Abstract
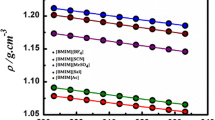
In comparison with the conventional ionic liquids, water-miscible amino acid ionic liquids (AAILs) are considered as more biodegradable and biocompatible, less toxic, and as able to enhance the biomaterials stability. An application of some long-chain ionic liquids in catalysis, extraction, etc. requests the detailed analysis of ionic and water transport properties of their diluted aqueous solutions close to the area of its critical micelle concentration (cmc). In this work, the molecular transport properties of two 1-methyl-3-octylimidazolium-based AAILs, [C8mim][Val], and [C8mim][Leu] (with anions of l-Leucine or l-Valine), in the aqueous solutions were studied by measuring the self-diffusion coefficients and the solution’ viscosities in the temperature ranges 273–343 K at the AAIL’s concentrations below and above its cmc. The data on self-diffusion coefficients of water molecules and cations/anions of AAILs are discussed in terms of activation energies and of hydration effects. Above the cmc, the [C8mim][Val] molecules demonstrate the strengthening effect on the solvent structure, while the molecules of [C8mim][Leu] have structure-destructive effect. The results obtained for the relative dynamic viscosities show a decrease of micellar size with increasing temperature. In addition, it was found that the degrees of counterion binding for both AAILs are higher than for 1-methyl-3-octylimidazolium halides.




Similar content being viewed by others
References
F. Tang, Q. Zhang, D. Ren, Z. Nie, Q. Liu, S. Yao, J. Chromatogr. A 1217, 4669–4674 (2010). https://doi.org/10.1016/j.chroma.2010.05.013
K. Fukumoto, M. Yoshizawa, H. Ohno, J. Am. Chem. Soc. 127, 2398–2399 (2005). https://doi.org/10.1021/ja043451i
R.L. Gardas, R. Ge, P. Goodrich, C. Hardacre, A. Hussain, D.W. Rooney, J. Chem. Eng. Data 55, 1505–1515 (2010). https://doi.org/10.1021/je900660x
E.V. Alopina, E.A. Safonova, I.B. Pukinsky, N.A. Smirnova, J. Chem. Eng. Data 61, 2013–2019 (2016). https://doi.org/10.1021/acs.jced.5b00948
Z. Yu, H. Gao, H. Wang, L. Chen, J. Chem. Eng. Data 56, 4295–4300 (2011). https://doi.org/10.1021/je200030k
Y. Wei, Y. Jin, Z.-J. Wu, Y. Yang, Q.-G. Zhang, Z.-H. Kang, J. Chem. Eng. Data 58, 349–356 (2013). https://doi.org/10.1021/je300932n
H. Gao, Z. Yu, H. Wang, J. Chem. Thermodyn. 42, 640–645 (2010). https://doi.org/10.1016/j.jct.2009.12.003
Y. Wei, Q.-G. Zhang, Y. Liu, X. Li, S. Lian, Z. Kang, J. Chem. Eng. Data 55, 2616–2619 (2010). https://doi.org/10.1021/je900865y
Q.-G. Zhang, K.-D. Cai, Z.-X. Jin, S.-L. Wang, S.-S. Sun, J. Chem. Eng. Data 55, 4044–4047 (2010). https://doi.org/10.1021/je100137g
O. Ben Ghanem, M.I.A. Mutalib, J.-M. Lévêque, G. Gonfa, C.F. Kait, M. El-Harbawi, J. Chem. Eng. Data 60, 1756–1763 (2015). https://doi.org/10.1021/je501162f
D. Fang, Q. Yan, D. Li, L.-X. Xia, S.-L. Zang, J. Chem. Thermodyn. 79, 12–18 (2014). https://doi.org/10.1016/j.jct.2014.06.022
