Abstract
Background
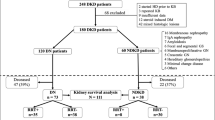
Studies on kidney function and histological findings in diabetic nephropathy (DN) with low urinary protein (UP) are few. We examined the differential impact of histological changes on kidney outcomes between non-proteinuric and proteinuric DN.
Methods
Patients diagnosed with DN by renal biopsy during 1981–2014 were divided into non-proteinuric (UP ≤ 0.5 g/day) and proteinuric (UP > 0.5 g/day) DN. The Cox proportional hazard model was used to examine the association of glomerular lesions (GLs) and interstitial fibrosis and tubular atrophy (IFTA) with end-stage kidney disease (ESKD) development after adjusting for relevant confounders.
Results
The non-proteinuric and proteinuric DN groups included 197 and 199 patients, respectively. During the 10.7-year median follow-up period, 16 and 83 patients developed ESKD in the non-proteinuric and proteinuric DN groups, respectively. In the multivariable Cox hazard model, hazard ratios (HRs) [95% confidence intervals (CIs)] of GL and IFTA for ESKD in proteinuric DN were 2.94 [1.67–5.36] and 3.82 [2.06–7.53], respectively. Meanwhile, HRs [95% CIs] of GL and IFTA in non-proteinuric DN were < 0.01 [0–2.48] and 4.98 [1.33–18.0], respectively. IFTA was consistently associated with higher incidences of ESKD regardless of proteinuria levels (P for interaction = 0.49). The prognostic impact of GLs on ESKD was significantly decreased as proteinuria levels decreased (P for interaction < 0.01).
Conclusions
IFTA is consistently a useful predictor of kidney prognosis in both non-proteinuric and proteinuric DN, while GLs are a significant predictor of kidney prognosis only in proteinuric DN.



Similar content being viewed by others
References
Afkarian M, Zelnick LR, Hall YN, Heagerty PJ, Tuttle K, Weiss NS, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988–2014. JAMA. 2016;316(6):602–10. https://doi.org/10.1001/jama.2016.10924.
Jiang G, Luk AOY, Tam CHT, Xie F, Carstensen B, Lau ESH, et al. Progression of diabetic kidney disease and trajectory of kidney function decline in Chinese patients with Type 2 diabetes. Kidney Int. 2019;95(1):178–87. https://doi.org/10.1016/j.kint.2018.08.026.
Yamanouchi M, Furuichi K, Hoshino J, Toyama T, Hara A, Shimizu M, et al. Nonproteinuric versus proteinuric phenotypes in diabetic kidney disease: a propensity score-matched analysis of a nationwide. Biopsy-Based Cohort Study Diabetes Care. 2019;42(5):891–902. https://doi.org/10.2337/dc18-1320.
Espinel E, Agraz I, Ibernon M, Ramos N, Fort J, Seron D. Renal biopsy in type 2 diabetic patients. J Clin Med. 2015;4(5):998–1009. https://doi.org/10.3390/jcm4050998.
Sharma SG, Bomback AS, Radhakrishnan J, Herlitz LC, Stokes MB, Markowitz GS, et al. The modern spectrum of renal biopsy findings in patients with diabetes. Clin J Am Soc Nephrol. 2013;8(10):1718–24. https://doi.org/10.2215/cjn.02510213.
Klessens CQ, Woutman TD, Veraar KA, Zandbergen M, Valk EJ, Rotmans JI, et al. An autopsy study suggests that diabetic nephropathy is underdiagnosed. Kidney Int. 2016;90(1):149–56. https://doi.org/10.1016/j.kint.2016.01.023.
Ekinci EI, Jerums G, Skene A, Crammer P, Power D, Cheong KY, et al. Renal structure in normoalbuminuric and albuminuric patients with type 2 diabetes and impaired renal function. Diabetes Care. 2013;36(11):3620–6. https://doi.org/10.2337/dc12-2572.
Yamanouchi M, Furuichi K, Hoshino J, Ubara Y, Wada T. Nonproteinuric diabetic kidney disease. Clin Exp Nephrol. 2020;24(7):573–81. https://doi.org/10.1007/s10157-020-01881-0.
Robles NR, Villa J, Gallego RH. Non-proteinuric diabetic nephropathy. J Clin Med. 2015;4(9):1761–73. https://doi.org/10.3390/jcm4091761.
