Abstract
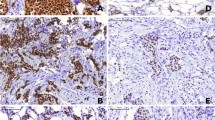
The luminal-A-like and luminal-B-like breast cancer groups have distinct biological features that lead to differences in the treatment response and clinical outcome. The aim of this study was to examine the value of the distribution pattern of ERα expression, ESR1 SNPs as well as ESR1 mRNA expression in predicting tamoxifen response and survival in patients with luminal-A-like and luminal-B-like breast cancer. A total of 135 patients with both subtypes were stratified into two groups depending on the tamoxifen response: tamoxifen-resistant patients (TR) and tamoxifen-sensitive patients (TS). ESR1 mRNA expression was measured by real-time quantitative reverse transcription-PCR. Three polymorphisms of ESR1 (rs2077647, rs2228480 and rs1801132) were genotyped using a TaqMan assay. The distribution pattern of ERα expression was analyzed immunohistochemically using the visual assessment of staining. The primary endpoint was progression-free survival (PFS). There was a significant decrease in ESR1 mRNA expression level in the TR group when compared to the TS group among patients with luminal-B-like subtype (P = 0.038). ESR1 2014AA mutant genotype of rs2228480 was more prevalent in the TR patients with luminal-B-like subtype than the TS group (P = 0.045). In the luminal-A-like group, tamoxifen-resistant tumors were more frequently heterogeneous for ERα expression than tamoxifen-sensitive tumors (P = 0.016). Multivariate analysis showed a strong association of lymph node status and the distribution pattern of ERα expression with tamoxifen responsiveness in this cohort of patients. In addition, a luminal-A-like patients with the heterogeneous ERα expression had a significantly shorter PFS time than those with the homogeneous ERα (P = 0.013). These results indicate that the heterogeneous expression of ERα is an accurate predictor of tamoxifen response and survival in luminal-A-like breast cancer patients. ESR1 rs2228480 may act as a marker with a high prognostic potential in luminal-B-like tumors.




Similar content being viewed by others
References
Semiglazov VF. Diverse biology of breast cancer: search for adequate treatment. Malig Tumours. 2016;3:5–10.
Murphy CG, Dickler MN. Endocrine resistance in hormone-responsive breast cancer: mechanisms and therapeutic strategies. Endocr Relat Cancer. 2016;23(8):R337–52.
Curigliano G, Burstein HJ, Winer EP, et al. De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann Oncol. 2017;28(8):1700–12.
Osborne CK, Schiff R. Mechanisms of endocrine resistance in breast cancer. Annu Rev Med. 2011;62:233–47.
Clarke R, Tyson JJ, Dixon JM. Endocrine resistance in breast cancer—an overview and update. Mol Cell Endocrinol. 2015;418(Pt 3):220–34.
de Castro Sant’ Anna C, Junior AGF, Soares P, et al. Molecular biology as a tool for the treatment of cancer. Clin Exp Med. 2018;18(4):457–64.
Pan Y, Liu G, Zhou F, Su B, Li Y. DNA methylation profiles in cancer diagnosis and therapeutics. Clin Exp Med. 2018;18(1):1–14.
Mueller C, Haymond A, Davis JB, Williams A, Espina V. Protein biomarkers for subtyping breast cancer and implications for future research. Expert Rev Proteom. 2018;15(2):131–52.
Brufsky AM, Dickler MN. Estrogen receptor positive breast cancer: exploiting signaling pathways implicated in endocrine resistance. Oncologist. 2018;23(5):528–39.
Mills JN, Rutkovsky AC, Giordano A. Mechanisms of resistance in estrogen receptor positive breast cancer: overcoming resistance to tamoxifen/aromatase inhibitors. Curr Opin Pharmacol. 2018;41:59–65.
Nardone A, De Angelis C, Trivedi MV, Osborne CK, Schiff R. The changing role of ER in endocrine resistance. Breast. 2015;24(Suppl 2):S60–6.
Badve S, Turbin D, Thorat MA, et al. FOXA1 expression in breast cancer—correlation with luminal subtype A and survival. Clin Cancer Res. 2007;13(15Pt 1):4415–21.
Babyshkina N, Vtorushin S, Zavyalova M, et al. The distribution pattern of ERα expression, ESR1 genetic variation and expression of growth factor receptors: association with breast cancer prognosis in Russian patients treated with adjuvant tamoxifen. Clin Exp Med. 2017;17(3):383–93.
Hammond ME, Hayes DF, Dowsett M, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch Pathol Lab Med. 2010;134(7):e48–72.
Lessey BA, Palomino WA, Apparao KB, Young SL, Lininger RA. Estrogen receptor-alpha (ER-alpha) and defects in uterine receptivity in women. Reprod Biol Endocrinol. 2006;4(Suppl 1):S9.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–8.
Dronova TA, Babyshkina NN, Zavyalova MV, et al. Relation of EGFR/PI3K/AKT signaling components with tamoxifen efficacy in patients with estrogen-dependent breast cancer. Adv Mol Oncol. 2018;5(3):40–50.
Kim C, Tang G, Pogue-Geile KL, et al. Estrogen receptor (ESR1) mRNA expression and benefit from tamoxifen in the treatment and prevention of estrogen receptor-positive breast cancer. J Clin Oncol. 2011;29(31):4160–7.
Bordeaux JM, Cheng H, Welsh AW, et al. Quantitative in situ measurement of estrogen receptor mRNA predicts response to tamoxifen. PLoS ONE. 2012;7(5):e36559.
Kim S, Lee J, Oh SJ, Nam SJ, Lee JE. Differential effect of EGFR inhibitors on tamoxifen-resistant breast cancer cells. Oncol Rep. 2015;34(3):1613–9.
Acknowledgements
The study was supported by the Grant of the Russian Foundation for Basic Research No. 17‑29‑06037. We acknowledge support of this work by the Tomsk State University Competitiveness Improvement Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
The study was approved by the Ethics Committee of the Tomsk Cancer Research Institute and has been performed in accordance with the Helsinki Declaration (1964, amended in 1975 and 1983).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Babyshkina, N., Vtorushin, S., Dronova, T. et al. Impact of estrogen receptor α on the tamoxifen response and prognosis in luminal-A-like and luminal-B-like breast cancer. Clin Exp Med 19, 547–556 (2019). https://doi.org/10.1007/s10238-019-00583-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-019-00583-6