Abstract
Objectives
Platelet-rich fibrin (PRF) in liquid form has shown advantages in tissue engineering including acting as injectable fillers and drug carriers. However, few studies have investigated the best relative centrifugal force (RCF) for preparing liquid PRF. The aim of the present study was to find out optimal centrifugation force for preparing liquid PRF.
Materials and methods
Liquid PRF was prepared using horizontal centrifugation (liquid H-PRF) with RCF ranging from 100 g, 300 g, 500 g, to 700 g for 8 min. The volume, weight, solidification time, and tensile properties were subsequently investigated. Scanning electron microscopy (SEM) and rheologic tests were carried out to investigate the microstructure and rheologic properties of liquid H-PRF after natural polymerization. The total number, concentration, and distribution of cells within each liquid H-PRF was evaluated by complete blood count (CBC) analysis and hematoxylin–eosin staining.
Results
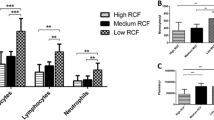
As RCF values increased, the volume and weight of liquid H-PRF both increased accordingly. SEM images revealed that as the centrifugal force increased, the fibrin bundles became thinner with a denser fibrin network, and rheologic tests revealed improved mechanical properties. CBC analysis demonstrated that 500 g group had the highest number of leukocytes and neutrophils, whereas 100 g group yielded the highest concentration of leukocytes and platelets. Furthermore, histological analysis suggests that cells obtained by 500 g for 8 min were most evenly distributed in liquid H-PRF.
Conclusions
In summary, the present study provided insights into the contents of liquid H-PRF prepared at different centrifugation forces, enabling clinicians to choose proper centrifugation forces based on their needs.
Clinical relevance
The present findings provide theoretical basis for clinical choice of liquid H-PRF protocol from mechanical, cell contents, and histological aspects.







Similar content being viewed by others
References
Raghavendra S, Wood MC, Taylor TD (2005) Early wound healing around endosseous implants: a review of the literature. Int J Oral Maxillofac Implants 20:425–431
Ozsagir ZB, Saglam E, Sen Yilmaz B, Choukroun J, Tunali M (2020) Injectable platelet-rich fibrin and microneedling for gingival augmentation in thin periodontal phenotype: a randomized controlled clinical trial. J Clin Periodontol 47:489–499. https://doi.org/10.1111/jcpe.13247
Zeitounlouian TS, Zeno KG, Brad BA, Haddad RA (2021) Effect of injectable platelet-rich fibrin (i-PRF) in accelerating orthodontic tooth movement: a randomized split-mouth-controlled trial. J Orofac Orthop 82:268–277. https://doi.org/10.1007/s00056-020-00275-x
Fabi S, Sundaram H (2014) The potential of topical and injectable growth factors and cytokines for skin rejuvenation. Facial Plast Surg 30:157–171. https://doi.org/10.1055/s-0034-1372423
Schär MO, Diaz-Romero J, Kohl S, Zumstein MA (2015) Nesic DJCO and Research® R Platelet-rich concentrates differentially release growth factors and induce cell migration in vitro. Clin Orthop Relat Res 473:1635–1643
Whitman DH, Berry RL, Green DM (1997) Platelet gel: an autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. J Oral Maxillofac Surg 55:1294–1299. https://doi.org/10.1016/s0278-2391(97)90187-7
Marx RE, Carlson ER, Eichstaedt RM, Schimmele SR, Strauss JE, Georgeff KR (1998) Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 85:638–646. https://doi.org/10.1016/s1079-2104(98)90029-4
Jia J, Wang SZ, Ma LY, Yu JB, Guo YD, Wang C (2018) The differential effects of leukocyte-containing and pure platelet-rich plasma on nucleus pulposus-derived mesenchymal stem cells: implications for the clinical treatment of intervertebral disc degeneration. Stem Cells Int 2018:7162084. https://doi.org/10.1155/2018/7162084
Yin W, Xu H, Sheng J, Zhu Z, Jin D, Hsu P, Xie X, Zhang CJE, Medicine T (2017) Optimization of pure platelet-rich plasma preparation: a comparative study of pure platelet-rich plasma obtained using different centrifugal conditions in a single-donor model. Exp Ther Med 14:2060–2070