An Y, Xu F, Le W, Ge Y, Zhou M, Chen H, et al. Renal histologic changes and the outcome in patients with diabetic nephropathy. Nephrol Dial Transplant. 2015;30(2):257–66. https://doi.org/10.1093/ndt/gfu250.
Shimizu M, Furuichi K, Toyama T, Kitajima S, Hara A, Kitagawa K, et al. Long-term outcomes of Japanese type 2 diabetic patients with biopsy-proven diabetic nephropathy. Diabetes Care. 2013;36(11):3655–62. https://doi.org/10.2337/dc13-0298.
Mise K, Hoshino J, Ubara Y, Sumida K, Hiramatsu R, Hasegawa E, et al. Renal prognosis a long time after renal biopsy on patients with diabetic nephropathy. Nephrol Dial Transplant. 2014;29(1):109–18. https://doi.org/10.1093/ndt/gft349.
Shimizu M, Furuichi K, Yokoyama H, Toyama T, Iwata Y, Sakai N, et al. Kidney lesions in diabetic patients with normoalbuminuric renal insufficiency. Clin Exp Nephrol. 2014;18(2):305–12. https://doi.org/10.1007/s10157-013-0870-0.
Gohda T, Murakoshi M, Koshida T, Ichikawa S, Li ZI, Adachi ERI, et al. Concept of diabetic kidney disease - paradigm shift from albuminuria-based to GFR-based kidney disease. Juntendo Medical Journal. 2019;65(6):510–6. https://doi.org/10.14789/jmj.2019.65.JMJ19-R16.
Morimoto K, Matsui M, Samejima K, Kanki T, Nishimoto M, Tanabe K, et al. Renal arteriolar hyalinosis, not intimal thickening in large arteries, is associated with cardiovascular events in people with biopsy-proven diabetic nephropathy. Diabet Med. 2020;37(12):2143–52. https://doi.org/10.1111/dme.14301.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53(6):982–92. https://doi.org/10.1053/j.ajkd.2008.12.034.
Geistanger A, Arends S, Berding C, Hoshino T, Jeppsson JO, Little R, et al. Statistical methods for monitoring the relationship between the IFCC reference measurement procedure for hemoglobin A1c and the designated comparison methods in the United States, Japan, and Sweden. Clin Chem. 2008;54(8):1379–85. https://doi.org/10.1373/clinchem.2008.103556.
Tervaert TW, Mooyaart AL, Amann K, Cohen AH, Cook HT, Drachenberg CB, et al. Pathologic classification of diabetic nephropathy. J Am Soc Nephrol. 2010;21(4):556–63. https://doi.org/10.1681/ASN.2010010010.
Furuichi K, Yuzawa Y, Shimizu M, Hara A, Toyama T, Kitamura H, et al. Nationwide multicentre kidney biopsy study of Japanese patients with type 2 diabetes. Nephrol Dial Transplant. 2018;33(1):138–48. https://doi.org/10.1093/ndt/gfw417.
Gilbert RE, Cooper ME. The tubulointerstitium in progressive diabetic kidney disease: more than an aftermath of glomerular injury? Kidney Int. 1999;56(5):1627–37. https://doi.org/10.1046/j.1523-1755.1999.00721.x.
Bonventre JV. Can we target tubular damage to prevent renal function decline in diabetes? Semin Nephrol. 2012;32(5):452–62. https://doi.org/10.1016/j.semnephrol.2012.07.008.
Okada T, Nagao T, Matsumoto H, Nagaoka Y, Wada T, Nakao T. Histological predictors for renal prognosis in diabetic nephropathy in diabetes mellitus type 2 patients with overt proteinuria. Nephrology (Carlton). 2012;17(1):68–75. https://doi.org/10.1111/j.1440-1797.2011.01525.x.
Eriguchi M, Lin M, Yamashita M, Zhao TV, Khan Z, Bernstein EA, et al. Renal tubular ACE-mediated tubular injury is the major contributor to microalbuminuria in early diabetic nephropathy. Am J Physiol Renal Physiol. 2018;314(4):F531–42. https://doi.org/10.1152/ajprenal.00523.2017.
