Abstract
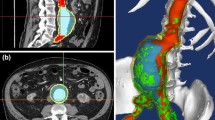
Elective repair of abdominal aortic aneurysm (AAA) is warranted when the risk of rupture exceeds that of surgery, and is mostly based on the AAA size as a crude rupture predictor. A methodology based on biomechanical considerations for a reliable patient-specific prediction of AAA risk of rupture is presented. Fluid–structure interaction (FSI) simulations conducted in models reconstructed from CT scans of patients who had contained ruptured AAA (rAAA) predicted the rupture location based on mapping of the stresses developing within the aneurysmal wall, additionally showing that a smaller rAAA presented a higher rupture risk. By providing refined means to estimate the risk of rupture, the methodology may have a major impact on diagnostics and treatment of AAA patients.






Similar content being viewed by others
References
Bengtsson, H., and D. Bergqvist. Ruptured abdominal aortic aneurysm: a population-based study. J. Vasc. Surg. 18:74–80, 1993.
Bettermann, K., J. F. Toole. Diagnostic evaluation and medical management of patients with ischemic cerebrovascular disease. In: Vascular Surgery, edited by R. Rutherford. Philadelphia: Elsevier, 2005, pp. 1904–1905.
Bluestein, D., K. Dumont, M. De Beule, J. Ricotta, P. Impellizzeri, B. Verhegghe, and P. Verdonck. Intraluminal thrombus and risk of rupture in patient specific abdominal aortic aneurysm—FSI modelling. Comput. Methods Biomech. Biomed. Eng. 12:73–81, 2008.
Bluestein, D., Y. Alemu, I. Avrahami, M. Gharib, K. Dumont, J. J. Ricotta, and S. Einav. Influence of microcalcifications on vulnerable plaque mechanics using FSI modeling. J. Biomech. 41:1111–1118, 2008.
Bonert, M., R. L. Leask, J. Butany, C. R. Ethier, J. G. Myers, K. W. Johnston, and M. Ojha. The relationship between wall shear stress distributions and intimal thickening in the human abdominal aorta. Biomed. Eng. Online 2:18, 2003.
Boyce, M. C., and E. M. Arruda. Constitutive models of rubber elasticity: a review. Rubber Chem. Technol. 73:504–523, 2000.
Brown, L. C., and J. T. Powell. Risk factors for aneurysm rupture in patients kept under ultrasound surveillance. UK small aneurysm trial participants. Ann. Surg. 230:289–296, 1999.
Cheng, C. P., R. J. Herfkens, and C. A. Taylor. Comparison of abdominal aortic hemodynamics between men and women at rest and during lower limb exercise. J. Vasc. Surg. 37:118–123, 2003.
Chuong, C. J., and Y. C. Fung. On residual stresses in arteries. J. Biomech. Eng. 108:189–192, 1986.
Cronenwett, J. L., T. F. Murphy, G. B. Zelenock, W. M. Whitehouse, S. M. Lindenauer, L. M. Graham, L. E. Quint, T. M. Silver, and J. C. Stanley. Actuarial analysis of variables associated with rupture of small abdominal aortic-aneurysms. Surgery 98:472–483, 1985.
de Putter, S., B. J. Wolters, M. C. Rutten, M. Breeuwer, F. A. Gerritsen, and F. N. van de Vosse. Patient-specific initial wall stress in abdominal aortic aneurysms with a backward incremental method. J. Biomech. 40:1081–1090, 2006.
Di Martino, E. S., and D. A. Vorp. Effect of variation in intraluminal thrombus constitutive properties on abdominal aortic aneurysm wall stress. Ann. Biomed. Eng. 31:804–809, 2003.
Di Martino, E., S. Mantero, F. Inzoli, G. Melissano, D. Astore, R. Chiesa, and R. Fumero. Biomechanics of abdominal aortic aneurysm in the presence of endoluminal thrombus: experimental characterisation and structural static computational analysis. Eur. J. Vasc. Endovasc. Surg. 15:290–299, 1998.
Di Martino, E. S., G. Guadagni, A. Fumero, G. Ballerini, R. Spirito, P. Biglioli, and A. Redaelli. Fluid-structure interaction within realistic three-dimensional models of the aneurysmatic aorta as a guidance to assess the risk of rupture of the aneurysm. Med. Eng. Phys. 23:647–655, 2001.