Y. Zhang, X.-Y. Chen, H.-J. Wang, K.-S. Diao, J.-M. Chen, J. Mol. Struct. Theochem. 952, 16–24 (2010). https://doi.org/10.1016/j.theochem.2010.03.033
S.S. Rao, S.P. Gejji, Comput. Theor. Chem. 2015, 24–38 (1057). https://doi.org/10.1016/j.comptc.2015.01.012
T. Fan, C. Chen, T. Fan, F. Liu, Q. Peng, J. Hazard. Mater. 297, 340–346 (2015). https://doi.org/10.1016/j.jhazmat.2015.05.034
K. Damodaran, Annu. Rep. NMR Spectrosc. 88, 215–244 (2016). https://doi.org/10.1016/BS.ARNMR.2015.11.002
R. Giernoth, in NMR Spectroscopy in Ionic Liquids (Springer, Berlin, 2008), pp. 263–283. https://doi.org/10.1007/128_2008_37
A. Noda, K. Hayamizu, M. Watanabe, J. Phys. Chem. B. 105, 4603–4610 (2001). https://doi.org/10.1021/jp004132q
H. Tokuda, K. Hayamizu, K. Ishii, M.A.B.H. Susan, M. Watanabe, J. Phys. Chem. B. 108, 16593–16600 (2004). https://doi.org/10.1021/jp047480r
K. Hayamizu, S. Tsuzuki, S. Seki, Y. Umebayashi, J. Phys. Chem. B. 116, 11284–11291 (2012)
A. Menjoge, J. Dixon, J.F. Brennecke, E.J. Maginn, S. Vasenkov, J. Phys. Chem. B. 113, 6353–6359 (2009). https://doi.org/10.1021/jp900902n
M. Nakakoshi, S. Ishihara, H. Utsumi, H. Seki, Y. Koga, K. Nishikawa, Chem. Phys. Lett. 427, 87–90 (2006). https://doi.org/10.1016/J.CPLETT.2006.06.052
O.V. Yarmolenko, A.V. Yudina, A.A. Ignatova, N.I. Shuvalova, V.M. Martynenko, L.M. Bogdanova, A.V. Chernyak, V.A. Zabrodin, V.I. Volkov, Russ. Chem. Bull. 64, 2505–2511 (2015). https://doi.org/10.1007/s11172-015-1184-z
G.Z. Tulibaeva, A.V. Chernyak, A.F. Shestakov, V.I. Volkov, O.V. Yarmolenko, Russ. Chem. Bull. 65, 1727–1733 (2016). https://doi.org/10.1007/s11172-016-1502-0
A.V. Chernyak, A.V. Yudina, O.V. Yarmolenko, V.I. Volkov, Russ. J. Electrochem. 51, 478–482 (2015). https://doi.org/10.1134/S1023193515050043
M. Figueira-González, V. Francisco, L. García-Río, E.F. Marques, M. Parajó, P. Rodríguez-Dafonte, J. Phys. Chem. B. 117, 2926–2937 (2013). https://doi.org/10.1021/jp3117962
V. Klimavicius, V. Bacevicius, Z. Gdaniec, V. Balevicius, J. Mol. Liq. 210, 223–226 (2015). https://doi.org/10.1016/J.MOLLIQ.2015.05.034
J.E. Tanner, J. Chem. Phys. 52, 2523–2526 (1970). https://doi.org/10.1063/1.1673336
A. Jerschow, N. Müller, J. Magn. Reson. 125, 372–375 (1997). https://doi.org/10.1006/jmre.1997.1123
W.S. Price, NMR Studies of Translational Motion (Cambridge University Press, Cambridge, 2009)
Y. Yasaka, C. Wakai, N. Matubayasi, M. Nakahara, J. Phys. Chem. A 111, 541–543 (2007). https://doi.org/10.1021/jp0673720
V. Klimavicius, Z. Gdaniec, J. Kausteklis, V. Aleksa, K. Aidas, V. Balevicius, J. Phys. Chem. B. 117, 10211–10220 (2013). https://doi.org/10.1021/jp4021245
S. Ohta, A. Shimizu, Y. Imai, H. Abe, N. Hatano, Y. Yoshimura, Open J. Phys. Chem. 1, 70–76 (2011). https://doi.org/10.4236/ojpc.2011.13010
K. Holmberg, B. Jönsson, B. Kronberg, B. Lindman, Surfactants and Polymers in Aqueous Solution (Wiley, Chichester, 2002). https://doi.org/10.1002/0470856424