Kobayashi E, Fluckiger L, Fujioka-Kobayashi M, Sawada K, Sculean A, Schaller B, Miron RJ (2016) Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin Oral Investig 20:2353–2360. https://doi.org/10.1007/s00784-016-1719-1
Mourão CFdAB, Valiense H, Melo ER, Mourão NBMF, Maia MD-CJRdCBdC (2015) Obtention of injectable platelets rich-fibrin (i-PRF) and its polymerization with bone graft. Rev Col Bras Cir 42:421–423
Miron RJ, Fujioka-Kobayashi M, Hernandez M, Kandalam U, Zhang Y, Ghanaati S, Choukroun J (2017) Injectable platelet rich fibrin (i-PRF): opportunities in regenerative dentistry? Clin Oral Investig 21:2619–2627. https://doi.org/10.1007/s00784-017-2063-9
Amaral Valladao CA, Jr., Freitas Monteiro M and Joly JC, (2020) Guided bone regeneration in staged vertical and horizontal bone augmentation using platelet-rich fibrin associated with bone grafts: a retrospective clinical study. Int J Implant Dent 6:72. https://doi.org/10.1186/s40729-020-00266-y
Saglam E, Ozsagir ZB, Unver T, Alinca SB, Toprak A, Tunali M (2021) Efficacy of injectable platelet-rich fibrin in the erosive oral lichen planus: a split-mouth, randomized, controlled clinical trial. J Appl Oral Sci 29:e20210180. https://doi.org/10.1590/1678-7757-2021-0180
Shashank B, Bhushan M (2021) Injectable platelet-rich fibrin (PRF): the newest biomaterial and its use in various dermatological conditions in our practice: a case series. J Cosmet Dermatol 20:1421–1426. https://doi.org/10.1111/jocd.13742
Hamid O, Quinlan DJ, Seemann R and Hassan H (2021) Injectable platelet-rich fibrin for perioral rejuvenation as assessed by 3D lip volume imaging. J Cosmet Dermatol 20:3270–3277. https://doi.org/10.1111/jocd.14394
Miron RJ, Chai J, Zheng S, Feng M, Sculean A and Zhang Y (2019) A novel method for evaluating and quantifying cell types in platelet rich fibrin and an introduction to horizontal centrifugation. J Biomed Mater Res A 107:2257–2271. https://doi.org/10.1002/jbm.a.36734
Feng M, Wang Y, Wei Y, Zhang X, Xiao L, Gong Z, Fujioka-Kobayashi M, Sculean A, Miron RJ and Froum S and Zhang Y (2021) Preparation, characterization and biological properties of a novel bone block composed of platelet rich fibrin and a deproteinized bovine bone mineral. Fundam Res 2:321–328. https://doi.org/10.1016/j.fmre.2021.08.003
Feng M, Wang Y, Zhang P, Zhao Q, Yu S, Shen K, Miron RJ, Zhang Y (2020) Antibacterial effects of platelet-rich fibrin produced by horizontal centrifugation. Int J Oral Sci 12:32. https://doi.org/10.1038/s41368-020-00099-w
Nickerson CS, Park J, Kornfield JA, Karageozian H (2008) Rheological properties of the vitreous and the role of hyaluronic acid. J Biomech 41:1840–1846. https://doi.org/10.1016/j.jbiomech.2008.04.015
Fagien S, Bertucci V, von Grote E, Mashburn JH (2019) Rheologic and physicochemical properties used to differentiate injectable hyaluronic acid filler products. Plast Reconstr Surg 143:707e–720e. https://doi.org/10.1097/Prs.0000000000005429
Salti G, Fundarò SP (2020) Evaluation of the rheologic and physicochemical properties of a novel hyaluronic acid filler range with excellent three-dimensional reticulation (XTR™) Technology. Polymers 12:1644. https://doi.org/10.3390/polym12081644
Sun C, Zeng X, Zheng S, Wang Y, Li Z, Zhang H, Nie L, Zhang Y, Zhao Y, Yang X (2022) Bio-adhesive catechol-modified chitosan wound healing hydrogel dressings through glow discharge plasma technique. Chem Eng J 427:130843. https://doi.org/10.1016/j.cej.2021.130843
Miron RJ, Chai J, Zhang P, Li Y, Wang Y, Mourao C, Sculean A, Fujioka Kobayashi M, Zhang Y (2020) A novel method for harvesting concentrated platelet-rich fibrin (C-PRF) with a 10-fold increase in platelet and leukocyte yields. Clin Oral Investig 24:2819–2828. https://doi.org/10.1007/s00784-019-03147-w
Miron RJ, Chai J, Fujioka-Kobayashi M, Sculean A, Zhang Y (2020) Evaluation of 24 protocols for the production of platelet-rich fibrin. BMC Oral Health 20:310. https://doi.org/10.1186/s12903-020-01299-w