Di Martino, E. S., A. Bohra, J. P. Vande Geest, N. Gupta, M. S. Makaroun, and D. A. Vorp. Biomechanical properties of ruptured versus electively repaired abdominal aortic aneurysm wall tissue. J. Vasc. Surg. 43:570–576, 2006.
Fillinger, M. The long-term relationship of wall stress to the natural history of abdominal aortic aneurysms (finite element analysis and other methods). Ann. N. Y. Acad. Sci. 1085:22–28, 2006.
Fillinger, M. F., M. L. Raghavan, S. P. Marra, J. L. Cronenwett, and F. E. Kennedy. In vivo analysis of mechanical wall stress and abdominal aortic aneurysm rupture risk. J. Vasc. Surg. 36:589–597, 2002.
Fillinger, M. F., S. P. Marra, M. L. Raghavan, and F. E. Kennedy. Prediction of rupture risk in abdominal aortic aneurysm during observation: wall stress versus diameter. J. Vasc. Surg. 37:724–732, 2003.
Finol, E. A., and C. H. Amon. Flow dynamics in anatomical models of abdominal aortic aneurysms: computational analysis of pulsatile flow. Acta Cient. Venez. 54:43–49, 2003.
Greve, J. M., A. S. Les, B. T. Tang, M. T. Draney Blomme, N. M. Wilson, R. L. Dalman, N. J. Pelc, and C. A. Taylor. Allometric scaling of wall shear stress from mice to humans: quantification using cine phase-contrast MRI and computational fluid dynamics. Am. J. Physiol. Heart Circ. Physiol. 291:H1700–H1708, 2006.
He, C. M., and M. R. Roach. The composition and mechanical properties of abdominal aortic aneurysms. J. Vasc. Surg. 20:6–13, 1994.
Holzapfel, G. A., and T. C. Gasser. A viscoelastic model for fiber-reinforced composites at finite strains: continuum basis, computational aspects and applications. Comput. Method Appl. Mech. 190:4379–4403, 2001.
Holzapfel, G. A., and T. C. Gasser. Computational stress-deformation analysis of arterial walls including high-pressure response. Int. J. Cardiol. 116:78–85, 2007.
Holzapfel, G. A., and H. W. Weizsacker. Biomechanical behavior of the arterial wall and its numerical characterization. Comput. Biol. Med. 28:377–392, 1998.
Holzapfel, G. A., T. C. Gasser, and R. W. Ogden. A new constitutive framework for arterial wall mechanics and a comparative study of material models. J. Elast. 61:1–48, 2000.
Holzapfel, G. A., T. C. Gasser, and M. Stadler. A structural model for the viscoelastic behavior of arterial walls: continuum formulation and finite element analysis. Eur. J. Mech. A Solid. 21:441–463, 2002.
Holzapfel, G. A., T. C. Gasser, and R. W. Ogden. Comparison of a multi-layer structural model for arterial walls with a fung-type model, and issues of material stability. J. Biomech. Eng. Trans. ASME 126:264–275, 2004.
Holzapfel, G., M. Stadler, and T. C. Gasser. Changes in the mechanical environment of stenotic arteries during interaction with stents: computational assessment of parametric stent designs. J Biomech. Eng. Trans. ASME 127:166–180, 2005.
Holzapfel, G. A., G. Sommer, C. T. Gasser, and P. Regitnig. Determination of layer-specific mechanical properties of human coronary arteries with nonatherosclerotic intimal thickening and related constitutive modeling. Am. J. Physiol. Heart Circ. Physiol. 289:H2048–H2058, 2005.
Hua, J., and W. R. Mower. Simple geometric characteristics fail to reliably predict abdominal aortic aneurysm wall stresses. J. Vasc. Surg. 34:308–315, 2001.
Lederle, F. A., S. E. Wilson, G. R. Johnson, D. B. Reinke, F. N. Littooy, C. W. Acher, D. J. Ballard, L. M. Messina, I. L. Gordon, E. P. Chute, W. C. Krupski, D. Bandyk, and A. D. M. Vet. Immediate repair compared with surveillance of small abdominal aortic aneurysms. N. Eng. J. Med. 346:1437–1444, 2002.
Limet, R., N. Sakalihassan, and A. Albert. Determination of the expansion rate and incidence of rupture of abdominal aortic aneurysms. J. Vasc. Surg. 14:540–548, 1991.
Mooney, M. A theory of large elastic deformation. J. Appl. Phys. 11:582–592, 1940.
