Abstract
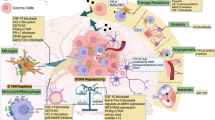
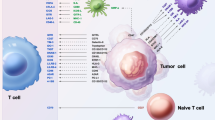
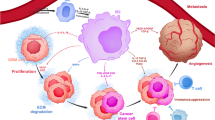
As a new concept of glioma therapy, immunotherapy combined with standard therapies is a promising modality to improve glioma patient survival. VEGF and its signaling pathway molecules not only inhibit angiogenesis but also may reinforce the immunosuppressive tumor microenvironment, including promotion of the accumulation of immunosuppressive tumor-associated macrophages (TAMs). In this review, we discuss VEGF-targeted therapy as a new treatment option of the TAM-targeted therapy for high-grade gliomas, as well as other TAM-targeted therapies. The authors also discuss the potential of these therapies combined with conventional immunotherapies.




Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Ishikawa E, Yamamoto T, Matsumura A (2017) Prospect of immunotherapy for glioblastoma: tumor vaccine, immune checkpoint inhibitors and combination therapy. Neurol Med Chir (Tokyo) 57(7):321–330
Akasaki Y, Kikuchi T, Homma S et al (2016) Phase I/II trial of combination of temozolomide chemotherapy and immunotherapy with fusions of dendritic and glioma cells in patients with glioblastoma. Cancer Immunol Immunother 65(12):1499–1509
Cloughesy TF, Mochizuki AY, Orpilla JR et al (2019) Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat Med 25(3):477–486
Ishikawa E, Muragaki Y, Yamamoto T et al (2014) Phase I/IIa trial of fractionated radiotherapy, temozolomide, and autologous formalin-fixed tumor vaccine for newly diagnosed glioblastoma. J Neurosurg 121(3):543–553
Narita Y (2015) Bevacizumab for glioblastoma. Ther Clin Risk Manag 11:1759–1765
Chen DS, Mellman I (2017) Elements of cancer immunity and the cancer-immune set point. Nature 541(7637):321–330
Miyazaki T, Ishikawa E, Matsuda M et al (2017) Assessment of PD-1 positive cells on initial and secondary resected tumor specimens of newly diagnosed glioblastoma and its implications on patient outcome. J Neurooncol 133(2):277–285
Reardon DA, Brandes AA, Omuro A et al (2020) Effect of nivolumab vs bevacizumab in patients with recurrent glioblastoma: the checkmate 143 phase 3 randomized clinical trial. JAMA Oncol 6(7):1003–1010
Zhao J, Chen AX, Gartrell RD et al (2019) Immune and genomic correlates of response to anti-PD-1 immunotherapy in glioblastoma. Nat Med 25(3):462–469
Weller M, Butowski N, Tran DD, ACT IV Trial Investigators et al (2017) Rindopepimut with temozolomide for patients with newly diagnosed, EGFRvIII-expressing glioblastoma (ACT IV): a randomised, double-blind, international phase 3 trial. Lancet Oncol 18(10):1373–1385
Hashimoto N, Tsuboi A, Kagawa N et al (2015) Wilms tumor 1 peptide vaccination combined with temozolomide against newly diagnosed glioblastoma: safety and impact on immunological response. Cancer Immunol Immunother 64(6):707–716
Johanns TM, Miller CA, Liu CJ et al (2019) Detection of neoantigen-specific T cells following a personalized vaccine in a patient with glioblastoma. Oncoimmunology 8(4):e1561106
Bota DA, Chung J, Dandekar MD et al (2018) Phase II study of ERC1671 plus bevacizumab versus bevacizumab plus placebo in recurrent glioblastoma: interim results and correlations with CD4(+) T-lymphocyte counts. CNS Oncol 7(3):CNS22
Akhavan D, Alizadeh D, Wang D et al (2019) CAR T cells for brain tumors: lessons learned and road ahead. Immunol Rev 290(1):60–84
Sekiguchi K, Ito Y, Hattori K et al (2019) VEGF receptor 1-expressing macrophages recruited from bone marrow enhances angiogenesis in endometrial tissues. Sci Rep 9(1):7037
Suzuki H, Onishi H, Wada J et al (2010) VEGFR2 is selectively expressed by FOXP3high CD4+ Treg. Eur J Immunol 40(1):197–203
Yao X, Ping Y, Liu Y et al (2013) Vascular endothelial growth factor receptor 2 (VEGFR-2) plays a key role in vasculogenic mimicry formation, neovascularization and tumor initiation by Glioma stem-like cells. PLoS ONE 8(3):e57188
Dikov MM, Ohm JE, Ray N et al (2005) Differential roles of vascular endothelial growth factor receptors 1 and 2 in dendritic cell differentiation. J Immunol 174(1):215–222
Mimura K, Kono K, Takahashi A et al (2007) Vascular endothelial growth factor inhibits the function of human mature dendritic cells mediated by VEGF receptor-2. Cancer Immunol Immunother 56(6):761–770
Chinot OL, Wick W, Mason W et al (2014) Bevacizumab plus radiotherapy-temozolomide for newly diagnosed glioblastoma. N Engl J Med 370(8):709–722
Wick W, Gorlia T, Bendszus M et al (2017) Lomustine and bevacizumab in progressive glioblastoma. N Engl J Med 377(20):1954–1963
van den Bent MJ, Klein M, Smits M et al (2018) Bevacizumab and temozolomide in patients with first recurrence of WHO grade II and III glioma, without 1p/19q co-deletion (TAVAREC): a randomised controlled phase 2 EORTC trial. Lancet Oncol 19(9):1170–1179
