Abstract
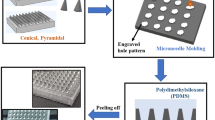
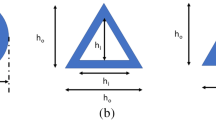
Hollow microneedles (MN) have been used for the past 20 years and exhibit controlled painless delivery of a wide range of drugs into the skin. Design and simulation analysis is needed before moving forward with microneedle production for successful penetration. In this study, we introduce a novel hollow microneedle array with integrated reservoir and cylindrical center lumen for transdermal drug delivery (TDD) applications. A conical and pyramidal geometry structure of hollow microneedle array was designed, with varying needle height (400–1000 μm), base diameter (150–300 μm), and tip diameter (10–30 μm). These microneedles are positioned in a 4 × 4 matrix array of 16 microneedles with center-to-center spacing of 650 µm. The numerical analysis is carried out with polyvinyl alcohol (PVA) as structure material using the COMSOL Multiphysics tool. A 4 × 4 conical and pyramidal hollow microneedle array is subjected to a stress study under axial and bending loading conditions. The effect of variation in the geometry parameter of the microneedle in relation to mechanical strength was investigated. The microfluidic analysis was performed with water and glucose as fluids with different viscosities. It has been found that the pyramidal microneedle array has a flow rate of 230 µL/min, whereas a sharper conical tip has stronger mechanical stability with less bending and has a higher flow rate of 320 µL/min.












Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Aggarwal P, Johnston CR (2004) Geometrical effects in mechanical characterizing of microneedle for biomedical applications. Sens Actuators B Chem 102(2):226–234. https://doi.org/10.1016/j.snb.2004.04.024
Ahmed Saeed AL-Japairai K et al (2020) Current trends in polymer microneedle for transdermal drug delivery. Int J Pharma 587:119673. https://doi.org/10.1016/j.ijpharm.2020.119673
Aldawood FK, Andar A, Desai S (2021) A comprehensive review of microneedles: types, materials, processes, characterizations and applications. Polymers 13(16):1–34. https://doi.org/10.3390/polym13162815
Amin F, Ahmed S (2013) Design, modeling and simulation of MEMS-based silicon microneedles. J Phys Conf Ser. https://doi.org/10.1088/1742-6596/439/1/012049
Aswani Kumar G et al (2021) Effect of insertion force for successful penetration of a conical shaped microneedle into the skin. In: Lecture notes in mechanical engineering. Springer, Singapore. https://doi.org/10.1007/978-981-15-4488-0_51
Bal SM et al (2010) Influence of microneedle shape on the transport of a fluorescent dye into human skin in vivo. J Control Release 147(2):218–224. https://doi.org/10.1016/j.jconrel.2010.07.104
Banga AK, Bose S, Ghosh TK (1999) Iontophoresis and electroporation: comparisons and contrasts. Int J Pharm 179(1):1–19. https://doi.org/10.1016/S0378-5173(98)00360-3
Bodhale DW, Nisar A, Afzulpurkar N (2010) Structural and microfluidic analysis of hollow side-open polymeric microneedles for transdermal drug delivery applications. Microfluid Nanofluid 8(3):373–392. https://doi.org/10.1007/s10404-009-0467-9
Bouwstra JA, Ponec M (2006) The skin barrier in healthy and diseased state. Biochim Biophys Acta Biomembr 1758(12):2080–2095. https://doi.org/10.1016/j.bbamem.2006.06.021
Byl NN (1995) The use of ultrasound as an enhancer for transcutaneous drug delivery: phonophoresis. Phys Ther 75(6):539–553. https://doi.org/10.1093/ptj/75.6.539
Cordeiro AS et al (2020) Two-photon polymerisation 3D printing of microneedle array templates with versatile designs: application in the development of polymeric drug delivery systems. Pharm Res. https://doi.org/10.1007/s11095-020-02887-9
Garcia J, Rios I, Fonthal F (2017) Design and analysis of microneedles structures for transdermal drug delivery. In: Proceedings of the 2016 IEEE ANDESCON, ANDESCON 2016. https://doi.org/10.1109/ANDESCON.2016.7836202
