Abstract
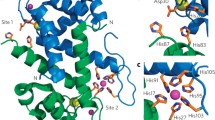
Human calprotectin (CP) is an innate immune protein that participates in the metal-withholding response to infection by sequestering essential metal nutrients from invading microbial pathogens. CP is comprised of S100A8 (α subunit, 10.8 kDa) and S100A9 (β subunit, 13.2 kDa). Two transition-metal binding sites of CP form at the S100A8/S100A9 dimer interface. Site 1 is a His3Asp motif comprised of His83 and His87 from the S100A8 subunit and His20 and Asp30 from the S100A9 subunit. Site 2 is an unusual hexahistidine motif composed of S100A8 residues His17 and His27 and S100A9 residues His91, His95, His103, and His105. In the present study, the His3Asp and His6 sites of CP were further characterized by utilizing Co2+ as a spectroscopic probe. Magnetic circular dichroism spectroscopy was employed in conjunction with electron paramagnetic resonance spectroscopy and density functional theory computations to characterize the Co2+-bound S100A8(C42S)/S100A9(C3S) CP-Ser variant and six site variants that allowed the His3Asp and His6 sites to be further probed. Our results provide new insight into the metal-binding sites of CP-Ser and the effect of amino acid substitutions on the structure of site 2.
Graphical abstract












Similar content being viewed by others
References
Hood MI, Skaar EP (2012) Nat Rev Microbiol 10:525–537
Palmer LD, Skaar EP (2016) Transition metals and virulence in bacteria. Ann Rev Genet 50:67–91. https://doi.org/10.1146/annurev-genet-120215-035146
Waldron KJ, Rutherford JC, Ford D, Robinson NJ (2009) Nature 460:823–830
Murdoch CC, Skaar EP (2022) Nat Rev Microbiol 20:657–670
Weinberg ED (1975) Jama-J Am Med Assoc 231:39–41
Zygiel EM, Nolan EM (2018) Transition metal sequestration by the host-defense protein calprotectin. Ann Rev Biochem 87:621–643. https://doi.org/10.1146/annurev-biochem-062917-012312
Hunter MJ, Chazin WJ (1998) J Biol Chem 273:12427–12435
Vogl T, Roth J, Sorg C, Hillenkamp F, Strupat K (1999) J Am Soc Mass Spectrom 10:1124–1130
Leukert N, Vogl T, Strupat K, Reichelt R, Sorg C, Roth J (2006) J Mol Biol 359:961–972
Strupat K, Rogniaux H, Van Dorsselaer A, Roth J, Vogl T (2000) J Am Soc Mass Spectrom 11:780–788
Damo SM, Kehl-Fie TE, Sugitani N, Holt ME, Rathi S, Murphy WJ, Zhang YF, Betz C, Hench L, Fritz G, Skaar EP, Chazin WJ (2013) Proc Natl Acad Sci USA 110:3841–3846
Gagnon DM, Brophy MB, Bowman SEJ, Stich TA, Drennan CL, Britt DR, Nolan EM (2015) JACS 137:3004–3016
Korndorfer IP, Brueckner F, Skerra A (2007) J Mol Biol 370:887–898
Nakashige TG, Zygiel EM, Drennan CL, Nolan EM (2017) J Am Chem Soc 139:8828–8836
Nakashige TG, Stephan JR, Cunden LS, Brophy MB, Wommack AJ, Keegan BC, Shearer JM, Nolan EM (2016) J Am Chem Soc 138:12243–12251
Nakashige TG, Zhang B, Krebs C, Nolan EM (2015) Nat Chem Biol 11:765
Adhikari J, Stephan JR, Rempel DL, Nolan EM, Gross ML (2020) J Am Chem Soc 142:13372–13383
Silvers R, Stephan JR, Griffin RG, Nolan EM (2021) J Am Chem Soc 143:18073–18090
Brophy MB, Hayden JA, Nolan EM (2012) J Am Chem Soc 134:18089–18100
Hayden JA, Brophy MB, Cunden LS, Nolan EM (2013) J Am Chem Soc 135:775–787
Stephan JR, Nolan EM (2016) Chem Sci 7:1962–1975
Brophy M, Nakashige T, Gaillard A, Nolan E (2013) J Am Chem Soc 135:17804–17817
Nakashige TG, Nolan EM (2017) Metallomics 9:1086–1095
Nakashige TG, Bowman SEJ, Zygiel EM, Drennan CL, Nolan EM (2018) Biophysical examination of the calcium-modulated nickel-binding properties of human calprotectin reveals conformational change in the EF-hand domains and His3Asp site. Biochemistry 57(28):4155–4164
