Abstract
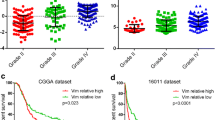
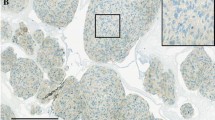
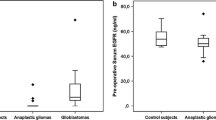
Early diagnosis of glioma is of great value to improve prognosis. We focused on serum vimentin levels as a useful biomarker for preoperative diagnosis. The aim of this study was to determine whether serum vimentin levels in patients with glioma are significantly higher than those of healthy adult volunteer and whether the serum vimentin level is associated with overall survival (OS) in patients with glioblastoma (GBM). This study included 52 consecutive patients with newly diagnosed glioma and a control group of 13 healthy adult volunteers. We measured serum vimentin levels in blood samples obtained from patients with glioma preoperatively and a control group. Furthermore, we investigated the correlation between serum vimentin levels and OS in patients with GBM. The serum vimentin levels of patients with glioma were significantly higher than those of the control group. The serum vimentin level of 2.9 ng/ml was the optimal value for differentiating patients with glioma from the control group with a sensitivity of 92.3% and specificity of 88.5%. The serum vimentin levels correlated significantly with immunoreactivity for survivin. In 27 patients with GBM, serum vimentin levels (cutoff value, median value 53.3 ng/ml) correlated with OS in univariate and multivariate analyses. Our study revealed that serum vimentin levels of patients with glioma are significantly higher than those of the control group. Therefore, we believe that serum vimentin level might be a useful and practical biomarker for preoperative diagnosis of glioma. Furthermore, high serum vimentin levels correlated significantly with shorter OS in patients with GBM.




Similar content being viewed by others
Data availability
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Aggarwal S, Singh B, Sharma SC, Das SN (2022) Circulating vimentin over-expression in patients with oral sub mucosal fibrosis and oral squamous cell carcinoma. Indian J Otolaryngol Head Neck Surg 74:510–515. https://doi.org/10.1007/s12070-021-03018-5
Andersson KM, Svensson MN, Erlandsson MC, Jonsson IM, Bokarewa MI (2015) Down-regulation of survivin alleviates experimental arthritis. J Leukoc Biol 97:135–145. https://doi.org/10.1189/jlb.3A0714-317R
Bonotti A, Simonini S, Pantani E, Giusti L, Donadio E, Mazzoni MR, Chella A, Marconi L, Ambrosino N, Lucchi M, Mussi A, Cristaudo A, Foddis R (2017) Serum mesothelin, osteopontin and vimentin: useful markers for clinical monitoring of malignant pleural mesothelioma. Int J Biol Mark 32:e126–e131. https://doi.org/10.5301/jbm.5000229
Brain Tumor Registry of Japan (2005–2008) (2017) Neurol Med Chir (Tokyo) 57:9–102. https://doi.org/10.2176/nmc.sup.2017-0001
Chen MH, Lu C, Sun J, Chen XD, Dai JX, Cai JY, Chen XL (2016) Diagnostic and prognostic value of serum vitronectin levels in human glioma. J Neurol Sci 371:54–59. https://doi.org/10.1016/j.jns.2016.10.022
DeWitt JC, Mock A, Louis DN (2017) The 2016 WHO classification of central nervous system tumors: what neurologists need to know. Curr Opin Neurol 30:643–649. https://doi.org/10.1097/WCO.0000000000000490
