Abstract
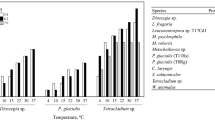
One hundred and three yeasts isolated from soil samples from King George Island and Tierra del Fuego province were screened in relation with their capability to produce pectinolytic enzymes. Although all the yeasts showed well-developed colonies at 20 °C, only eight showed a clear halo around the colony, indicative of pectin degradation. A secondary screening demonstrated that only four yeasts were capable to produce pectinases at low temperatures (8 °C). It could be seen that the selected yeasts were able to grow and produce high levels of polygalacturonase activity when submerged fermentations were performed using pectin-containing fruit wastes as substrates. None of the strains produced neither lyase nor rhamnogalacturonan hydrolase activities. Regarding pectin esterase activity, it was only produced in lower amounts by G. pullulans 8E (0.022 U ml−1). A TLC analysis of the substrate cleavage pattern of the pectinolytic systems was consistent with an endo-type activity. The clarification of apple juice was only accomplished by G. pullulans pectinolytic system, with a clarification of 80% (%T650) using 4 U/ml of enzyme at 20 °C. As far as we concern this work describes for the first time the production of pectinases by the cold-adapted yeasts species Cystofilobasidium infirmominiatum, Cryptococcus adeliensis and G. pullulans.





Similar content being viewed by others
References
Abe F, Minegishi H, Miura T et al (2006) Characterization of cold- and high-pressure-active polygalacturonases from a deep-sea yeast, Cryptococcus liquefaciens strain N6. Biosci Biotech Bioch 70:296–299
Albersheim P (1966) Pectin lyase from fungi. Methods Enzymol 8:628–631
Alkorta I, Garbisu C, MaJ Llama, Serra JL (1998) Industrial applications of pectic enzymes: a review. Process Biochem 33:21–28
Barnby FM, Morpeth FF, Pyle DL (1990) Endopolygalacturonase production from Kluyveromyces marxianus. I. Resolution, purification, and partial characterisation of the enzyme. Enzyme Microb Technol 12:891–897
Birgisson H, Delgado O, Garcia Arroyo L et al (2003) Cold-adapted yeasts as producers of cold-active polygalacturonases. Extremophiles 7:185–193
Blanco P, Sieiro C, Villa TG (1999) Production of pectic enzymes in yeasts. FEMS Microbiol Lett 175:1–9
Branda E, Turchetti B, Diolaiuti G et al (2010) Yeast and yeast-like diversity in the southernmost glacier of Europe (Calderone Glacier, Apennines, Italy). FEMS Microbiol Ecol 72:354–369
Brandão LR, Libkind D, Vaz ABM et al (2011) Yeasts from an oligotrophic lake in Patagonia (Argentina): diversity, distribution and synthesis of photoprotective compounds and extracellular enzymes. FEMS Microbiol Ecol 76:1–13
Brizzio S, Turchetti B, de Garcia V et al (2007) Extracellular enzymatic activities of basidiomycetous yeasts isolated from glacial and subglacial waters of northwest Patagonia (Argentina). Can J Microbiol 53:519–525
Buzzini P, Martini A (2002) Extracellular enzymatic activity profiles in yeast and yeast-like strains isolated from tropical environments. J Appl Microbiol 93:1020–1025
Cavalitto FS, Hours AR, Mignone FC (2000) Growth and protopectinase production of Geotrichum klebahnii in batch and continuous cultures with synthetic media. J Ind Microbiol Biotech 25:260–265
Christensen ET, Nielsen EJ, Kreiberg DJ et al (1998) Pectin methyl esterase from orange fruit: characterization and localization by in situ hybridization and immunohistochemistry. Planta 206:493–503
Connell L, Redman R, Craig S et al (2008) Diversity of soil yeasts isolated from South Victoria Land, Antarctica. Microb Ecol 56:448–459
Contreras Esquivel JC, Voget CE (2004) Purification and partial characterization of an acidic polygalacturonase from Aspergillus kawachii. J Biotechnol 110:21–28
de García V, Brizzio S, Libkind D et al (2007) Biodiversity of cold-adapted yeasts from glacial meltwater rivers in Patagonia, Argentina. FEMS Microbiol Ecol 59:331–341
DePriest T, Ivanova N, Fahselt D et al (2002) Sequences of psychrophilic fungi amplified from glacier-preserved ascolichens. Can J Bot 78:1450–1459
Dey TB, Banerjee R (2014) Application of decolourized and partially purified polygalacturonase and α-amylase in apple juice clarification. Braz J Microbiol 45:97–104
Fell JW, Hunter IL (1968) Isolation of heterothallic yeast strains of Metschnikowia kamienski and their mating reactions with Chlamydozyma wickerham spp. Antonie Van Leeuwenhoek 34:365–376
García-Carreño FL, Dimes LE, Haard NF (1993) Substrate-gel electrophoresis for composition and molecular weight of proteinases or proteinaceous proteinase inhibitors. Anal Biochem 214:65–69
Gunde-Cimerman N, Butinar L, Sonjak S et al. (2005) Halotolerant and halophilic fungi from coastal environments in the Arctics. In: Gunde-Cimerman N, Oren O, Plemenitaš A (eds) Adaptation to life at high salt concentrations in Archaea, Bacteria, and Eukarya, Springer, pp 397–423
