post-impact in an equine cartilage injury model. J. Biomech. Eng. 133(6):061005, 2011.
Chen, X., O. Nadiarynkh, S. Plotnikov, and P. J. Campagnola. Second harmonic generation microscopy for quantitative analysis of collagen fibrillar structure. Nat. Protocols. 7:654–669, 2012.
Cicchi, R., D. Kapsokalyvas, V. De Giorgi, V. Maio, A. Van Wiechen, D. Massi, T. Lotti, and F. S. Pavone. Scoring of collagen organization in healthy and diseased human dermis by multiphoton microscopy. J. Biophoton. 3:34–43, 2010.
Dacre, J. E., D. L. Scott, J. A. Da Silva, G. Welsh, and E. C. Huskisson. Joint space in radiologically normal knees. Br. J. Rheumatol. 30:426–428, 1991.
Ebrahimi, M., S. Ojanen, A. Mohammadi, M. A. Finnilä, A. Joukainen, H. Kröger, S. Saarakkala, R. K. Korhonen, and P. Tanska. Elastic, viscoelastic and fibril-reinforced poroelastic material properties of healthy and osteoarthritic human tibial cartilage. Ann. Biomed. Eng. 47:953–966, 2019.
Ebrahimi, M., M. J. Turunen, M. A. Finnilä, A. Joukainen, H. Kröger, S. Saarakkala, R. K. Korhonen, and P. Tanska. Structure-function relationships of healthy and osteoarthritic human tibial cartilage: experimental and numerical investigation. Ann. Biomed. Eng. 48:2887–2900, 2020.
Favre, J., J. C. Erhart-Hledik, K. Blazek, B. Fasel, G. E. Gold, and T. P. Andriacchi. Anatomically standardized maps reveal distinct patterns of cartilage thickness with increasing severity of medial compartment knee osteoarthritis. J. Orthop. Res. 35:2442–2451, 2017.
Franz, T., E. M. Hasler, R. Hagg, C. Weiler, R. P. Jakob, and P. Mainil-Varlet. In situ compressive stiffness, biochemical composition, and structural integrity of articular cartilage of the human knee joint. Osteoarthr. Cartil. 9:582–592, 2001.
Gelse, K., A. Olk, S. Eichhorn, B. Swoboda, M. Schoene, and K. Raum. Quantitative ultrasound biomicroscopy for the analysis of healthy and repair cartilage tissue. Eur. Cell Mater. 19:58–71, 2010.
Hadjab, I., S. Sim, S. S. Karhula, S. Kauppinen, M. Garon, E. Quenneville, P. Lavigne, P. P. Lehenkari, S. Saarakkala, and M. D. Buschmann. Electromechanical properties of human osteoarthritic and asymptomatic articular cartilage are sensitive and early detectors of degeneration. Osteoarthr. Cartil. 26:405–413, 2018.
Hui Mingalone, C. K., Z. Liu, J. M. Hollander, K. D. Garvey, A. L. Gibson, R. E. Banks, M. Zhang, T. E. McAlindon, H. C. Nielsen, I. Georgakoudi, and L. Zeng. Bioluminescence and second harmonic generation imaging reveal dynamic changes in the inflammatory and collagen landscape in early osteoarthritis. Lab. Invest. 98:656–669, 2018.
Huynh, R. N., G. Nehmetallah, and C. B. Raub. Noninvasive assessment of articular cartilage surface damage using reflected polarized light microscopy. J. Biomed. Opt. 22:65001, 2017.
Huynh, R. N., G. Nehmetallah, and C. B. Raub. Mueller matrix polarimetry and polar decomposition of articular cartilage imaged in reflectance. Biomed. Opt. Express. 12:5160–5178, 2021.
Huynh, R. N., B. Pesante, G. Nehmetallah, and C. B. Raub. Polarized reflectance from articular cartilage depends upon superficial zone collagen network microstructure. Biomed. Opt. Express. 10:5518–5534, 2019.
