Abstract
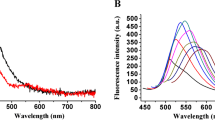
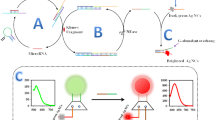
A fluorescence biosensor has been developed based on hybridisation chain reaction (HCR) amplification coupled with silver nanoclusters (AgNCs) for nucleic acid detection. The fluorescence was activated via end-to-end transfer of dark AgNCs caged within a DNA template to another DNA sequence that could enhance their red fluorescence emission at 611 nm. Such cluster-transfer approach allows us to introduce fluorogenic AgNCs as external signal transducers, thereby enabling HCR to perform in a predictable manner. The resulted HCR-AgNC biosensor was able to detect target DNA with a detection limit of 3.35 fM, and distinguish the DNA target from single-base mismatch sequences. Moreover, the bright red fluorescence emission was detectable with the naked eye, with concentration of target DNA down to 1 pM. The biosensor also performed well in human serum samples with good recovery. Overall, our cluster-transfer approach provides a good alternative to construct HCR-AgNC assay with less risk of circuit leakage and produce AgNCs in a controllable manner.
Graphical Abstract






Similar content being viewed by others
References
Obliosca JM, Liu C, Yeh HC (2013) Fluorescent silver nanoclusters as DNA probes. Nanoscale 5:8443–8461. https://doi.org/10.1039/C3NR01601C
Obliosca JM, Babin MC, Liu C, Liu YL, Chen YA, Batson RA, Ganguly M, Petty JT, Yeh HC (2014) A complementary palette of NanoCluster beacons. ACS Nano 8:10150–10160. https://doi.org/10.1021/nn505338e
Yeh HC, Sharma J, Han JJ, Martinez JS, Werner JH (2010) A DNA−silver nanocluster probe that fluoresces upon hybridization. Nano Lett 10:3106–3110. https://doi.org/10.1021/nl101773c
Yeh HC, Sharma J, Han JJ, Martinez JS, Werner JH (2011) A beacon of light: a new molecular probe for homogeneous detection of nucleic acid targets. IEEE Nanotechnol Mag 5:28–33. https://doi.org/10.1109/MNANO.2011.940951
Teng Y, Jia X, Zhang S, Zhu J, Wang E (2016) A nanocluster beacon based on the template transformation of DNA-templated silver nanoclusters. Chem Commun 52:1721–1724. https://doi.org/10.1039/C5CC09138A
Asiello PJ, Baeumner AJ (2011) Miniaturized isothermal nucleic acid amplification, a review. Lab Chip 11:1420–1430. https://doi.org/10.1039/C0LC00666A
Yan L, Zhou J, Zheng Y, Gamson AS, Roembke BT, Nakayama S, Sintim HO (2014) Isothermal amplified detection of DNA and RNA. Mol Biosyst 10:970–1003. https://doi.org/10.1039/C3MB70304E
Dirks RM, Pierce NA (2004) Triggered amplification by hybridization chain reaction. Proc Natl Acad Sci 101:15275–15278. https://doi.org/10.1073/pnas.0407024101
Bi S, Yue S, Zhang S (2017) Hybridization chain reaction: a versatile molecular tool for biosensing, bioimaging, and biomedicine. Chem Soc Rev 46:4281–4298. https://doi.org/10.1039/c7cs00055c
Green SJ, Lubrich D, Turberfield AJ (2006) DNA hairpins: fuel for autonomous DNA devices. Biophys J 91:2966–2975. https://doi.org/10.1529/biophysj.106.084681
Ang YS, Yung LYL (2016) Rational design of hybridization chain reaction monomers for robust signal amplification. Chem Commun 52:4219–4222. https://doi.org/10.1039/c5cc08907g
Li S, Li P, Ge M, Wang H, Cheng Y, Li G, Huang Q, He H, Cao C, Lin D, Yang L (2020) Elucidation of leak-resistance DNA hybridization chain reaction with universality and extensibility. Nucleic Acids Res 48:2220–2231. https://doi.org/10.1093/nar/gkaa016
Zhang S, Wang K, Li KB, Shi W, Jia WP, Chen X, Sun T, Han DM (2017) A DNA-stabilized silver nanoclusters/graphene oxide-based platform for the sensitive detection of DNA through hybridization chain reaction. Biosens Bioelectron 91:374–379. https://doi.org/10.1016/j.bios.2016.12.060
Liu L, Li Q, Tang LJ, Yu RQ, Jiang JH (2016) Silver nanocluster-lightened hybridization chain reaction. RSC Adv 6:57502–57506. https://doi.org/10.1039/c6ra09337j
Qiu X, Wang P, Cao Z (2014) Hybridization chain reaction modulated DNA-hosted silver nanoclusters for fluorescent identification of single nucleotide polymorphisms in the let-7 miRNA family. Biosens Bioelectron 60:351–357. https://doi.org/10.1016/j.bios.2014.04.040
Wong ZW, Ng JF, New SY (2021) Ratiometric detection of microRNA using hybridization chain reaction and fluorogenic silver nanoclusters. Chem Asian J 16:4081–4086. https://doi.org/10.1002/asia.202101145
Clarivate, ISI web of science. (2021). www.webofscience.com (accessed 01.07.22)
Shang L, Dörlich RM, Trouillet V, Bruns M, Nienhaus GU (2012) Ultrasmall fluorescent silver nanoclusters: protein adsorption and its effects on cellular responses. Nano Res 5:531–542. https://doi.org/10.1007/S12274-012-0238-X
Penna E, Orso F, Taverna D (2015) miR-214 as a key hub that controls cancer networks: small player, multiple functions. J Invest Dermatol 135:960–969. https://doi.org/10.1038/jid.2014.479