Sawaf H, Thomas G, Taliercio JJ, Nakhoul G, Vachharajani TJ, Mehdi A. Therapeutic Advances in Diabetic Nephropathy. J Clin Med. 2022;11(2). doi: https://doi.org/10.3390/jcm11020378.
Chao EC, Henry RR. SGLT2 inhibition–a novel strategy for diabetes treatment. Nat Rev Drug Discov. 2010;9(7):551–9. https://doi.org/10.1038/nrd3180.
Cherney DZ, Perkins BA, Soleymanlou N, Maione M, Lai V, Lee A, et al. Renal hemodynamic effect of sodium-glucose cotransporter 2 inhibition in patients with type 1 diabetes mellitus. Circulation. 2014;129(5):587–97. https://doi.org/10.1161/circulationaha.113.005081.
Anders H-J, Huber TB, Isermann B, Schiffer M. CKD in diabetes: diabetic kidney disease versus nondiabetic kidney disease. Nat Rev Nephrol. 2018;14(6):361–77. https://doi.org/10.1038/s41581-018-0001-y.
Pirklbauer M, Schupart R, Fuchs L, Staudinger P, Corazza U, Sallaberger S, et al. Unraveling reno-protective effects of SGLT2 inhibition in human proximal tubular cells. American Journal of Physiology-Renal Physiology. 2019;316(3):F449–62. https://doi.org/10.1152/ajprenal.00431.2018.
Bauersachs J, Jaisser F, Toto R. Mineralocorticoid receptor activation and mineralocorticoid receptor antagonist treatment in cardiac and renal diseases. Hypertension. 2015;65(2):257–63. https://doi.org/10.1161/HYPERTENSIONAHA.114.04488.
Agarwal R, Kolkhof P, Bakris G, Bauersachs J, Haller H, Wada T, et al. Steroidal and non-steroidal mineralocorticoid receptor antagonists in cardiorenal medicine. Eur Heart J. 2021;42(2):152–61. https://doi.org/10.1093/eurheartj/ehaa736.
Haller H, Bertram A, Stahl K, Menne J. Finerenone: a New Mineralocorticoid Receptor Antagonist Without Hyperkalemia: an Opportunity in Patients with CKD? Curr Hypertens Rep. 2016;18(5):41. https://doi.org/10.1007/s11906-016-0649-2.
Ito S, Shikata K, Nangaku M, Okuda Y, Sawanobori T. Efficacy and Safety of Esaxerenone (CS-3150) for the treatment of type 2 diabetes with microalbuminuria: a randomized, double-blind, placebo-controlled, phase II Trial. Clin J Am Soc Nephrol. 2019;14(8):1161–72. https://doi.org/10.2215/cjn.14751218.
Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, et al. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med. 2020;383(23):2219–29. https://doi.org/10.1056/NEJMoa2025845.
Tomita I, Kume S, Sugahara S, Osawa N, Yamahara K, Yasuda-Yamahara M, et al. SGLT2 inhibition mediates protection from diabetic kidney disease by promoting ketone body-induced mTORC1 Inhibition. Cell Metab. 2020;32(3):404-19.e6. https://doi.org/10.1016/j.cmet.2020.06.020.
Lattenist L, Lechner SM, Messaoudi S, Le Mercier A, El Moghrabi S, Prince S, et al. Nonsteroidal mineralocorticoid receptor antagonist finerenone protects against acute kidney injury-mediated chronic kidney disease: role of oxidative stress. Hypertension. 2017;69(5):870–8. https://doi.org/10.1161/hypertensionaha.116.08526.
Acknowledgements
None.
Funding
This work was supported by grants from the Japan Society for the Promotion of Science (Grant-in-Aid for Scientific Research 18K15984) to KS.
Author information
Authors and Affiliations
Contributions
Research idea and study design: FF and ME; Drafting the manuscript: FF; Revising the manuscript: ME, data analysis/interpretation: FF and ME; statistical analysis: FF; supervision or mentorship: HT, TU, HT, RF, MN, TK, KTa, KM, KO, MM, KS, and KTs. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions on the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
All authors have nothing to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Fukata, F., Eriguchi, M., Tamaki, H. et al. Differential impact of glomerular and tubule-interstitial histological changes on kidney outcome between non-proteinuric and proteinuric diabetic nephropathy. Clin Exp Nephrol 28, 282–292 (2024). https://doi.org/10.1007/s10157-023-02433-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-023-02433-y