Ishihara, M., M. Sato, N. Kaneshiro, G. Mitani, S. Sato, J. Mochida, and M. Kikuchi. Development of a diagnostic system for osteoarthritis using a photoacoustic measurement method. Lasers Surg. Med. 38:249–255, 2006.
Johansson, A., T. Sundqvist, J.-H. Kuiper, and P. Å. Öberg. A spectroscopic approach to imaging and quantification of cartilage lesions in human knee joints. Phys. Med. Biol. 56:1865, 2011.
Jurvelin, J. S., T. Räsänen, P. Kolmonen, and T. Lyyra. Comparison of optical, needle probe and ultrasonic techniques for the measurement of articular cartilage thickness. J. Biomech. 28:231–235, 1995.
Karjalainen, K., P. Tanska, S. C. Sibole, S. Mikkonen, W. Herzog, R. Korhonen, and E. K. Moo. Effect of cells on spatial quantification of proteoglycans in articular cartilage of small animals. Connect Tissue Res. 63:603–614, 2022.
Kashimura, Y., R. Matsuda, N. Yamato, and M. Hashimoto. Second-harmonic generation arthroscope with integrated femtosecond Yb fiber laser. , 2022.at <https://opg.optica.org/abstract.cfm?uri=CLEOPR-2022-P_CM15_07>
Kiyomatsu, H., Y. Oshima, T. Saitou, T. Miyazaki, A. Hikita, H. Miura, T. Iimura, and T. Imamura. Quantitative SHG imaging in osteoarthritis model mice, implying a diagnostic application. Biomed. Opt. Express. 6:405–420, 2015.
Kleemann, R. U., D. Krocker, A. Cedraro, J. Tuischer, and G. N. Duda. Altered cartilage mechanics and histology in knee osteoarthritis: relation to clinical assessment (ICRS Grade). Osteoarthr. Cartil. 13:958–963, 2005.
Kumar, R., K. M. Grønhaug, C. L. Davies, J. O. Drogset, and M. B. Lilledahl. Nonlinear optical microscopy of early stage (ICRS Grade-I) osteoarthritic human cartilage. Biomed. Opt. Express. 6:1895–1903, 2015.
Laasanen, M. S., J. Töyräs, J. Hirvonen, S. Saarakkala, R. K. Korhonen, M. T. Nieminen, I. Kiviranta, and J. S. Jurvelin. Novel mechano-acoustic technique and instrument for diagnosis of cartilage degeneration. Physiol. Meas. 23:491–503, 2002.
Lyyra, T., I. Kiviranta, U. Väätäinen, H. J. Helminen, and J. S. Jurvelin. In vivo characterization of indentation stiffness of articular cartilage in the normal human knee. J. Biomed. Mater. Res. 48:482–487, 1999.
Mäkelä, J. T. A., Z. S. Rezaeian, S. Mikkonen, R. Madden, S.-K. Han, J. S. Jurvelin, W. Herzog, and R. K. Korhonen. Site-dependent changes in structure and function of lapine articular cartilage 4 weeks after anterior cruciate ligament transection. Osteoarthr. Cartil. 22:869–878, 2014.
Mansfield, J. C., V. Mandalia, A. Toms, C. P. Winlove, and S. Brasselet. Collagen reorganization in cartilage under strain probed by polarization sensitive second harmonic generation microscopy. J. R. Soc. Interface. 16:20180611, 2019.
Moo, E. K., Z. Abusara, N. A. Abu Osman, B. Pingguan-Murphy, and W. Herzog. Dual photon excitation microscopy and image threshold segmentation in live cell imaging during compression testing. J. Biomech. 46:2024–2031, 2013.
Moo, E. K., Y. Al-Saffar, T. Le, R. A. Seerattan, B. Pingguan-Murphy, R. K. Korhonen, and W. Herzog. Deformation behaviors and mechanical impairments of tissue cracks in immature and mature cartilages. J. Orthop. Res. 40(9):2103–12, 2021.