Gera AK, Burra RK (2022) The rise of polymeric microneedles: recent developments, advances, challenges, and applications with regard to transdermal drug delivery. J Funct Biomater 13(2):81. https://doi.org/10.3390/jfb13020081
Gill HS et al (2008) Effect of microneedle design on pain in human subjects. Clin J Pain 24(7):585
Gittard SD et al (2013) The effects of geometry on skin penetration and failure of polymer microneedles. J Adhes Sci Technol 27(3):227–243. https://doi.org/10.1080/01694243.2012.705101
Griss P, Stemme G (2002) Novel, side opened out-of-plane microneedles for microfluidic transdermal interfacing. In: Proceedings of the IEEE micro electro mechanical systems (MEMS), (February), pp 467–470. https://doi.org/10.1109/memsys.2002.984303
Henry S et al (1998a) Microfabricated microneedles: a novel approach to transdermal drug delivery. J Pharm Sci 87(8):922–925. https://doi.org/10.1021/js980042+
Henry S et al (1998b) Micromachined needles for the transdermal delivery of drugs. In: Proceedings of the IEEE micro electro mechanical systems (MEMS), pp 494–498. https://doi.org/10.1109/memsys.1998.659807
Hu Z et al (2020) Solid and hollow metallic glass microneedles for transdermal drug-delivery. Appl Phys Lett 116(20):203703. https://doi.org/10.1063/5.0008983
Ita K (2015) Transdermal delivery of drugs with microneedles—potential and challenges. Pharmaceutics 7(3):90–105. https://doi.org/10.3390/pharmaceutics7030090
Jain N, Singh VK, Chauhan S (2017) A review on mechanical and water absorption properties of polyvinyl alcohol based composites/films. J Mech Behav Mater 26(5–6):213–222. https://doi.org/10.1515/jmbm-2017-0027
Jung JH, Jin SG (2021) Microneedle for transdermal drug delivery: current trends and fabrication. J Pharm Investig 51:503–517. https://doi.org/10.1007/s40005-021-00512-4
Khumpuang S, Maeda R, Sugiyama S (2003) Design and fabrication of a coupled microneedle array and insertion guide array for safe penetration through skin. In: MHS 2003—proceedings of 2003 international symposium on micromechatronics and human science, vol 1, pp 233–237. https://doi.org/10.1109/MHS.2003.1249938
Kochhar JS et al (2013) Effect of microneedle geometry and supporting substrate on microneedle array penetration into skin. J Pharm Sci 102(11):4100–4108. https://doi.org/10.1002/jps.23724
Li Y et al (2019) Fabrication of sharp silicon hollow microneedles by deep-reactive ion etching towards minimally invasive diagnostics. Microsyst Nanoeng. https://doi.org/10.1038/s41378-019-0077-y
Loizidou EZ et al (2016) ‘Evaluation of geometrical effects of microneedles on skin penetration by CT scan and finite element analysis. Eur J Pharm Biopharm 107:1–6. https://doi.org/10.1016/j.ejpb.2016.06.023
Mcallister DV, Allen MG, Prausnitz RM (2000) M icrofabricated M icroneedles for G ene. Molecules 2:289–313
Mccrudden MTC et al (2013) Microneedles for intradermal and transdermal delivery. Eur J Pharm Sci 50(5):623–637. https://doi.org/10.1016/j.ejps.2013.05.005.Microneedles
Mishra R, Maiti TK, Bhattacharyya TK (2018) Design and scalable fabrication of hollow SU-8 microneedles for transdermal drug delivery. IEEE Sens J IEEE 18(14):5635–5644. https://doi.org/10.1109/JSEN.2018.2840335
Ogiso T, Iwaki M, Paku T (1995) Effect of various enhancers on transdermal penetration of indomethacin and urea, and relationship between penetration parameters and enhancement factors. J Pharm Sci 84(4):482–488. https://doi.org/10.1002/jps.2600840418
Oh NG, Hwang SY, Na YH (2022) Fabrication of a PVA-based hydrogel microneedle patch. ACS Omega 7(29):25179–25185. https://doi.org/10.1021/acsomega.2c01993
Pradeep Narayanan S, Raghavan S (2017) Solid silicon microneedles for drug delivery applications. Int J Adv Manuf Technol 93(1–4):407–422. https://doi.org/10.1007/s00170-016-9698-6