Miron RJ, Chai J, Zheng S, Feng M, Sculean A, Zhang Y (2019) A novel method for evaluating and quantifying cell types in platelet rich fibrin and an introduction to horizontal centrifugation. J Biomed Mater Res A 107:2257–2271. https://doi.org/10.1002/jbm.a.36734
Arasteh S, Khanjani S, Golshahi H, Mobini S, Jahed MT, Heidari-Vala H, Edalatkhah H, Kazemnejad S (2020) Efficient wound healing using a synthetic nanofibrous bilayer skin substitute in murine model. J Surg Res 245:31–44. https://doi.org/10.1016/j.jss.2019.07.017
Fujioka-Kobayashi M, Kono M, Katagiri H, Schaller B, Zhang Y, Sculean A, Miron RJ (2021) Histological comparison of Platelet rich fibrin clots prepared by fixed-angle versus horizontal centrifugation. Platelets 32:413–419. https://doi.org/10.1080/09537104.2020.1754382
Zhang J, Qi X, Luo X, Li D, Wang H, Li T (2017) Clinical and immunohistochemical performance of lyophilized platelet-rich fibrin (Ly-PRF) on tissue regeneration. Clin Implant Dent Relat Res 19:466–477. https://doi.org/10.1111/cid.12473
Liu Z, Jin H, Xie Q, Jiang Z, Guo S, Li Y, Zhang B (2019) Controlled release strategies for the combination of fresh and lyophilized platelet-rich fibrin on bone tissue regeneration. Biomed Res Int 2019:4923767. https://doi.org/10.1155/2019/4923767
Agrawal DR, Jaiswal PG (2020) Injectable platelet rich fibrin (i-PRF): a gem in dentistry. Int J Cur Res Rev 12:25. http://doi.org/10.31782/IJCRR.2020.122116
Nacopoulos C, Vesala AM (2020) Lower facial regeneration with a combination of platelet-rich fibrin liquid matrices based on the low speed centrifugation concept-Cleopatra technique. J Cosmet Dermatol 19:185–189. https://doi.org/10.1111/jocd.13196
Martínez-Zapata MJ, Martí-Carvajal A, Solà I, Bolibar I, Angel Exposito J, Rodriguez L, García J (2009) Efficacy and safety of the use of autologous plasma rich in platelets for tissue regeneration: a systematic review. Transfusion 49:44–56. https://doi.org/10.1111/j.1537-2995.2008.01945.x
Li W, Sigley J, Pieters M, Helms CC, Nagaswami C, Weisel JW, Guthold M (2016) Fibrin fiber stiffness is strongly affected by fiber diameter, but not by fibrinogen glycation. Biophys J 110:1400–1410. https://doi.org/10.1016/j.bpj.2016.02.021
Fagien S, Bertucci V, von Grote E, Mashburn JH (2019) Rheologic and physicochemical properties used to differentiate injectable hyaluronic acid filler products. Plast Reconstr Surg 143:707e–720e. https://doi.org/10.1097/PRS.0000000000005429
Hee CK, Shumate GT, Narurkar V, Bernardin A, Messina DJ (2015) Rheological properties and in vivo performance characteristics of soft tissue fillers. Dermatol Surg 41(Suppl 1):S373–S381. https://doi.org/10.1097/DSS.0000000000000536
Lee W, Yoon J-H, Koh I-S, Oh W, Kim K-W, Yang E-J (2018) Clinical application of a new hyaluronic acid filler based on its rheological properties and the anatomical site of injection. Biomed Dermatol 2:1–5. https://doi.org/10.1186/s41702-018-0032-9
Funding
The study was supported by the National Natural Science Foundation of China (82101019).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The study was conducted under the supervision of Yulan Wang and Yufeng Zhang. Material preparation, data collection, and analysis were performed by Mengge Feng, Yan Wei, Hongjiang Wei, and Yunxiao Wang. The first draft of the manuscript was written by Mengge Feng, revised by Yulan Wang and Richard J Miron. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
The research involves collecting blood from human participants. All protocols used were approved by the Ethics Committee of the School and Hospital of Stomatology, Wuhan University (B52/2020) in this study.
Consent to participate
Written informed consents were obtained from volunteers.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, M., Wei, Y., Wei, H. et al. Effect of relative centrifugal force on the biological properties of liquid platelet-rich fibrin produced via horizontal centrifugation. Clin Oral Invest 27, 399–409 (2023). https://doi.org/10.1007/s00784-022-04745-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04745-x