Moo, E. K., M. Amrein, M. Epstein, M. Duvall, N. A. Abu Osman, B. Pingguan-Murphy, and W. Herzog. The properties of chondrocyte membrane reservoirs and their role in impact-induced cell death. Biophys. J. 105:1590–1600, 2013.
Moo, E. K., M. Ebrahimi, S. C. Sibole, P. Tanska, and R. K. Korhonen. The intrinsic quality of proteoglycans, but not collagen fibres, degrades in osteoarthritic cartilage. Acta Biomater. 153:178–189, 2022.
Moo, E. K., S. K. Han, S. Federico, S. C. Sibole, A. Jinha, N. A. Abu Osman, B. Pingguan-Murphy, and W. Herzog. Extracellular matrix integrity affects the mechanical behaviour of in-situ chondrocytes under compression. J. Biomech. 47:1004–1013, 2014.
Moo, E. K., and W. Herzog. Unfolding of membrane ruffles of in situ chondrocytes under compressive loads. J. Orthop. Res. 35:304–310, 2017.
Moo, E. K., S. C. Sibole, S. Federico, R. K. Korhonen, and W. Herzog. Microscale investigation of the anisotropic swelling of cartilage tissue and cells in response to hypo-osmotic challenges. J. Orthop. Res. 2023. https://doi.org/10.1002/jor.25657.
Moo, E. K., S. C. Sibole, S. K. Han, and W. Herzog. Three-dimensional micro-scale strain mapping in living biological soft tissues. Acta Biomater. 70:260–269, 2018.
Moo, E. K., P. Tanska, S. Federico, Y. Al-Saffar, W. Herzog, and R. K. Korhonen. Collagen fibres determine the crack morphology in articular cartilage. Acta Biomater. 2021. https://doi.org/10.1016/j.actbio.2021.03.031.
Niederauer, G. G., G. M. Niederauer, L. C. Cullen, K. A. Athanasiou, J. B. Thomas, and M. Q. Niederauer. Correlation of cartilage stiffness to thickness and level of degeneration using a handheld indentation probe. Ann. Biomed. Eng. 32:352–359, 2004.
Novakofski, K. D., S. L. Pownder, M. F. Koff, R. M. Williams, H. G. Potter, and L. A. Fortier. High-resolution methods for diagnosing cartilage damage in vivo. Cartilage. 7:39–51, 2016.
Novakofski, K. D., R. M. Williams, L. A. Fortier, H. O. Mohammed, W. R. Zipfel, and L. J. Bonassar. Identification of cartilage injury using quantitative multiphoton microscopy. Osteoarthr. Cartil. 22:355–362, 2014.
Park, S. H., J. M. Goo, and C.-H. Jo. Receiver operating characteristic (ROC) curve: practical review for radiologists. Korean J. Radiol. 5:11–18, 2004.
Pedregosa, F., G. Varoquaux, A. Gramfort, V. Michel, B. Thirion, O. Grisel, M. Blondel, P. Prettenhofer, R. Weiss, V. Dubourg, J. Vanderplas, A. Passos, D. Cournapeau, M. Brucher, M. Perrot, and É. Duchesnay. Scikit-learn: machine learning in Python. J. Mach. Learn. Res. 12:2825–2830, 2011.
Plotnikov, S. V., A. M. Kenny, S. J. Walsh, B. Zubrowski, C. Joseph, V. L. Scranton, G. A. Kuchel, D. Dauser, M. Xu, C. C. Pilbeam, D. J. Adams, R. P. Dougherty, P. J. Campagnola, and W. A. Mohler. Measurement of muscle disease by quantitative second-harmonic generation imaging. J. Biomed. Opt 13:044018-044018–11, 2008.
Saarakkala, S., P. Julkunen, P. Kiviranta, J. Mäkitalo, J. S. Jurvelin, and R. K. Korhonen. Depth-wise progression of osteoarthritis in human articular cartilage: investigation of composition, structure and biomechanics. Osteoarthr. Cartil. 18:73–81, 2010.