Moore, Jr., J. E., S. E. Maier, D. N. Ku, and P. Boesiger. Hemodynamics in the abdominal aorta: a comparison of in vitro and in vivo measurements. J. Appl. Physiol. 76:1520–1527, 1994.
Olufsen, M. S., C. S. Peskin, W. Y. Kim, E. M. Pedersen, A. Nadim, and J. Larsen. Numerical simulation and experimental validation of blood flow in arteries with structured-tree outflow conditions. Ann. Biomed. Eng. 28:1281–1299, 2000.
Papaharilaou, Y., J. A. Ekaterinaris, E. Manousaki, and A. N. Katsamouris. A decoupled fluid structure approach for estimating wall stress in abdominal aortic aneurysms. J. Biomech. 40:367–377, 2007.
Paszkowiak, J. J., and A. Dardik. Arterial wall shear stress: observations from the bench to the bedside. Vasc. Endovascular Surg. 37:47–57, 2003.
Powell, J. T., A. R. Brady, L. C. Brown, J. F. Forbes, F. G. R. Fowkes, R. M. Greenhalgh, C. V. Ruckley, S. G. Thompson, and U. S. A. T. Participants. Mortality results for randomised controlled trial of early elective surgery or ultrasonographic surveillance for small abdominal aortic aneurysms. Lancet 352:1649–1655, 1998.
Raghavan, M. L., and D. A. Vorp. Toward a biomechanical tool to evaluate rupture potential of abdominal aortic aneurysm: identification of a finite strain constitutive model and evaluation of its applicability. J. Biomech. 33:475–482, 2000.
Raghavan, M. L., M. W. Webster, and D. A. Vorp. Ex vivo biomechanical behavior of abdominal aortic aneurysm: assessment using a new mathematical model. Ann. Biomed. Eng. 24:573–582, 1996.
Raghavan, M. L., J. Kratzberg, E. M. C. de Tolosa, M. M. Hanaoka, P. Walker, and E. S. da Silva. Regional distribution of wall thickness and failure properties of human abdominal aortic aneurysm. J. Biomech. 39:3010–3016, 2006.
Reneman, R. S., T. Arts, and A. P. Hoeks. Wall shear stress—an important determinant of endothelial cell function and structure—in the arterial system in vivo. Discrepancies with theory. J. Vasc. Res. 43:251–269, 2006.
Ricotta, J. J., J. Pagan, M. Xenos, Y. Alemu, S. Einav, and D. Bluestein. Cardiovascular disease management: the need for better diagnostics. Med. Biol. Eng. Comput. 46:1059–1068, 2008.
Rissland, P., Y. Alemu, S. Einav, J. Ricotta, and D. Bluestein. Abdominal aortic aneurysm risk of rupture—patient specific FSI simulations using anisotropic model. J. Biomech. Eng. 13:031001–031010, 2009.
Rivlin, R. S. Large elastic deformations of isotropic materials. 1. Fundamental concepts. Philos. Trans. R. Soc. Lond. A Math. Phys. Sci. 240:459–508, 1948.
Roach, M. R., and A. C. Burton. The reason for the shape of the distensibility curves of arteries. Can. J. Biochem. Physiol. 35:681–690, 1957.
Rodriguez, J. F., C. Ruiz, M. Doblare, and G. A. Holzapfel. Mechanical stresses in abdominal aortic aneurysms: influence of diameter, asymmetry, and material anisotropy. J. Biomech. Eng. 130:021023, 2008.
Scotti, C., A. Shkolnik, S. Muluk, and E. Finol. Fluid-structure interaction in abdominal aortic aneurysms: effects of asymmetry and wall thickness. Biomed. Eng. Online 4:64, 2005.
Sussman, T., and K. J. Bathe. A finite-element formulation for nonlinear incompressible elastic and inelastic analysis. Comput. Struct. 26:357–409, 1987.
Swillens, A., L. Lanoye, J. De Backer, N. Stergiopulos, P. R. Verdonck, F. Vermassen, and P. Segers. Effect of an abdominal aortic aneurysm on wave reflection in the aorta. IEEE Trans. Biomed. Eng. 55:1602–1611, 2008.
Thubrikar, M. Vascular Mechanics and Pathology. New York: Springer, 436 pp., 2007.
Truijers, M., J. A. Pol, L. J. Schultzekool, S. M. van Sterkenburg, M. F. Fillinger, and J. D. Blankensteijn. Wall stress analysis in small asymptomatic, symptomatic and ruptured abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. 33:401–407, 2007.
