Abstract
Background and purpose
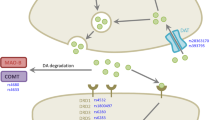
Polymorphisms of the catechol-O-methyl transferase (COMT) or monoamine oxidase B (MAO-B) genes may affect the occurrence of dyskinesia in Parkinson’s disease (PD) patients. However, the findings are inconsistent. Thus, we performed a meta-analysis to assess whether COMT and MAO-B genetic variants are associated with an increased incidence of levodopa-induced dyskinesia (LID) in PD patients.
Methods
A literature search of PubMed, Embase, and Cochrane Library was conducted to identify relevant studies published up to January 2021. The strength of the association between the polymorphisms and LID susceptibility was estimated by odds ratio (OR) and associated 95% confidence interval (CI). The pooled ORs were assessed in different genetic models.
Results
Ten studies involving 2385 PD patients were included in the meta-analysis. Analysis of pooled ORs and 95% CIs suggested that the AA genotype of COMT(rs4680) was associated with LID (OR = 1.39, 95%CI: 1.02–1.89, P = 0.039) in the recessive model, and this correlation was more obvious in Brazilian samples in the analysis stratified by ethnicity. For the AG genotype of MAO-B(rs1799836), the pooled OR was 1.66 (95% CI: 1.04–2.65, P = 0.03) in patients with LID versus those without LID in the heterozygote model.
Conclusions
Our meta-analysis implicates the AA genotype of the COMT rs4680 polymorphism as potentially increasing the risk of LID in a recessive genetic model for PD patients. Furthermore, the AG genotype of the MAO-B rs1799836 polymorphism may influence the prevalence of LID in PD patients in the heterozygote model. However, further well-designed studies with larger PD patient cohorts are required to validate these results after adjusting for confounding factors.




Similar content being viewed by others
References
Kalia LV, Lang AE (2015) Parkinson’s disease. Lancet 386(9996):896–912
Tambasco N, Romoli M, Calabresi P (2018) Levodopa in Parkinson’s disease: current status and future developments. Curr Neuropharmacol 16(8):1239–1252
Ondo WG (2011) Motor complications in Parkinson’s disease. Int J Neurosci 121(Suppl 2):37–44
Péchevis M, Clarke CE, Vieregge P et al (2005) Effects of dyskinesias in Parkinson’s disease on quality of life and health-related costs: a prospective European study. Eur J Neurol 12(12):956–963
Tran TN, Vo TNN, Frei K, Truong DD (2018) Levodopa-induced dyskinesia: clinical features, incidence, and risk factors. J Neural Transm 125(8):1109–1117
Fahn S, Oakes D, Shoulson I et al (2004) Levodopa and the progression of Parkinson’s disease. N Engl J Med 351(24):2498–2508
Eusebi P, Romoli M, Paoletti FP, Tambasco N, Calabresi P, Parnetti L (2018) Risk factors of levodopa-induced dyskinesia in Parkinson’s disease: results from the PPMI cohort. NPJ Parkinsons Dis 4:33
Strong JA, Dalvi A, Revilla FJ et al (2006) Genotype and smoking history affect risk of levodopa-induced dyskinesias in Parkinson’s disease. Mov Disord 21(5):654–659
Rieck M, Schumacher-Schuh AF, Altmann V et al (2012) DRD2 haplotype is associated with dyskinesia induced by levodopa therapy in Parkinson’s disease patients. Pharmacogenomics 13(15):1701–1710
Lee JY, Cho J, Lee EK, Park SS, Jeon BS (2011) Differential genetic susceptibility in diphasic and peak-dose dyskinesias in Parkinson’s disease. Mov Disord 26(1):73–79
Talkowski ME, McCann KL, Chen M et al (2010) Fine-mapping reveals novel alternative splicing of the dopamine transporter. Am J Med Genet Part B Neuropsychiatr Genet 153B(8):1434–1447
Kaiser R, Hofer A, Grapengiesser A (2003) L -Dopa-induced adverse effects in PD and dopamine transporter gene polymorphism. Neurology 60(11):1750–1755
Watanabe M, Harada S, Nakamura T et al (2003) Association between catechol-O-methyltrans ferase gene polymorphisms and wearing-off and dyskinesia in Parkinson’s disease. Neuropsychobiology 48(4):190–193
Contin M, Martinelli P, Mochi M, Riva R, Albani F, Baruzzi A (2005) Genetic polymorphism of catechol-O-methyltrans ferase and levodopa pharmacokinetic-pharmacodynamic pattern in patients with Parkinson’s disease. Mov Disord 20(6):734–739
Torkaman-Boutorabi A, Shahidi GA, Choopani S et al (2012) The catechol-O-methyltransferase and monoamine oxidase B polymorphisms and levodopa therapy in the Iranian patients with sporadic Parkinson’s disease. Acta Neurobiol Exp (Wars) 72(3):272–82
Hao H, Shao M, An J et al (2014) Association of catechol-O-methyltransferase and monoamine oxidase B gene polymorphisms with motor complications in parkinson’s disease in a Chinese population. Parkinsonism Relat Disord 20(10):1041–1045
Cheshire P, Bertram K, Ling H et al (2013) Influence of single nucleotide polymorphisms in COMT, MAO-A and BDNF genes on dyskinesias and levodopa use in Parkinson’s disease. Neurodegener Dis 13(1):24–28
Xiao Q, Qian Y, Liu J, Xu S, Yang X (2017) Roles of functional catechol-O-methyltransferase genotypes in Chinese patients with Parkinson’s disease. Transl Neurodegener 6:11