Hoondal G, Tiwari R, Tewari R et al (2002) Microbial alkaline pectinases and their industrial applications: a review. Appl Microbiol Biotech 59:409–418
Kutty SN, Philip R (2008) Marine yeasts—a review. Yeast 25:465–483
Margesin R, Fauster V, Fonteyne PA (2005) Characterization of cold-active pectate lyases from psychrophilic Mrakia frigida. Lett Appl Microbiol 40:453–459
Martinez A, Cavello I, Garmendia G et al (2016) Yeasts from sub-Antarctic region: biodiversity, enzymatic activities and their potential as oleaginous microorganisms. Extremophiles 20:759–769
Merín MG, Morata de Ambrosini VI (2015) Highly cold-active pectinases under wine-like conditions from non-Saccharomyces yeasts for enzymatic production during winemaking. Lett Appl Microbiol 60:467–474
Morgan-Kiss RM, Priscu JC, Pocock T et al (2006) Adaptation and acclimation of photosynthetic microorganisms to permanently cold environments. Microbiol Mol Biol Rev 70:222–252
Nakagawa T, Kaichiro Y, Tatsuro M, Noboru T (2002) Cold-active pectinolytic activity of psychrophilic-basidiomycetous yeast Cystofilobasidium capitatum strain PPY-1. J Biosci Bioeng 94:175–177
Nakagawa T, Nagaoka T, Taniguchi S et al (2004) Isolation and characterization of psychrophilic yeasts producing cold-adapted pectinolytic enzymes. Lett Appl Microbiol 38:383–387
Nakagawa T, Nagaoka T, Miyaji T, Tomizuka N (2005) Cold-active polygalacturonase from psychrophilic-basidiomycetous yeast Cystofilobasidium capitatum strain PPY-1. Biosci Biotech Bioch 69:419–421
Nakkeeran E, Umesh-Kumar S, Subramanian R (2011) Aspergillus carbonarius polygalacturonases purified by integrated membrane process and affinity precipitation for apple juice production. Biores Technol 102:3293–3297
Normand J, Ralet MC, Thibault JF et al (2010) Purification, characterization, and mode of action of a rhamnogalacturonan hydrolase from Irpex lacteus, tolerant to an acetylated substrate. Appl Microbiol Biotech 86:577–588
Pedrolli DB, Gomes E, Monti R, Carmona EC (2008) Studies on productivity and characterization of polygalacturonase from Aspergillus giganteus submerged culture using citrus pectin and orange waste. Appl Biochem Biotech 144:191–200
Sakai T, Sakamoto T, Hallaert J, Vandamme EJ (1993) Pectin, pectinase, and protopectinase: production, properties, and applications. Adv Appl Microbiol 39:213–294
Sampaoi JP (2011) Cystofilobasidium Oberwinkler & Bandoni (1983). Fifth: 1429–1430
Sandri IG, Fontana RC, Barfknecht DM, da Silveira MM (2011) Clarification of fruit juices by fungal pectinases. LWT-Food Sci Technol 44:2217–2222
Slavikova E, Kosikova B, Mikulasova M (2002) Biotransformation of waste lignin products by the soil-inhabiting yeast Trichosporon pullulans. Can J Microbiol 48:200–203
Sunnotel O, Nigam P (2002) Pectinolytic activity of bacteria isolated from soil and two fungal strains during submerged fermentation. World J Microbiol Biotechnol 18:835–839
Vaz ABM, Rosa LH, Vieira MLA et al (2011) The diversity, extracellular enzymatic activities and photoprotective compounds of yeasts isolated in Antarctica. Braz J Microbiol 42:937–947
Vishniac HS (2005) Yeast biodiversity in the Antarctic. In: Peter G, Rosa C (eds) Biodiversity and ecophysiology of yeasts. Springer, pp 419–440
Whitaker JR (1984) Pectic substances, pectic enzymes and haze formation in fruit juices. Enzyme Microb Technol 6:341–349
Yukimura K, Nakai R, Kohshima S et al (2009) Spore-forming halophilic bacteria isolated from Arctic terrains: implications for long-range transportation of microorganisms. Polar Sci 3:163–169
Acknowledgements
This work was supported by grants from Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, PIP 112-201101-00662) and Agencia Nacional de Promoción Científica y Tecnológica (PICT 2014-1655).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no commercial or financial conflict of interest.
Additional information
Communicated by A. Driessen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
792_2016_904_MOESM2_ESM.tif
Supplementary Fig. 2. Effect of carbon sources on PGase production. PGase activity (U/ml) produced in a culture medium containing 10 g l−1 citrus pectin (dark gray color box), 5 g l−1 glucose and 0.5 g l−1 citrus pectin (light gray color box) and 10 g l−1 glucose (medium gray color box) (TIFF 71 kb)
Rights and permissions
About this article
Cite this article
Cavello, I., Albanesi, A., Fratebianchi, D. et al. Pectinolytic yeasts from cold environments: novel findings of Guehomyces pullulans, Cystofilobasidium infirmominiatum and Cryptococcus adeliensis producing pectinases. Extremophiles 21, 319–329 (2017). https://doi.org/10.1007/s00792-016-0904-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-016-0904-0