Abstract
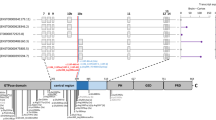
GNAO1 variants are associated with a wide range of neurodevelopmental disorders including epileptic encephalopathies and movement disorders. It has been reported that some GNAO1 variants are associated with movement disorders, and the 207–246 amino acid region was proposed as a mutational hotspot. Here, we report an intronic variant (NM_020988.3:c.724-8G>A) in GNAO1 in a Japanese girl who showed mild developmental delay and movement disorders including dystonia and myoclonus. Her movement disorders were improved by deep brain stimulation treatment as previously reported. This variant has been recurrently reported in four patients and was transmitted from her mother who possessed the variant as low-prevalent mosaicism. Using RNA extracted from lymphoblastoid cells derived from the patient, we demonstrated that the variant caused abnormal splicing of in-frame 6-bp intronic retention, leading to 2 amino acid insertion (p.Thr241_Asn242insProGln). Immunoblotting and immunostaining using WT and mutant GNAO1 vectors showed no significant differences in protein expression levels, but the cellular localization pattern of this mutant was partially shifted to the cytoplasm whereas WT was exclusively localized in the cellular membrane. Our report first clarified abnormal splicing and resulting mutant protein caused by the c.724-8G>A variant.


Similar content being viewed by others
Data accessibility statement
Genetic data is not available.
References
Feng H, Khalil S, Neubig RR, Sidiropoulos C (2018) A mechanistic review on GNAO1-associated movement disorder. Neurobiol Dis 08(116):131–141. https://doi.org/10.1016/j.nbd.2018.05.005
Worley PF, Baraban JM, Van Dop C et al (1986) Go, a guanine nucleotide-binding protein: immunohistochemical localization in rat brain resembles distribution of second messenger systems. Proc Natl Acad Sci U S A 83(12):4561–4565. https://doi.org/10.1073/pnas.83.12.4561
Solis GP, Bilousov O, Koval A, Lüchtenborg AM, Lin C, Katanaev VL (2017) Golgi-resident gαo promotes protrusive membrane dynamics. Cell 170(5):939-955.e24. https://doi.org/10.1016/j.cell.2017.07.015
Abela L, Kurian MA (2018) Postsynaptic movement disorders: clinical phenotypes, genotypes, and disease mechanisms. J Inherit Metab Dis 41(6):1077–1091. https://doi.org/10.1007/s10545-018-0205-0
Feng H, Sjögren B, Karaj B et al (2017) Movement disorder in gnao1 encephalopathy associated with gain-of-function mutations. Neurology 89(8):762–770. https://doi.org/10.1212/wnl.0000000000004262
Nakamura K, Kodera H, Akita T et al (2013) De novo mutations in GNAO1, encoding a gαo subunit of heterotrimeric g proteins, cause epileptic encephalopathy. Am J Hum Genet 93(3):496–505. https://doi.org/10.1016/j.ajhg.2013.07.014
Kelly M, Park M, Mihalek I et al (2019) Spectrum of neurodevelopmental disease associated with the GNAO1 guanosine triphosphate-binding region. Epilepsia 60(3):406–418. https://doi.org/10.1111/epi.14653
Schirinzi T, Garone G, Travaglini L et al (2019) Phenomenology and clinical course of movement disorder in GNAO1 variants: results from an analytical review. Parkinsonism Relat Disord 04(61):19–25. https://doi.org/10.1016/j.parkreldis.2018.11.019
Danti FR, Galosi S, Romani M et al (2017) GNAO1 encephalopathy: broadening the phenotype and evaluating treatment and outcome. Neurol Genet 3(2):e143. https://doi.org/10.1212/nxg.0000000000000143
Muntean BS, Masuho I, Dao M et al (2021) Gαo is a major determinant of cAMP signaling in the pathophysiology of movement disorders. Cell Rep 34(5):108718. https://doi.org/10.1016/j.celrep.2021.108718
Di Rocco M, Galosi S, Lanza E et al (2021) Caenorhabditis elegans provides an efficient drug screening platform for GNAO1-related disorders and highlights the potential role of caffeine in controlling dyskinesia. Hum Mol Genet. https://doi.org/10.1093/hmg/ddab296 (ahead of print)