Saarakkala, S., M. S. Laasanen, J. S. Jurvelin, and J. Töyräs. Quantitative ultrasound imaging detects degenerative changes in articular cartilage surface and subchondral bone. Phys. Med. Biol. 51:5333, 2006.
Schenke-Layland, K., N. Madershahian, I. Riemann, B. Starcher, K.-J. Halbhuber, K. König, and U. A. Stock. Impact of cryopreservation on extracellular matrix structures of heart valve leaflets. Ann. Thorac. Surg. 81:918–926, 2006.
Sherlock, B. E., J. Chen, J. C. Mansfield, E. Green, and C. P. Winlove. Biophotonic tools for probing extracellular matrix mechanics. Matrix Biol. Plus. 2021. https://doi.org/10.1016/j.mbplus.2021.100093.
Sibole, S. C., E. K. Moo, S. Federico, and W. Herzog. The protective function of directed asymmetry in the pericellular matrix enveloping chondrocytes. Ann. Biomed. Eng. 50:39–55, 2022.
Sim, S., A. Chevrier, M. Garon, E. Quenneville, P. Lavigne, A. Yaroshinsky, C. D. Hoemann, and M. D. Buschmann. Electromechanical probe and automated indentation maps are sensitive techniques in assessing early degenerated human articular cartilage. J. Orthopaedic Res. 35:858–867, 2017.
Sim, S., A. Chevrier, M. Garon, E. Quenneville, A. Yaroshinsky, C. D. Hoemann, and M. D. Buschmann. Non-destructive electromechanical assessment (Arthro-BST) of human articular cartilage correlates with histological scores and biomechanical properties. Osteoarthr. Cartil. 22:1926–1935, 2014.
Sim, S., I. Hadjab, M. Garon, E. Quenneville, P. Lavigne, and M. D. Buschmann. Development of an electromechanical grade to assess human knee articular cartilage quality. Ann. Biomed. Eng. 45:2410–2421, 2017.
Spahn, G., H. M. Klinger, M. Baums, U. Pinkepank, and G. O. Hofmann. Reliability in arthroscopic grading of cartilage lesions: results of a prospective blinded study for evaluation of inter-observer reliability. Arch. Orthop. Trauma Surg. 131:377–381, 2011.
Stoller, P. C., P. M. Celliers, K. M. Reiser, and A. M. Rubenchik. Imaging collagen orientation using polarization-modulated second harmonic generation. , 2002.
Wieland, H. A., M. Michaelis, B. J. Kirschbaum, and K. A. Rudolphi. Osteoarthritis—an untreatable disease? Nat. Rev. Drug Discov. 4:331–344, 2005.
Acknowledgements
We would like to express our gratitude to Ms. Christiana Choi who helped in tissue procurement from arthroplasty procedure, the PLC, Surgical Suite, and Mr. Azim Jinha who provided technical support during data analysis.
Funding
This study was supported by the Joint Transplantation Program at the McCaig Institute for Bone and Joint Heath, University of Calgary, the Calgary Health Trust, the Carleton University internal start-up research fund (186725), the Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery grant (funding reference no: DGECR-2023-00346), The Killam Foundation, The Canadian Institutes of Health Research, and the Nigg Chair for Mobility and Longevity.
Author information
Authors and Affiliations
Contributions
(1) The conception and design of the study, or acquisition of data, or analysis and interpretation of the data: ZA, EKM, IH, CT, SM, ST and WH. (2) Drafting the article or revising it critically for important intellectual content: ZA, EKM, IH, CT, SM, ST and WH. (3) Final approval of the version to be submitted: ZA, EKM, IH, CT, SM, ST and WH.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Associate Editor Michael S. Detamore oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abusara, Z., Moo, E.K., Haider, I. et al. Functional Assessment of Human Articular Cartilage Using Second Harmonic Generation (SHG) Imaging: A Feasibility Study. Ann Biomed Eng 52, 1009–1020 (2024). https://doi.org/10.1007/s10439-023-03437-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-023-03437-1