I. Goodchild, L. Collier, S.L. Millar, I. Prokeš, J.C.D. Lord, C.P. Butts, J. Bowers, J.R.P. Webster, R.K. Heenan, J. Colloid Interface Sci. 307, 455–468 (2007). https://doi.org/10.1016/j.jcis.2006.11.034
V.V. Mank, N.I. Lebovka, NMR Spectroscopy of water in Heterogeneous Systems (Naukova Dumka, Kiev, 1988)
J.C. Hindman, J. Chem. Phys. 60, 4488–4496 (1974). https://doi.org/10.1063/1.1680928
D.H. Dagade, K.R. Madkar, S.P. Shinde, S.S. Barge, J. Phys. Chem. B. 117, 1031–1043 (2013). https://doi.org/10.1021/jp310924m
B. Palecz, J. Am. Chem. Soc. 124, 6003–6008 (2002). https://doi.org/10.1021/ja011937i
A. Bagno, G. Lovato, G. Scorrano, J.W. Wijnen, J. Phys. Chem. 97, 4601–4607 (1993). https://doi.org/10.1021/j100120a008
Y. Maeda, N. Tsukida, H. Kitano, T. Terada, J. Yamanaka, J. Phys. Chem. 97, 13903–13906 (1993). https://doi.org/10.1021/j100153a073
M. Ide, Y. Maeda, H. Kitano, J. Phys. Chem. B. 101, 7022–7026 (1997). https://doi.org/10.1021/jp971334m
D. Hecht, L. Tadesse, L. Walters, J. Am. Chem. Soc. 115, 3336–3337 (1993). https://doi.org/10.1021/ja00061a045
A.V. Kustov, N.L. Smirnova, R. Neueder, W. Kunz, J. Phys. Chem. B. 116, 2325–2329 (2012). https://doi.org/10.1021/jp2121559
A.A. Nikonorova, E.V. Alopina, Y.G. Dobryakov, N.A. Smirnova Khimicheskie Nauki. “Chronos”, pp. 86–90 (2017) (in Russian)
H. Hoffmann, G. Platz, W. Ulbricht, J. Phys. Chem. 85, 1418–1428 (1981). https://doi.org/10.1021/j150610a029
N.A. Smirnova, A.A. Vanin, E.A. Safonova, I.B. Pukinsky, Y.A. Anufrikov, A.L. Makarov, J. Colloid Interface Sci. 336, 793–802 (2009). https://doi.org/10.1016/j.jcis.2009.04.004
J. Łuczak, C. Jungnickel, M. Joskowska, J. Thöming, J. Hupka, J. Colloid Interface Sci. 336, 111–116 (2009). https://doi.org/10.1016/J.JCIS.2009.03.017
J. Bowers, C.P. Butts, P.J. Martin, M.C. Vergara-Gutierrez, R.K. Heenan, Langmuir 20, 2191–2198 (2004). https://doi.org/10.1021/la035940m
U. Preiss, C. Jungnickel, J. Thöming, I. Krossing, J. Łuczak, M. Diedenhofen, A. Klamt, Chem. A Eur. J. 15, 8880–8885 (2009). https://doi.org/10.1002/chem.200900024
J. Wang, H. Wang, in Aggregation in Systems of Ionic Liquids (Springer, Berlin, 2014), pp. 39–77. https://doi.org/10.1007/978-3-642-38619-0_2
N.M. Vaghela, N.V. Sastry, V.K. Aswal, Colloid Polym. Sci. 289, 309–322 (2011). https://doi.org/10.1007/s00396-010-2332-5
Acknowledgements
We thank Dr. E. V. Alopina and Dr. Yu. G. Dobryakov from St. Petersburg State University for AAILs provided. NMR measurements were performed at the Center for Magnetic Resonance, SPbSU. The reported study was funded by the Russian Foundation for Basic Research according to the Research Project No. 16-03-00723a.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chernyshev, Y.S., Koneva, A.S. & Safonova, E.A. NMR Self-diffusion Study of Amino Acid Ionic Liquids Based on 1-Methyl-3-Octylimidazolium in Water. Appl Magn Reson 49, 607–618 (2018). https://doi.org/10.1007/s00723-018-1008-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-018-1008-z