Prausnitz MR (2004) Microneedles for transdermal drug delivery. Adv Drug Deliv Rev 56(5):581–587. https://doi.org/10.1016/j.addr.2003.10.023
Ranamukhaarachchi SA et al (2016) Integrated hollow microneedle-optofluidic biosensor for therapeutic drug monitoring in sub-nanoliter volumes. Sci Rep 6(July):1–10. https://doi.org/10.1038/srep29075
Sawon MA, Samad MF (2021) Design and optimization of a microneedle with skin insertion analysis for transdermal drug delivery applications. J Drug Deliv Sci Technol. https://doi.org/10.1016/j.jddst.2021.102477
Schuetz YB et al (2005) Emerging strategies for the transdermal delivery of peptide and protein drugs. Expert Opin Drug Deliv 2(3):533–548. https://doi.org/10.1517/17425247.2.3.533
Sharma D (2018) Microneedles: an approach in transdermal drug delivery: a review. Pharmatutor 6(1):07. https://doi.org/10.29161/pt.v6.i1.2018.7
Shibata T et al (2007) Fabrication and mechanical characterization of microneedle array for cell surgery. In: Transducers and EUROSENSORS ’07—4th international conference on solid-state sensors, actuators and microsystems, vol 3(September), pp 719–722. https://doi.org/10.1109/SENSOR.2007.4300231
Shu W et al (2021) Insights into the mechanics of solid conical microneedle array insertion into skin using the finite element method. Acta Biomater 135:403–413. https://doi.org/10.1016/j.actbio.2021.08.045
Tariq N et al (2022) Numerical simulation, analysis, and fabrication of MEMS-based solid Ag and Cu microneedles for biomedical applications. Math Problems Eng 2022:1–19. https://doi.org/10.1155/2022/1633183
Tayyaba S, Ashraf MW, Afzulpurkar N (2013) Design, simulation, and fabrication of microneedles and a blood filter for use in a hemofiltration system. IEEE Trans Autom Sci Eng 10(2):252–266. https://doi.org/10.1109/TASE.2012.2230166
Vinayakumar KB et al (2016) Out-of-plane cup shaped stainless steel microneedle array for drug delivery. In: 2016 IEEE 11th annual international conference on nano/micro engineered and molecular systems, NEMS 2016, pp 172–175. https://doi.org/10.1109/NEMS.2016.7758225
Waghule T et al (2019) Microneedles: a smart approach and increasing potential for transdermal drug delivery system. Biomed Pharmacother 109:1249–1258. https://doi.org/10.1016/j.biopha.2018.10.078
Wang PC et al (2009) Hollow polymer microneedle array fabricated by photolithography process combined with micromolding technique. In: Proceedings of the 31st annual international conference of the IEEE engineering in medicine and biology society: engineering the future of biomedicine, EMBC 2009, pp 7026–7029. https://doi.org/10.1109/IEMBS.2009.5333317
Yan Q et al (2022) The finite element analysis research on microneedle design strategy and transdermal drug delivery system. Pharmaceutics 14(8):1625. https://doi.org/10.3390/pharmaceutics14081625
Acknowledgements
This work was supported in part by the Department of Science and Technology under Grant TDP/BDTD/23/2019. The authors would like to thank Indian Science Technology and Engineering facilities Map (I-STEM), a program supported by Office of the Principal Scientific Adviser to the Govt. of India, for enabling access to the COMSOL Multiphysics 5.6 software suite used to carry out this work.
Author information
Authors and Affiliations
Contributions
AG, BSS and SR contributed to the study concept and design. Simulation, analysis and fabrication process flow work done by AG. The result validation performed by BSS and SR. AG, BSS and SR participated in the writing and revision of the manuscript equally.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anbazhagan, G., Suseela, S.B. & Sankararajan, R. Effect of hollow microneedle geometry structure on mechanical stability and microfluidic flow for transdermal drug delivery applications. Microfluid Nanofluid 27, 25 (2023). https://doi.org/10.1007/s10404-023-02636-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-023-02636-5