Egger J, Kapur T, Fedorov A, Pieper S, Miller JV, Veeraraghavan H, Freisleben B, Golby AJ, Nimsky C, Kikinis R (2013) GBM volumetry using the 3D Slicer medical image computing platform. Sci Rep 3:1364. https://doi.org/10.1038/srep01364
Franke WW, Schmid E, Osborn M, Weber K (1978) Different intermediate-sized filaments distinguished by immunofluorescence microscopy. Proc Natl Acad Sci U S A 75:5034–5038. https://doi.org/10.1073/pnas.75.10.5034
Fuchs E, Weber K (1994) Intermediate filaments: structure, dynamics, function, and disease. Annu Rev Biochem 63:345–382. https://doi.org/10.1146/annurev.bi.63.070194.002021
Fujii Y, Muragaki Y, Maruyama T, Nitta M, Saito T, Ikuta S, Iseki H, Hongo K, Kawamata T (2018) Threshold of the extent of resection for WHO Grade III gliomas: retrospective volumetric analysis of 122 cases using intraoperative MRI. J Neurosurg 129:1–9. https://doi.org/10.3171/2017.3.JNS162383
Fukui A, Muragaki Y, Saito T, Maruyama T, Nitta M, Ikuta S, Kawamata T (2017) Volumetric analysis using low-field intraoperative magnetic resonance imaging for 168 newly diagnosed supratentorial glioblastomas: effects of extent of resection and residual tumor volume on survival and recurrence. World Neurosurg 98:73–80. https://doi.org/10.1016/j.wneu.2016.10.109
Hilbert M, Kuzman P, Mueller WC, Meixensberger J, Nestler U (2022) Feasibility of ApoC1 serum levels as tumor biomarker in glioblastoma patients: a pilot study. Sci Rep 12:16981. https://doi.org/10.1038/s41598-022-21216-1
Huo Y, Zheng Z, Chen Y, Wang Q, Zhang Z, Deng H (2016) Downregulation of vimentin expression increased drug resistance in ovarian cancer cells. Oncotarget 7:45876–45888. https://doi.org/10.18632/oncotarget.9970
Ivaska J, Pallari HM, Nevo J, Eriksson JE (2007) Novel functions of vimentin in cell adhesion, migration, and signaling. Exp Cell Res 313:2050–2062. https://doi.org/10.1016/j.yexcr.2007.03.040
Lai NS, Wu DG, Fang XG, Lin YC, Chen SS, Li ZB, Xu SS (2015) Serum microRNA-210 as a potential noninvasive biomarker for the diagnosis and prognosis of glioma. Br J Cancer 112:1241–1246. https://doi.org/10.1038/bjc.2015.91
Lan F, Yue X, Xia T (2020) Exosomal microRNA-210 is a potentially non-invasive biomarker for the diagnosis and prognosis of glioma. Oncol Lett 19:1967–1974. https://doi.org/10.3892/ol.2020.11249
Lilina AV, Leekens S, Hashim HM, Vermeire PJ, Harvey JN, Strelkov SV (2022) Stability profile of vimentin rod domain. Protein Sci 31:e4505. https://doi.org/10.1002/pro.4505
Liu Y, Zhang J, Wang D, Yang X (2019) Serum ROCK1 mRNA is of great diagnostic value for glioma patients. Medicine (Baltimore) 98:e15413. https://doi.org/10.1097/MD.0000000000015413
Mahotka C, Liebmann J, Wenzel M, Suschek CV, Schmitt M, Gabbert HE, Gerharz CD (2002) Differential subcellular localization of functionally divergent survivin splice variants. Cell Death Differ 9:1334–1342. https://doi.org/10.1038/sj.cdd.4401091
Maruno M, Yoshimine T, Ushio Y, Hayakawa T, Jamshidi J, Arita N, Bitoh S, Mogami H (1987) Immunohistochemical study of human brain tumors with vimentin and astroprotein (GFAP). No To Shinkei 39:579–585
Morokoff A, Jones J, Nguyen H, Ma C, Lasocki A, Gaillard F, Bennett I, Luwor R, Stylli S, Paradiso L, Koldej R, Paldor I, Molania R, Speed TP, Webb A, Infusini G, Li J, Malpas C, Kalincik T, Drummond K, Siegal T, Kaye AH (2020) Serum microRNA is a biomarker for post-operative monitoring in glioma. J Neurooncol 149:391–400. https://doi.org/10.1007/s11060-020-03566-w
