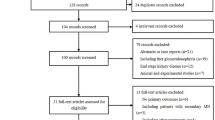
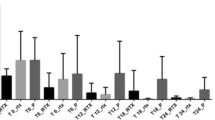
Abstract
Rituximab (RTX) has been the first option in idiopathic membranous nephropathy (IMN). However, the clinical effect was not very satisfactory. This study aimed to explore the clinical efficacy and safety of the combination of RTX and glucocorticoids (GC) in anti-phospholipase A2 receptor (anti-PLA2R) antibody positive IMN. Sixty-six patients were randomly divided into RTX/GC group (RTX infusion plus short-term oral GC) and RTX group (RTX infusion alone) in this prospective cohort study. Complete remission (CR) and partial remission (PR) were the primary outcomes. Adverse events were the secondary outcomes. The laboratory index including serum albumin, 24 h urinary protein, serum creatinine, estimated glomerular filtration rate, and anti-PLA2R antibody titer were also monitored. All patients were followed for at least 12 months. During the 12-month follow-up, the composite remission rates in RTX/GC and RTX groups were 74.3% and 67.7%, and the CR rates were 34.3% and 19.4%, respectively. The median time of remission in RTX/GC group was shorter than the RTX group (P < 0.001). Compared with RTX monotherapy, the combination of RTX and GC significantly decreased the anti-PLA2R antibody titer (P = 0.028). No significant difference was observed in the incidence of adverse events. The results of the Kaplan–Meier survival analysis indicated that the cumulative CR rate and cumulative composite remission rate in RTX/GC group were all better than the RTX group (P = 0.043, P = 0.040, respectively). The combination of RTX and GC was better than RTX monotherapy without increasing the adverse events in the treatment of IMN.



Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this article could be acquired from the authors.
References
Ronco P, Beck L, Debiec H, Fervenza FC, Hou FF, Jha V, Sethi S, Tong A, Vivarelli M, Wetzels J. Membranous nephropathy. Nat Rev Dis Primers. 2021. https://doi.org/10.1038/s41572-021-00303-z.
Rovin BH, Adler SG, Barratt J, Bridoux F, Burdge KA, Chan TM, Cook HT, Fervenza FC, Gibson KL, Glassock RJ, et al. Executive summary of the kdigo 2021 guideline for the management of glomerular diseases. Kidney Int. 2021. https://doi.org/10.1016/j.kint.2021.05.015.
Rojas-Rivera JE, Ortiz A, Fervenza FC. Novel treatments paradigms: membranous nephropathy. Kidney Int Rep. 2023. https://doi.org/10.1016/j.ekir.2022.12.011.
von Groote TC, Williams G, Au EH, Chen Y, Mathew AT, Hodson EM, Tunnicliffe DJ. Immunosuppressive treatment for primary membranous nephropathy in adults with nephrotic syndrome. Cochrane Database Syst Rev. 2021. https://doi.org/10.1002/14651858.CD004293.pub4.
Fervenza FC, Appel GB, Barbour SJ, Rovin BH, Lafayette RA, Aslam N, Jefferson JA, Gipson PE, Rizk DV, Sedor JR, et al. Rituximab or cyclosporine in the treatment of membranous nephropathy. N Engl J Med. 2019. https://doi.org/10.1056/NEJMoa1814427.
Scolari F, Delbarba E, Santoro D, Gesualdo L, Pani A, Dallera N, Mani LY, Santostefano M, Feriozzi S, Quaglia M, et al. Rituximab or cyclophosphamide in the treatment of membranous nephropathy: the ri-cyclo randomized trial. J Am Soc Nephrol. 2021. https://doi.org/10.1681/asn.2020071091.
Hofstra JM, Fervenza FC, Wetzels JF. Treatment of idiopathic membranous nephropathy. Nat Rev Nephrol. 2013. https://doi.org/10.1038/nrneph.2013.125.
Rice JB, White AG, Scarpati LM, Wan G, Nelson WW. Long-term systemic corticosteroid exposure: a systematic literature review. Clin Ther. 2017. https://doi.org/10.1016/j.clinthera.2017.09.011.