Montana V, Sontheimer H (2011) Bradykinin promotes the chemotactic invasion of primary brain tumors. J Neurosci 31(13):4858–4867
Uemae Y, Ishikawa E, Osuka S et al (2014) CXCL12 secreted from glioma stem cells regulates their proliferation. J Neurooncol 117(1):43–51
Kovalchuk B, Berghoff AS, Karreman MA et al (2020) Nintedanib and a bi-specific anti-VEGF/Ang2 nanobody selectively prevent brain metastases of lung adenocarcinoma cells. Clin Exp Metastasis 37(6):637–648
Sharma I, Singh A, Siraj F et al (2018) IL-8/CXCR1/2 signalling promotes tumor cell proliferation, invasion and vascular mimicry in glioblastoma. J Biomed Sci 25(1):62
Le Joncour V, Filppu P, Hyvönen M et al (2019) Vulnerability of invasive glioblastoma cells to lysosomal membrane destabilization. EMBO Mol Med 11(6):e9034
Auf G, Jabouille A, Guérit S et al (2010) Inositol-requiring enzyme 1alpha is a key regulator of angiogenesis and invasion in malignant glioma. Proc Natl Acad Sci USA 107(35):15553–15558
Krusche B, Ottone C, Clements NP et al (2016) EphrinB2 drives perivascular invasion and proliferation of glioblastoma stem-like cells. Elife 5:e14845
Griveau A, Seano G, Shelton SJ et al (2018) A glial signature and Wnt7 signaling regulate glioma-vascular interactions and tumor microenvironment. Cancer Cell 33(5):874-889.e7
Huang H, Langenkamp E, Georganaki M et al (2015) VEGF suppresses T-lymphocyte infiltration in the tumor microenvironment through inhibition of NF-kappaB-induced endothelial activation. FASEB J 29(1):227–238
Gabrilovich DI, Chen HL, Girgis KR et al (1996) Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med 2(10):1096–1103
Wheeler KC, Jena MK, Pradhan BS et al (2018) VEGF may contribute to macrophage recruitment and M2 polarization in the decidua. PLoS ONE 13(1):e0191040
Lisi L, Pia Ciotti GM et al (2020) Vascular endothelial growth factor receptor 1 in glioblastoma-associated microglia/macrophages. Oncol Rep 43(6):2083–2092
Min AKT, Mimura K, Nakajima S et al (2021) Therapeutic potential of anti-VEGF receptor 2 therapy targeting for M2-tumor-associated macrophages in colorectal cancer. Cancer Immunol Immunother 70(2):289–298
Tamura R, Tanaka T, Akasaki Y et al (2019) The role of vascular endothelial growth factor in the hypoxic and immunosuppressive tumor microenvironment: perspectives for therapeutic implications. Med Oncol 37(1):2
Voron T, Colussi O, Marcheteau E et al (2015) VEGF-A modulates expression of inhibitory checkpoints on CD8+ T cells in tumors. J Exp Med 212(2):139–148
Friebel E, Kapolou K, Unger S et al (2020) Single-cell mapping of human brain cancer reveals tumor-specific instruction of tissue-invading leukocytes. Cell 181(7):1626-1642.e20
Chuang HY, Su YK, Liu HW et al (2019) Preclinical evidence of STAT3 inhibitor pacritinib overcoming temozolomide resistance via downregulating miR-21-enriched exosomes from M2 glioblastoma-associated macrophages. J Clin Med 8(7):959
Takenaka MC, Gabriely G, Rothhammer V et al (2019) Control of tumor-associated macrophages and T cells in glioblastoma via AHR and CD39. Nat Neurosci 22(5):729–740
Kaneda MM, Messer KS, Ralainirina N et al (2016) PI3Kγ is a molecular switch that controls immune suppression. Nature 539(7629):437–442
Pyonteck SM, Akkari L, Schuhmacher AJ et al (2013) CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat Med 19(10):1264–1272
Miyazaki T, Ishikawa E, Matsuda M et al (2020) Infiltration of CD163-positive macrophages in glioma tissues after treatment with anti-PD-L1 antibody and role of PI3Kγ inhibitor as a combination therapy with anti-PD-L1 antibody in in vivo model using temozolomide-resistant murine glioma-initiating cells. Brain Tumor Pathol 37(2):41–49
Miyazaki T, Ishikawa E, Sugii N et al (2020) Therapeutic strategies for overcoming immunotherapy resistance mediated by immunosuppressive factors of the glioblastoma microenvironment. Cancers (Basel) 12(7):1960
Acknowledgements
We thank Dr. Alexander Zaboronok, Department of Neurosurgery, Faculty of Medicine of the University of Tsukuba for professional and English revision. This study was supported by a Grant-in-Aid for Scientific Research in Japan (Grant number: 18K08962).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Materials for the AFTV described in this review article were partly provided by Cell-Medicine, Inc. (CMI), a venture company for research and development of immunotherapy established by the initiative of RIKEN (The Institute of Physical and Chemical Research) and the University of Tsukuba in Japan. The authors declare that T.M. is a member of CMI, and E.I, S.T., and H.A. have no conflicts of interest regarding this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ishikawa, E., Miyazaki, T., Takano, S. et al. Anti-angiogenic and macrophage-based therapeutic strategies for glioma immunotherapy. Brain Tumor Pathol 38, 149–155 (2021). https://doi.org/10.1007/s10014-021-00402-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10014-021-00402-5