Abstract
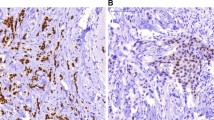
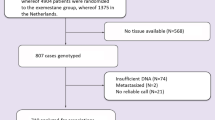
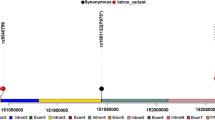
Identification of additional biomarkers associated with ER genomic and nongenomic pathways could be very useful to distinguish patients who will benefit from tamoxifen treatment. The aim of this study was to analyze the prognostic significance of the distribution pattern of ERα expression, ESR1 gene single-nucleotide polymorphisms and expression levels of growth factor receptors in Russian hormone receptor-positive breast cancer patients treated with adjuvant tamoxifen. Formalin-fixed paraffin-embedded tumor tissue samples from 97 patients were examined for the distribution pattern of ERα expression, as well as for EGFR and TGF-βR1 expression by immunohistochemistry. Genotypes for ESR1 +30T>C (rs2077647) and ESR1 2014G>A (rs2228480) were analyzed using a TaqMan assay. Progression-free survival (PFS) was used as an endpoint for the survival analyses. We found that patients with the heterogeneous distribution of ERα expression had poor prognosis on tamoxifen treatment (P = 0.021). We identified a high EGFR expression in patients who developed distant metastasis or recurrence during tamoxifen treatment (a tamoxifen-resistant group—TR) in contrast to the distant metastasis-free patients (a tamoxifen-sensitive group—TS) (80.0 vs. 41.9 %, respectively, P = 0.009). Carriers of the ESR12014A mutant allele were more prevalent among the TR patients compared to the TS patients (26.3 vs. 8.0 %, respectively, P = 0.009). EGFR expression and the distribution pattern of ERα expression were associated with the response to tamoxifen by both univariate and multivariate logistic regression analyses. The presence of these markers either alone or in combination was correlated with the worse PFS for all patients. Analysis of the distribution pattern of ERα expression and the EGFR status in tumor tissue may be valuable for patient selection for tamoxifen adjuvant therapy.




Similar content being viewed by others
References
Goldhirsch A, Winer EP, Coates AS, et al. Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer. Ann Oncol. 2013;24(9):2206–23.
Semiglazov VF. New approaches to the treatment of breast cancer. Vopr Onkol. 2013;59(3):288–91.
Osborne CK, Schiff R. Mechanisms of endocrine resistance in breast cancer. Annu Rev Med. 2011;62:233–47.
Semiglazova TY, Semiglazov VV, Filatova LV, et al. A new approach to the overcoming resistance to hormone therapy of breast cancer. Pharmateca. 2012;18:50–6.
Chang J, Fan W. Endocrine therapy resistance: current status, possible mechanisms and overcoming strategies. Anticancer Agents Med. Chem. 2013;13(3):464–75.
Jordan VC, O’Malley BW. Selective estrogen-receptor modulators and antihormonal resistance in breast cancer. J Clin Oncol. 2007;25:5815–24.
Gee JM, Robertson LF, Gutteridge E, et al. Epidermal growth factor receptor/HER2/insulin-like growth factor receptor signalling and oestrogen receptor activity in clinical breast cancer. Endocr Relat Cancer. 2005;12(Suppl 1):S99–111.
Dunning AM, Healey CS, Baynes C, et al. Association of ESR1 gene tagging SNPs with breast cancer risk. Hum Mol Genet. 2009;18:1131–9.
Ding SL, Yu JC, Chen ST, et al. Diverse associations between ESR1 polymorphism and breast cancer development and progression. Clin Cancer Res. 2010;16(13):3473–84.
Markiewicz A, Wełnicka-Jaśkiewicz M, Skokowski J, et al. Prognostic significance of ESR1 amplification and ESR1 PvuII, CYP2C19*2, UGT2B15*2 polymorphisms in breast cancer patients. PLoS ONE. 2013;8(8):e72219.
Lipphardt MF, Deryal M, Ong MF, Schmidt W, Mahlknecht U. ESR1 single nucleotide polymorphisms predict breast cancer susceptibility in the central European Caucasian population. Int J Clin Exp Med. 2013;6(4):282–8.
Sobti RC, Askari M, Nikbakht M, Singh N, Sharma SC, Abitew AM. Genetic variants of EGFR (142285G > A) and ESR1 (2014G > A) gene polymorphisms and risk of breast cancer. Mol Cell Biochem. 2012;369(1–2):217–25.
Xia P, Jin T, Geng T, et al. Polymorphisms in ESR1 and FLJ43663 are associated with breast cancer risk in the Han population. Tumour Biol. 2014;35(3):2187–90.
Madeira KP, Daltoé RD, Sirtoli GM, Carvalho AA, Rangel LB, Silva IV. Estrogen receptor alpha (ERS1) SNPs c454-397T > C (PvuII) and c454-351A > G (XbaI) are risk biomarkers for breast cancer development. Mol Biol Rep. 2014;41(8):5459–66.
Akisik E, Dalay N. Estrogen receptor codon 594 and HER1 codon 655 polymorphisms and breast cancer risk. Exp Mol Pathol. 2004;76(3):160–3.
Anghel A, Raica M, Narita D. Estrogen receptor alpha polymorphisms: correlation with clinicopathological parameters in breast cancer. Neoplasma. 2010;57(4):306–15.
Osborne CK. Steroid hormone receptors in breast cancer management. Breast Cancer Res Treat. 1998;51:227–38.