Ma Q, Li X, Xu G. New-onset and relapsed membranous nephropathy post sars-cov-2 and covid-19 vaccination. Viruses. 2022. https://doi.org/10.3390/v14102143.
Fernández-Juárez G, Rojas-Rivera J, Logt AV, Justino J, Sevillano A, Caravaca-Fontán F, Ávila A, Rabasco C, Cabello V, Varela A, et al. The starmen trial indicates that alternating treatment with corticosteroids and cyclophosphamide is superior to sequential treatment with tacrolimus and rituximab in primary membranous nephropathy. Kidney Int. 2021;99:986–98. https://doi.org/10.1016/j.kint.2020.10.014.
Ruggenenti P, Fervenza FC, Remuzzi G. Treatment of membranous nephropathy: time for a paradigm shift. Nat Rev Nephrol. 2017;13:563–79. https://doi.org/10.1038/nrneph.2017.92.
Gorriz JL, Martinez-Castelao A. Proteinuria: detection and role in native renal disease progression. Transplant Rev (Orlando). 2012. https://doi.org/10.1016/j.trre.2011.10.002.
Brantsma AH, Bakker SJ, de Zeeuw D, de Jong PE, Gansevoort RT. Extended prognostic value of urinary albumin excretion for cardiovascular events. J Am Soc Nephrol. 2008. https://doi.org/10.1681/asn.2007101065.
Ruggenenti P, Perna A, Mosconi L, Pisoni R, Remuzzi G. Urinary protein excretion rate is the best independent predictor of esrf in non-diabetic proteinuric chronic nephropathies. Gruppo italiano di studi epidemiologici in nefrologia (gisen). Kidney Int. 1998. https://doi.org/10.1046/j.1523-1755.1998.00874.x.
van de Logt AE, Fresquet M, Wetzels JF, Brenchley P. The anti-pla2r antibody in membranous nephropathy: What we know and what remains a decade after its discovery. Kidney Int. 2019. https://doi.org/10.1016/j.kint.2019.07.014.
Rougé L, Chiang N, Steffek M, Kugel C, Croll TI, Tam C, Estevez A, Arthur CP, Koth CM, Ciferri C, et al. Structure of cd20 in complex with the therapeutic monoclonal antibody rituximab. Science. 2020. https://doi.org/10.1126/science.aaz9356.
Mishima Y, Terui Y, Takeuchi K, Matsumoto-Mishima Y, Matsusaka S, Utsubo-Kuniyoshi R, Hatake K. The identification of irreversible rituximab-resistant lymphoma caused by cd20 gene mutations. Blood Cancer J. 2011. https://doi.org/10.1038/bcj.2011.11.
Parrillo JE, Fauci AS. Mechanisms of glucocorticoid action on immune processes. Annu Rev Pharmacol Toxicol. 1979. https://doi.org/10.1146/annurev.pa.19.040179.001143.
Flammer JR, Rogatsky I. Minireview: glucocorticoids in autoimmunity: Unexpected targets and mechanisms. Mol Endocrinol. 2011. https://doi.org/10.1210/me.2011-0068.
Zhao Q, Dai H, Liu X, Jiang H, Liu W, Feng Z, Zhang N, Gao Y, Dong Z, Zhou X, et al. Helper t cells in idiopathic membranous nephropathy. Front Immunol. 2021. https://doi.org/10.3389/fimmu.2021.665629.
Funding
This work was supported by the Key Clinical Research Project of the Second Affiliated Hospital of Nanchang University (No. 2022efyB01), the “Thousand Talents Plan” Project of Introducing and Training High‐level Talents of Innovation and Entrepreneurship in Jiangxi Province (No. JXSQ2023201030), and the Jiangxi Graduate Innovation Special Fund (No. YC2023-B090).
Author information
Authors and Affiliations
Contributions
QM conducted conception, design, data collection and manuscript writing. ML conducted data collection; GX was responsible for the idea, funds and paper revision. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, Q., Li, M. & Xu, G. Combination of rituximab and short-term glucocorticoids in the treatment of anti-phospholipase A2 receptor antibody positive idiopathic membranous nephropathy. Clin Exp Med 23, 5337–5343 (2023). https://doi.org/10.1007/s10238-023-01183-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-01183-1