Ostrom QT, Gittleman H, Truitt G, Boscia A, Kruchko C, Barnholtz-Sloan JS (2018) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2011–2015. Neuro Oncol 20:iv1–iv86. https://doi.org/10.1093/neuonc/noy131
Qu S, Chen Z, Liu B, Liu J, Wang H (2021) N6-methyladenine-related genes affect biological behavior and the prognosis of glioma. Cancer Med 10:98–108. https://doi.org/10.1002/cam4.3574
Qu S, Qiu O, Hu Z (2021) The prognostic factors and nomogram for patients with high-grade gliomas. Fundam Res 1:824–828. https://doi.org/10.1016/j.fmre.2021.07.005
Ramos I, Stamatakis K, Oeste CL, Perez-Sala D (2020) Vimentin as a multifaceted player and potential therapeutic target in viral infections. Int J Mol Sci 21. https://doi.org/10.3390/ijms21134675
Sah NK, Khan Z, Khan GJ, Bisen PS (2006) Structural, functional and therapeutic biology of survivin. Cancer Lett 244:164–171. https://doi.org/10.1016/j.canlet.2006.03.007
Saito T, Arifin MT, Hama S, Kajiwara Y, Sugiyama K, Yamasaki F, Hidaka T, Arita K, Kurisu K (2007) Survivin subcellular localization in high-grade astrocytomas: simultaneous expression in both nucleus and cytoplasm is negative prognostic marker. J Neurooncol 82:193–198. https://doi.org/10.1007/s11060-006-9267-1
Saito T, Maruyama T, Muragaki Y, Tanaka M, Nitta M, Shinoda J, Aki T, Iseki H, Kurisu K, Okada Y (2013) 11C-methionine uptake correlates with combined 1p and 19q loss of heterozygosity in oligodendroglial tumors. AJNR Am J Neuroradiol 34:85–91. https://doi.org/10.3174/ajnr.A3173
Saito T, Muragaki Y, Maruyama T, Komori T, Nitta M, Tsuzuki S, Fukui A, Kawamata T (2019) Influence of wide opening of the lateral ventricle on survival for supratentorial glioblastoma patients with radiotherapy and concomitant temozolomide-based chemotherapy. Neurosurg Rev. https://doi.org/10.1007/s10143-019-01185-2
Saito T, Muragaki Y, Maruyama T, Tamura M, Nitta M, Tsuzuki S, Konishi Y, Kamata K, Kinno R, Sakai KL, Iseki H, Kawamata T (2016) Difficulty in identification of the frontal language area in patients with dominant frontal gliomas that involve the pars triangularis. J Neurosurg 125:803–811. https://doi.org/10.3171/2015.8.JNS151204
Saito T, Muragaki Y, Shioyama T, Komori T, Maruyama T, Nitta M, Yasuda T, Hosono J, Okamoto S, Kawamata T (2019) Malignancy index using intraoperative flow cytometry is a valuable prognostic factor for glioblastoma treated with radiotherapy and concomitant temozolomide. Neurosurgery 84:662–672. https://doi.org/10.1093/neuros/nyy089
Saito T, Muragaki Y, Tamura M, Maruyama T, Nitta M, Tsuzuki S, Fukuchi S, Ohashi M, Kawamata T (2019) Awake craniotomy with transcortical motor evoked potential monitoring for resection of gliomas in the precentral gyrus: utility for predicting motor function. J Neurosurg 1–11. https://doi.org/10.3171/2018.11.JNS182609
Saito T, Sugiyama K, Takeshima Y, Amatya VJ, Yamasaki F, Takayasu T, Nosaka R, Muragaki Y, Kawamata T, Kurisu K (2018) Prognostic implications of the subcellular localization of survivin in glioblastomas treated with radiotherapy plus concomitant and adjuvant temozolomide. J Neurosurg 128:679–684. https://doi.org/10.3171/2016.11.JNS162326
Sun S, Poon RT, Lee NP, Yeung C, Chan KL, Ng IO, Day PJ, Luk JM (2010) Proteomics of hepatocellular carcinoma: serum vimentin as a surrogate marker for small tumors (<or=2 cm). J Proteome Res 9:1923–1930. https://doi.org/10.1021/pr901085z