Benabid A, Pollak P, Hoffmann D et al (1991) Long-term suppression of tremor by chronic stimulation of the ventral intermediate thalamic nucleus. Lancet 337(8738):403–406. https://doi.org/10.1016/0140-6736(91)91175-t
Rodrigues FB, Duarte GS, Prescott D, Ferreira J, Costa J (2019) Deep brain stimulation for dystonia. Cochrane Database Sys Rev 1(1):CD012405. https://doi.org/10.1002/14651858.cd012405.pub2
Koy A, Cirak S, Gonzalez V et al (2018) Deep brain stimulation is effective in pediatric patients with GNAO1 associated severe hyperkinesia. J Neurol Sci 391:31–39. https://doi.org/10.1016/j.jns.2018.05.018
Yamashita Y, Ogawa T, Ogaki K et al (2020) Neuroimaging evaluation and successful treatment by using directional deep brain stimulation and levodopa in a patient with gnao1-associated movement disorder: a case report. J Neurol Sci 411:116710. https://doi.org/10.1016/j.jns.2020.116710
Watanabe K, Nakashima M, Kumada S et al (2021) Identification of two novel de novo TUBB variants in cases with brain malformations: case reports and literature review. J Hum Genet 66(12):1193–1197. https://doi.org/10.1038/s10038-021-00956-4
Retterer K, Juusola J, Cho MT et al (2016) Clinical application of whole-exome sequencing across clinical indications. Genet Med 18(7):696–704. https://doi.org/10.1038/gim.2015.148
Monies D, Abouelhoda M, Assoum M et al (2019) Lessons learned from large-scale, first-tier clinical exome sequencing in a highly consanguineous population. Am J Hum Genet 104(6):1182–1201. https://doi.org/10.1016/j.ajhg.2019.04.011
Yang X, Niu X, Yang Y et al (2021) Phenotypes of GNAO1 variants in a Chinese cohort. Front Neurol 12:662162. https://doi.org/10.3389/fneur.2021.662162
Garcia-Marcos M, Ghosh P, Farquhar MG (2011) Molecular basis of a novel oncogenic mutation in GNAO1. Oncogene 30(23):2691–2696. https://doi.org/10.1038/onc.2010.645
Coubes P, Roubertie A, Vayssiere N, Hemm S, Echenne B (2000) Treatment of DYT1-generalised dystonia by stimulation of the internal globus pallidus. Lancet 355(9222):2220–2221. https://doi.org/10.1016/s0140-6736(00)02410-7
Tisch S, Kumar KR (2021) Pallidal deep brain stimulation for monogenic dystonia: the effect of gene on outcome. Front Neurol 11:630391. https://doi.org/10.3389/fneur.2020.630391
Møller RS, Liebmann N, Larsen LHG et al (2019) Parental mosaicism in epilepsies due to alleged de novo variants. Epilepsia 60(6):e63–e66. https://doi.org/10.1111/epi.15187
Myers CT, Hollingsworth G, Muir AM et al (2018) Parental mosaicism in “de novo” epileptic encephalopathies. N Engl J Med 378(17):1646–1648. https://doi.org/10.1056/nejmc1714579
Acknowledgements
We would like to thank the patient’s family for participating in this work. We also thank K. Shibazaki, M. Tsujimura, and A. Kitamoto for their technical assistance. This work was supported by the Grants-in-Aid for Scientific Research (B) (JP20H03641), HUSM Grant-in-Aid from Hamamatsu University School of Medicine, and the Takeda Science Foundation.
Author information
Authors and Affiliations
Contributions
SM, MN, and HS contributed data analysis. SM performed RT-PCR, cell culture, transfection, and immunoblotting analysis; SF and SK evaluated the patient and provided samples. HS conceived and designed the study. All authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Consent to participate
Informed consent was obtained from all individual participants included in the study and the participant has consented to the submission of the case report to the journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miyamoto, S., Nakashima, M., Fukumura, S. et al. An intronic GNAO1 variant leading to in-frame insertion cause movement disorder controlled by deep brain stimulation. Neurogenetics 23, 129–135 (2022). https://doi.org/10.1007/s10048-022-00686-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10048-022-00686-5