Higgins MJ, Stearns V. Understanding resistance to tamoxifen in hormone receptor-positive breast cancer. Clin Chem. 2009;55:1453e5.
Kucher AN, Ivanova OF, Puzyrev VP, Tsimbalyuk IV, Trotsenko BA. Genetic-demographic characteristics of a contemporary Siberian city Tomsk. Genetika. 1994;30(2):276–81.
Zavyalova MV, Denisov EV, Tashireva LA, et al. Phenotypic drift as a cause for intratumoral morphological heterogeneity of invasive ductal breast carcinoma not otherwise specified. Biores Open Access. 2013;2(2):148–54.
Hammond ME, Hayes DF, Dowsett M, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch Pathol Lab Med. 2010;134(7):e48–72.
Lessey BA, Palomino WA, Apparao KB, Young SL, Lininger RA. Estrogen receptor-alpha (ER-alpha) and defects in uterine receptivity in women. Reprod Biol Endocrinol. 2006;4(Suppl 1):S9.
Vtorushin SV, Perelmuter VM, Kritskaia NG, et al. (2007) Prediction of the breast cancer clinical course. Patent. 2007; 2300111 Russian Federation: MPK51 G1N33/74, G1N33/50.
Goldhirsch A, Ingle JN, Gelber RD, et al. Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2009. Ann Oncol. 2009;20:1319–29.
Vtorushin SV. Features of morphological structure of primary tumors in multicentric/multifocal growth of breast cancer. Sib Med J. 2011;26(1):92–7.
Vtorushin SV, Slonimskaya EM, Perelmuter VM, et al. Prediction of lymph node metastasis and disease outcome in breast cancer patients. Women Reprod Syst Tumors. 2007;1:43–5.
Shaikh T, Tam TY, Li T, Hayes SB, et al. Multifocal and multicentric breast cancer is associated with increased local recurrence regardless of surgery type. Breast J. 2015;21(2):121–6.
Weissenbacher TM, Zschage M, Janni W, et al. Multicentric and multifocal versus unifocal breast cancer: is the tumor-node-metastasis classification justified? Breast Cancer Res Treat. 2010;122(1):27–34.
Nicholson RI, McClelland RA, Gee JM, et al. Epidermal growth factor receptor expression in breast cancer: association with response to endocrine therapy. Breast Cancer Res Treat. 1994;29:117–25.
Xuan QJ, Wang JX, Nanding A, et al. Tumor associated macrophages are correlated with tamoxifen resistance in the postmenopausal breast cancer patients. Pathol Oncol Res. 2014;20(3):619–24.
Arpino G, Weiss H, Lee AV, et al. Estrogen receptor-positive, progesterone receptor-negative breast cancer: association with growth factor receptor expression and tamoxifen resistance. J Natl Cancer Inst. 2005;97(17):1254–61.
Giltnane JM, Rydén L, Cregger M, Bendahl PO, Jirström K, Rimm DL. Quantitative measurement of epidermal growth factor receptor is a negative predictive factor for tamoxifen response in hormone receptor positive premenopausal breast cancer. J Clin Oncol. 2007;25(21):3007–14.
Witton CJ, Reeves JR, Going JJ, Cooke TG, Bartlett JM. Expression of the HER1-4 family of receptor tyrosine kinases in breast cancer. J Pathol. 2003;200(3):290–7.
Dihge L, Bendahl PO, Grabau D, et al. Epidermal growth factor receptor (EGFR) and the estrogen receptor modulator amplified in breast cancer (AIB1) for predicting clinical outcome after adjuvant tamoxifen in breast cancer. Breast Cancer Res Treat. 2008;109(2):255–62.
Dowsett M, Houghton J, Iden C, et al. Benefit from adjuvant tamoxifen therapy in primary breast cancer patients according oestrogen receptor, progesterone receptor, EGF receptor and HER2 status. Ann Oncol. 2006;17(5):818–26.
Kallel I, Rebai M, Khabir A, Farid NR, Rebaï A. Genetic polymorphisms in the EGFR (R521 K) and estrogen receptor (T594T) genes, EGFR and ErbB-2 protein expression, and breast cancer risk in Tunisia. J Biomed Biotechnol. 2009;2009:753683.
Hsiao WC, Young KC, Lin SL, Lin PW. Estrogen receptor-alpha polymorphism in a Taiwanese clinical breast cancer population: a case-control study. Breast Cancer Res. 2004;6(3):R180–6.
Yang J, Singleton DW, Shaughnessy EA, Khan SA. The F-domain of estrogen receptor-alpha inhibits ligand induced receptor dimerization. Mol Cell Endocrinol. 2008;295(1–2):94–100.
Acknowledgments
The study was supported by the Russian Scientific Foundation, Grant #14-15-00350 (tumor sample collection, clinical data for patients within follow-up period). We acknowledge support of this work by the Tomsk State University Competitiveness Improvement Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical statement
The study was approved by the Ethics Committee of the Tomsk Cancer Research Institute and has been performed in accordance with the Helsinki Declaration (1964, amended in 1975 and 1983).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Babyshkina, N., Vtorushin, S., Zavyalova, M. et al. The distribution pattern of ERα expression, ESR1 genetic variation and expression of growth factor receptors: association with breast cancer prognosis in Russian patients treated with adjuvant tamoxifen. Clin Exp Med 17, 383–393 (2017). https://doi.org/10.1007/s10238-016-0428-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-016-0428-z