Turner JD, Williamson R, Almefty KK, Nakaji P, Porter R, Tse V, Kalani MY (2010) The many roles of microRNAs in brain tumor biology. Neurosurg Focus 28:E3. https://doi.org/10.3171/2009.10.FOCUS09207
van Asperen JV, Fedorushkova DM, Robe P, Hol EM (2022) Investigation of glial fibrillary acidic protein (GFAP) in body fluids as a potential biomarker for glioma: a systematic review and meta-analysis. Biomarkers 27:1–12. https://doi.org/10.1080/1354750X.2021.2006313
Wei X, Chen D, Lv T, Li G, Qu S (2016) Serum MicroRNA-125b as a potential biomarker for glioma diagnosis. Mol Neurobiol 53:163–170. https://doi.org/10.1007/s12035-014-8993-1
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507. https://doi.org/10.1056/NEJMra0708126
Willems E, Dedobbeleer M, Digregorio M, Lombard A, Goffart N, Lumapat PN, Lambert J, Van den Ackerveken P, Szpakowska M, Chevigne A, Scholtes F, Rogister B (2019) Aurora A plays a dual role in migration and survival of human glioblastoma cells according to the CXCL12 concentration. Oncogene 38:73–87. https://doi.org/10.1038/s41388-018-0437-3
Yang F, Ma J, Wan J, Ha W, Fang C, Lu H, Zhang W (2020) Epithelial-mesenchymal transition of circulating tumor cells in prostate cancer is promoted by survivin. J Int Med Res 48:300060519892395. https://doi.org/10.1177/0300060519892395
Zhao J, Zhang L, Dong X, Liu L, Huo L, Chen H (2018) High Expression of vimentin is associated with progression and a poor outcome in glioblastoma. Appl Immunohistochem Mol Morphol 26:337–344. https://doi.org/10.1097/PAI.0000000000000420
Acknowledgements
We thank Takashi Sakayori, Department of Pathology, Tokyo Women’s Medical University, Tokyo, Japan, for the immunohistochemical staining and Saori Okamoto, Shinjuku Hiro Clinic, Tokyo, Japan, for the ELISA method. We would like to thank Editage (www.editage.com) for the English language editing.
Funding
This study was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant No. 21K09138.
Author information
Authors and Affiliations
Contributions
Conceptualization: Taiichi Saito, Yoshihiro Muragaki; formal analysis and investigation: Asuka Komori, Masayuki Nitta; writing—original draft preparation: Taiichi Saito; writing—review and editing: Yoshihiro Muragaki, Taiichi Saito, Takakazu Kawamata; funding acquisition: Taiichi Saito; resources: Shunsuke Tsuzuki, Shunichi Koriyama, Bunto Ro; visualization: Shunsuke Tsuzuki, Shunichi Koriyama; supervision: Takakazu Kawamata; approved the final version: all authors.
Corresponding author
Ethics declarations
Ethics approval
Ethical approval was obtained from the ethics committee at Tokyo Women’s Medical University (No. 2621-R2).
Consent to participate
We obtained informed consent from all patients and healthy adult volunteers. To protect patient privacy, we removed all identifiers from our records on the completion of our analyses.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saito, T., Muragaki, Y., Komori, A. et al. Increase in serum vimentin levels in patients with glioma and its correlation with prognosis of patients with glioblastoma. Neurosurg Rev 46, 202 (2023). https://doi.org/10.1007/s10143-023-02112-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10143-023-02112-2