Sampaio TF, Dos Santos EUD, de Lima GDC et al (2018) MAO-B and COMT genetic variations associated with levodopa treatment response in patients with Parkinson’s disease. J Clin Pharmacol 58(7):920–926
Kakinuma S, Beppu M, Sawai S et al (2020) Monoamine oxidase B rs1799836 G allele polymorphism is a risk factor for early development of levodopa-induced dyskinesia in Parkinson’s disease. eNeurologicalSci 19:100239
Dos Santos EUD, da Silva IIFG, Asano AGC, Asano NMJ, De MascenaDiniz Maia M, de Souza PRE (2020) Pharmacogenetic profile and the development of the dyskinesia induced by levodopa-therapy in Parkinson’s disease patients: a population-based cohort study. Mol Biol Rep 47(11):8997–9004
Zhao C, Wang Y, Zhang B, Yue Y, Zhang J (2020) Genetic variations in catechol-O-methyltransferase gene are associated with levodopa response variability in Chinese patients with Parkinson’s disease. Sci Rep 10(1):9521
Białecka M, Droździk M, Kłodowska-Duda G et al (2004) The effect of monoamine oxidase B (MAOB) and catechol-O- methyltransferase (COMT) polymorphisms on levodopa therapy in patients with sporadic Parkinson’s disease. Acta Neurol Scand 110(4):260–266
Bialecka M, Kurzawski M, Klodowska-Duda G, Opala G, Tan EK, Drozdzik M (2008) The association of functional catechol-O- methyltransferase haplotypes with risk of Parkinsons disease, levodopa treatment response, and complications. Pharmacogenet Genomics 18(9):815–821
de Lau LM, Verbaan D, Marinus J, Heutink P, van Hilten JJ (2012) Catechol-O-methyltransferase Val158Met and the risk of dyskinesias in Parkinson’s disease. Mov Disord 27(1):132–135
Ziegler DA, Ashourian P, Wonderlick JS et al (2014) Associations with COMT and DRD2 polymorphisms. Scand J Psychol 55(3):278–286
Ivanova SA, Alifirova VM, Pozhidaev IV et al (2018) Polymorphisms of catechol-O-methyl transferase (COMT) gene in vulnerability to levodopa-induced dyskinesia. J Pharm Pharm Sci 21(1):340–346
Redenšek S, Flisar D, Kojović M et al (2019) Dopaminergic pathway genes influence adverse events related to dopaminergic treatment in Parkinson’s disease. Front Pharmacol 10:8
Michałowska M, Chalimoniuk M, Jówko E et al (2020) Gene polymorphisms and motor levodopa-induced complications in Parkinson’s disease. Brain Behav 10(3):e01537
Yin B, Chen Y, Zhang L (2014) Association between catechol-O-methyltransferase (COMT) gene polymorphisms, Parkinson’s disease, and levodopa efficacy. Mol Diagnosis Ther 18:253–260
Falla M, Di Fonzo A, Hicks AA, Pramstaller PP, Fabbrini G (2021) Genetic variants in levodopa-induced dyskinesia (LID): a systematic review and meta-analysis. Parkinsonism Relat Disord 84:52–60
Thakkinstian A, McElduff P, D’Este C, Duffy D, Attia J (2005) A method for meta-analysis of molecular association studies. Stat Med 24(9):1291–1306
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558
Egger M, Smith GD, Phillips AN (1997) Meta-analysis: principles and procedures. Br Med J 315(7121):1533–1537
Chen J, Lipska BK, Halim N et al (2004) Functional analysis of genetic variation in catechol-O-methyltransferase (COMT): effects on mRNA, protein, and enzyme activity in postmortem human brain. Am J Hum Genet 75(5):807–821
de la Fuente-Fernández R, Sossi V, Huang Z et al (2004) Levodopa-induced changes in synaptic dopamine levels increase with progression of Parkinson’s disease: implications for dyskinesias. Brain 127(Pt 12):2747–2754
Garpenstrand H, Ekblom J, Forslund K, Rylander G, Oreland L (2000) Platelet monoamine oxidase activity is related to MAOB intron 13 genotype. J Neural Transm (Vienna) 107(5):523–30
Costa-Mallen P, Afsharinejad Z, Kelada SN et al (2004) DNA sequence analysis of monoamine oxidase B gene coding and promoter regions in Parkinson’s disease cases and unrelated controls. Mov Disord 19(1):76–83
Balciuniene J, Emilsson L, Oreland L, Pettersson U, Jazin E (2002) Investigation of the functional effect of monoamine oxidase polymorphisms in human brain. Hum Genet 110(1):1–7
Young WF Jr, Laws ER Jr, Sharbrough FW, Weinshilboum RM (1986) Human monoamine oxidase. Lack of brain and platelet correlation. Arch Gen Psychiatry 43(6):604–9
Talati R, Reinhart K, Baker W, White CM, Coleman CI (2009) Pharmacologic treatment of advanced Parkinson’s disease: a meta-analysis of COMT inhibitors and MAO-B inhibitors. Park Relat Disord 15(7):500–505
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Not involved Ethical content.
Conflict of interest
The authors declare no competing interests.
Informed consent
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yin, Y., Liu, Y., Xu, M. et al. Association of COMT rs4680 and MAO-B rs1799836 polymorphisms with levodopa-induced dyskinesia in Parkinson’s disease—a meta-analysis. Neurol Sci 42, 4085–4094 (2021). https://doi.org/10.1007/s10072-021-05509-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-021-05509-3