Sharma T, Hamilton R, Mandal CC (2015) miR-214: a potential biomarker and therapeutic for different cancers. Future Oncol 11:349–363. https://doi.org/10.2217/fon.14.193
Wang Q, Liu Y, Wu Y, Wen J, Man C (2021) Immune function of miR-214 and its application prospects as molecular marker. PeerJ 9:e10924. https://doi.org/10.7717/peerj.10924
Yu J, Choi S, Dickson RM (2009) Shuttle-based fluorogenic silver-cluster biolabels. Angew Chemie Int Ed 48:318–320. https://doi.org/10.1002/ANIE.200804137
Lin X, Xiao F, Li X, Li F, Liu C, Xiao X, Hu N, Yang S (2019) A cytosine-rich hairpin DNA loaded with silver nanoclusters as a fluorescent probe for uranium(IV) and mercury(II) ions. Microchim Acta 186:519. https://doi.org/10.1007/S00604-019-3625-0
Filipczuk P, Świtalska A, Kosman J, Nowaczyk G, Dembska A (2021) Fluorescent AgNCs formed on bifunctional DNA template for potassium ion detection. Chemosensors 9:349. https://doi.org/10.3390/chemosensors9120349
Volkov IL, Smirnova A, Makarova AA, Reveguk ZV, Ramazanov RR, Usachov DY, Adamchuk VK, Kononov AI (2017) DNA with ionic, atomic, and clustered silver: an XPS study. J Phys Chem B 121:2400–2406. https://doi.org/10.1021/acs.jpcb.6b11218
Huaccallo-Aguilar Y, Álvarez-Torrellas S, Larriba M, Águeda VI, Delgado JA, Ovejero G, García J (2019) Optimization parameters, kinetics, and mechanism of naproxen removal by catalytic wet peroxide oxidation with a hybrid iron-based magnetic catalyst. Catal 9:287. https://doi.org/10.3390/catal9030287
Jones B (2021) The prediction profiler at 30. Qual Eng 33:417–424. https://doi.org/10.1080/08982112.2021.1874015
Max KEA, Bertram K, Akat KM, Bogardus KA, Li J, Morozov P, Ben-Dov IZ, Li X, Weiss ZR, Azizian A, Sopeyin A, Diacovo TG, Adamidi C, Williams Z, Tuschl T (2018) Human plasma and serum extracellular small RNA reference profiles and their clinical utility. Proc Natl Acad Sci USA 115:E5334–E5343. https://doi.org/10.1073/PNAS.1714397115
Meng Y, Chen F, Jiang M, Guo Q, Wang Y, Wang J, Zhang DW (2021) A Homogeneous label-free electrochemical microRNA biosensor coupling with G-Triplex/methylene blue complex and λ-exonuclease-assisted recycling amplification. Front Chem 9:753253. https://doi.org/10.3389/fchem.2021.753253
Arévalo B, Serafín V, Sánchez-Paniagua M, Montero-Calle A, Barderas R, López-Ruíz B, Campuzano S, Yáñez- Sedeño P, Pingarrón JM (2020) Fast and sensitive diagnosis of autoimmune disorders through amperometric biosensing of serum anti-dsDNA autoantibodies. Biosens Bioelectron 160:112233. https://doi.org/10.1016/J.BIOS.2020.112233
Max KEA, Bertram K, Akat KM et al (2018) Human plasma and serum extracellular small RNA reference profiles and their clinical utility. Proc Natl Acad Sci USA 115:E5334–E5343. https://doi.org/10.1073/PNAS.1714397115
Geekiyanage H, Rayatpisheh S, Wohlschlegel JA et al (2020) Extracellular microRNAs in human circulation are associated with miRISC complexes that are accessible to anti-AGO2 antibody and can bind target mimic oligonucleotides. Proc Natl Acad Sci USA 117:24213–24223. https://doi.org/10.1073/PNAS.2008323117
Matsuzaki J, Ochiya T (2020) Circulating microRNAs: next-generation cancer detection. Keio J Med 69:88–96. https://doi.org/10.2302/KJM.2019-0011-OA
Song W, Zhu K, Cao Z, Lau C, Lu J (2012) Hybridization chain reaction-based aptameric system for the highly selective and sensitive detection of protein. Analyst 137:1396–1401. https://doi.org/10.1039/c2an16232f
del Río JS, Lobato IM, Mayboroda O et al (2017) Enhanced solid-phase recombinase polymerase amplification and electrochemical detection. Anal Bioanal Chem 409:3261–3269. https://doi.org/10.1007/S00216-017-0269-Y
Acknowledgements
ZWW. acknowledges the Faculty of Science and Engineering, University of Nottingham Malaysia, for sponsoring his PhD study. ZWW. also acknowledges Dr Nur Aliana Hidayah Mohamed (Universiti Teknologi MARA) for her support in providing the human serum sample used in this work.
Funding
This work was supported by the Fundamental Research Grant Scheme (FRGS) under the project code: FRGS/1/2018/STG07/UNIM/02/2.
Author information
Authors and Affiliations
Contributions
ZWW and SYN have contributed equally: conceptualisation, writing — original draft and revision. ZWW: data curation, formal analysis, investigation, methodology, project administration, software and visualization. SYN: funding acquisition, resources, supervision and validation.
Corresponding author
Ethics declarations
Ethics approval
The study involving human serum was reviewed and approved by the Science and Engineering Research Ethics Committee of the University of Nottingham Malaysia.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wong, Z.W., New, S.Y. An enzyme-free turn-on fluorescent strategy for nucleic acid detection based on hybridization chain reaction and transferable silver nanoclusters. Microchim Acta 190, 16 (2023). https://doi.org/10.1007/s00604-022-05591-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-022-05591-0