Valencia, A., H. Morales, R. Rivera, E. Bravo, and M. Galvez. Blood flow dynamics in patient-specific cerebral aneurysm models: the relationship between wall shear stress and aneurysm area index. Med. Eng. Phys. 30:329–340, 2008.
Vande Geest, J. P., M. S. Sacks, and D. A. Vorp. Age dependency of the biaxial biomechanical behavior of human abdominal aorta. J. Biomech. Eng. 126:815–822, 2004.
Vande Geest, J. P., D. H. Wang, S. R. Wisniewski, M. S. Makaroun, and D. A. Vorp. Towards a noninvasive method for determination of patient-specific wall strength distribution in abdominal aortic aneurysms. Ann. Biomed. Eng. 34:1098–1106, 2006.
Vande Geest, J. P., M. S. Sacks, and D. A. Vorp. The effects of aneurysm on the biaxial mechanical behavior of human abdominal aorta. J. Biomech. 39:1324–1334, 2006.
Vande Geest, J. P., D. E. Schmidt, M. S. Sacks, and D. A. Vorp. The effects of anisotropy on the stress analyses of patient-specific abdominal aortic aneurysms. Ann. Biomed. Eng. 36:921–932, 2008.
Vardulaki, K. A., N. M. Walker, N. E. Day, S. W. Duffy, H. A. Ashton, and R. A. Scott. Quantifying the risks of hypertension, age, sex and smoking in patients with abdominal aortic aneurysm. Br. J. Surg. 87:195–200, 2000.
Vengrenyuk, Y., L. Cardoso, and S. Weinbaum. Micro-CT based analysis of a new paradigm for vulnerable plaque rupture: cellular microcalcifications in fibrous caps. Mol. Cell. Biomech. 5:37–47, 2008.
Venkatasubramaniam, A. K., M. J. Fagan, T. Mehta, K. J. Mylankal, B. Ray, G. Kuhan, I. C. Chetter, and P. T. McCollum. A comparative study of aortic wall stress using finite element analysis for ruptured and non-ruptured abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. 28:168–176, 2004.
Vito, R. P., and J. Hickey. The mechanical properties of soft tissues-II: the elastic response of arterial segments. J. Biomech. 13:951–957, 1980.
Vorp, D. A., M. L. Raghavan, S. C. Muluk, M. S. Makaroun, D. L. Steed, R. Shapiro, and M. W. Webster. Wall strength and stiffness of aneurysmal and nonaneurysmal abdominal aorta. Ann. N. Y. Acad. Sci. 800:274–276, 1996.
Wang, D. H., M. Makaroun, M. W. Webster, and D. A. Vorp. Mechanical properties and microstructure of intraluminal thrombus from abdominal aortic aneurysm. J. Biomech. Eng. 123:536–539, 2001.
Wilmink, A. B., C. S. Hubbard, N. E. Day, and C. R. Quick. The incidence of small abdominal aortic aneurysms and the change in normal infrarenal aortic diameter: implications for screening. Eur. J. Vasc. Endovasc. Surg. 21:165–170, 2001.
Wolters, B. J., M. C. Rutten, G. W. Schurink, U. Kose, J. de Hart, and F. N. van de Vosse. A patient-specific computational model of fluid-structure interaction in abdominal aortic aneurysms. Med. Eng. Phys. 27:871–883, 2005.
Yang, C., R. G. Bach, J. Zheng, I. E. Naqa, P. K. Woodard, Z. Teng, K. Billiar, and D. Tang. In vivo IVUS-based 3-D fluid-structure interaction models with cyclic bending and anisotropic vessel properties for human atherosclerotic coronary plaque mechanical analysis. IEEE Trans. Biomed. Eng. 56:2420–2428, 2009.
Zankl, A. R., H. Schumacher, U. Krumsdorf, H. A. Katus, L. Jahn, and C. P. Tiefenbacher. Pathology, natural history and treatment of abdominal aortic aneurysms. Clin. Res. Cardiol. 96:140–151, 2007.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Peter E. McHugh oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Xenos, M., Rambhia, S.H., Alemu, Y. et al. Patient-Based Abdominal Aortic Aneurysm Rupture Risk Prediction with Fluid Structure Interaction Modeling. Ann Biomed Eng 38, 3323–3337 (2010). https://doi.org/10.1007/s10439-010-0094-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-010-0094-3