Moroz OV, Blagova EV, Wilkinson AJ, Wilson KS, Bronstein IB (2009) J Mol Biol 391:536–551
Brodersen DE, Nyborg J, Kjeldgaard M (1999) Biochemistry 38:1695–1704
Moroz OV, Antson AA, Grist SJ, Maitland NJ, Dodson GG, Wilson KS, Lukanidin E, Bronstein IB (2003) Acta Crystallogr Sect D Biol Crystallogr 59:859–867
Baker TM, Nakashige TG, Nolan EM, Neidig ML (2017) Chem Sci 8:1369–1377
Besold AN, Gilston BA, Radin JN, Ramsoomair C, Culbertson EM, Li CX, Cormack BP, Chazin WJ, Kehl-Fie TE, Culotta VC (2018) Infect Immunity 86:2
Wang JF, Lonergan ZR, Gonzalez-Gutierrez G, Nairn BL, Maxwell CN, Zhang YX, Andreini C, Karty JA, Chazin WJ, Trinidad JC, Skaar EP, Giedroc DP (2019) Cell Chem Biol 26:745
Zygiel EM, Nelson CE, Brewer LK, Oglesby-Sherrouse AG, Nolan EM (2019) J Biol Chem 294:3549–3562
Maret W, Vallee BL (1993) Metallobiochem Part C 226:52–71
Vallee BL (1971). In: Mertz W, Cornatzer WE (eds) Newer trace elements in nutrition. Dekker, New York, p 33
Vallee BL, Rupley JA, Coombs TL, Neurath H (1958) J Am Chem Soc 80:4750
Vallee BL (1973) Adv Exp Med Biol 40:1–12
Harding MM, Nowicki MW, Walkinshaw MD (2010) Crystallogr Rev 16:247–302
Johnson MK (2000). In: Que L (ed) Physical methods in bioinorganic chemistry. University Science Books, Sausalito, pp 233–285
Solomon EI, Lever ABP (1999) Inorganic electronic structure and spectroscopy. Wiley, New York
Kaden TA, Holmqui B, Vallee BL (1974) Inorg Chem 13:2585–2590
Holmquist B, Kaden TA, Vallee BL (1975) Biochemistry 14:1454–1461
Tomkowicz Z, Ostrovsky S, Foro S, Calvo-Perez V, Haase W (2012) Inorg Chem 51:6046–6055
Harding MJ, Briat B (1973) Mol Phys 25:745–776
Larrabee JA, Schenk G, Mitic N, Riley MJ (2015) Eur Biophys J Biophys Lett 44:393–415
Neese F (2012) The ORCA program system. Wiley Interdiscip Rev Comput Mol Sci 2:73–78. https://doi.org/10.1002/wcms.81
Schafer A, Huber C, Ahlrichs R (1994) J Chem Phys 100:5829–5835
Schafer A, Horn H, Ahlrichs R (1992) J Chem Phys 97:2571–2577
Lee CT, Yang WT, Parr RG (1988) Phys Rev B 37:785–789
Kaden TA, Vallee BL, Holmquis B (1972) Biochem Biophys Res Commun 46:1654
Solomon EI, Neidig ML, Schenk G (2004) Magnetic circular dichroism of paramagnetic species. In: McCleverty JA, Meyer TJ (ed) Comprehensive coordination chemistry II: from biology to nanotechnology. Elsevier/Pergamon, Oxford, UK, pp 339–349. https://doi.org/10.1016/B0-08-043748-6/01216-0
Neese F (2006) J Biol Inorg Chem 11:702–711
Neese F (2009) Coord Chem Rev 253:526–563
Krzystek J, Zvyagin SA, Ozarowski A, Fiedler AT, Brunold TC, Telser J (2004) J Am Chem Soc 126:2148–2155
Larrabee JA, Alessi CM, Asiedu ET, Cook JO, Hoerning KR, Klingler LJ, Okin GS, Santee SG, Volkert TL (1997) J Am Chem Soc 119:4182–4196
Acknowledgements
This work was supported by the National Science Foundation Grant no. CHE-1352132 (CAREER to EMN), the National Science Foundation Graduate Research Fellowship Program under Grant no. DGE-1256259 (fellowship to MMK), and the National Science Foundation grant CHE-0840494 (computational resources).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Killian, M.M., Brophy, M.B., Nolan, E.M. et al. Spectroscopic and computational investigations of Cobalt(II) binding to the innate immune protein human calprotectin. J Biol Inorg Chem 29, 127–137 (2024). https://doi.org/10.1007/s00775-023-02034-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-023-02034-w