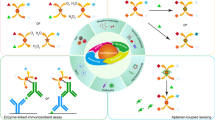
Abstract
A Glypican-3 (GPC3) electrochemical aptamer nanobiosensor based on hemin/graphene nanohybrids (HGNs) peroxidase-like catalytic silver deposition and GPC3 aptamer has been constructed for the determination of GPC3. The HGNs were prepared by an one-step reduction method. Fourier transform infrared spectroscopy (FT-IR), ultraviolet spectroscopy (UV-vis), and transmission electron microscopy (TEM) were used to study the structure and morphological characteristics of the HGNs. The GPC3 electrochemical aptamer nanobiosensor was constructed using HGNs-aptamer (HGNs-Apt) as the signal probe and GPC3 aptamer as the capture probe. With the help of the catalytic action of peroxidase-like properties of HGNs, H2O2 reduces the silver (Ag) ions in solution to metallic Ag, which deposit on the surface of the electrode. The amount of deposited Ag, which was derived from the amount of GPC3, was quantified by differential pulse voltammetry (DPV). Under optimal conditions, the current response of Ag had a good positive correlation with the GPC3 concentration in the range 10.0–100.0 μg mL−1 with a correlation coefficient of 0.9958. The detection limit was 3.16 μg mL−1 at a signal-to-noise ratio of 3, and the sensitivity was calculated to be 0.807 μA μM−1 cm−2. The method is validated by analyzing spiked human serum samples with good recovery ranging from 101 to 122%. In addition, the GPC3 electrochemical aptamer nanobiosensor has acceptable selectivity, stability, and reproducibility.

A Glypican-3 electrochemical aptamer nanobiosensor based on hemin/graphene nanohybrids (HGNs) peroxidase-like catalytic silver deposition and GPC3 aptamer has been constructed for the determination of GPC3. The electrochemical aptamer nanobiosensor exhibits high selectivity, acceptance reproducibility, and good recovery performances.





Similar content being viewed by others
References
Chen CN, Tsai YT, Lai JN (2019) 8 years post-marketing surveillance between asari radix and hepatocellular carcinoma: nationwide population-based evidence against an association. J Ethnopharmacol 243:112094. https://doi.org/10.1016/j.jep.2019.112094
Tretiakova M, Zynger DL, Luan CY, Andeen NK, Finn LS, Kocherginsky M, Teh BT, Yang XMJ (2015) Glypican 3 overexpression in primary and metastatic wilms tumors. Virchows Arch 466(1):67–76. https://doi.org/10.1007/s00428-014-1669-4
Cao WQ, Sharma M, Imam RM, Yu JZ (2019) Study on diagnostic values of astrocyte elevated gene 1 (AEG-1) and glypican 3 (GPC-3) in hepatocellular carcinoma. Am J Clin Pathol 152(5):647–655. https://doi.org/10.1093/ajcp/aqz086
Zhou FB, Shang WT, Yu XL, Tian J (2018) Glypican-3: a promising biomarker for hepatocellular carcinoma diagnosis and treatment. Med Res Rev 38(2):741–767. https://doi.org/10.1002/med.21455
Wu YL, Liu H, Ding HG (2016) GPC-3 in hepatocellular carcinoma: current perspectives. J Hepatocell Carcinoma 3:63–67. https://doi.org/10.2147/JHC.S116513
Xie CM, Tiede C, Zhang XY, Wang CR, Li ZX, Xu X, McPherson MJ, Tomlinson DC, Xu WW (2017) Development of an affimer antibody combined immunological diagnosis kit for glypican-3. Sci Rep-Uk 7:9608. https://doi.org/10.1038/s41598-017-10083-w
Chen JJ, Xie CM, Wang CR, Wan Y, Dong ZN, Li M, Xu WW (2017) Development of a time-resolved fluorescence immunoassay for the diagnosis of hepatocellular carcinoma based on the detection of glypican-3. J Fluoresc 27(4):1479–1485. https://doi.org/10.1007/s10895-017-2087-1
Yu JP, Xu XG, Ma RJ, Qin SN, Wang CR, Wang XB, Li M, Li MS, Ma Q, Xu WW (2015) Development of a clinical chemiluminescent immunoassay for serum GPC3 and simultaneous measurements alone with AFP and CK19 in diagnosis of hepatocellular carcinoma. J Clin Lab Anal 29(2):85–93. https://doi.org/10.1002/jcla.21733
Chikhaliwala P, Rai R, Chandra S (2019) Simultaneous voltammetric immunodetection of alpha-fetoprotein and glypican-3 using a glassy carbon electrode modified with magnetite-conjugated dendrimers. Microchim Acta 186(4):255. https://doi.org/10.1007/s00604-019-3354-4
Negahdary M (2020) Aptamers in nanostructure-based electrochemical biosensors for cardiac biomarkers and cancer biomarkers: a review. Biosens Bioelectron 152:112018. https://doi.org/10.1016/j.bios.2020.112018
Ladju RB, Pascut D, Massi MN, Tiribelli C, Sukowati CHC (2018) Aptamer: a potential oligonucleotide nanomedicine in the diagnosis and treatment of hepatocellular carcinoma. Oncotarget 9(2):2951–2961. https://doi.org/10.18632/oncotarget.23359
Tsao SM, Lai JC, Horng HE, Hong CY (2017) Generation of aptamers from a primer-free randomized ssDNA library using magnetic-assisted rapid aptamer selection. Sci Rep-Uk 7:45478. https://doi.org/10.1038/srep45478
Mira TA, Yoon JH, Gurudatt NG, Won MS, Shim YB (2015) Ultrasensitive cytosensing based on an aptamer modified nanobiosensor with a bioconjugate: detection of human non-small-cell lung cancer cells. Biosens Bioelectron 74:594–600. https://doi.org/10.1016/j.bios.2015.07.012
Wang T, Zhang P, Zhang KH, Luo FB, Wan QS, Han FL, Yuan HL, Huang DQ, Lv NH (2015) Diagnostic value of glypican-3 aptamers for primary hepatic carcinoma. J Gastroen Hepatol 32:190–190
Li GY, Zeng JX, Liu HL, Ding P, Liang JT, Nie XM, Zhou ZD (2019) A fluorometric aptamer nanoprobe for alpha-fetoprotein by exploiting the FRET between 5-carboxyfluorescein and palladium nanoparticles. Mikrochim Acta 186:314–322. https://doi.org/10.1007/s00604-019-3403-z
Guan JF, Huang ZN, Zou J, Jiang XY, Peng DM, Yu JG (2020) A sensitive non-enzymatic electrochemical sensor based on acicular manganese dioxide modified graphene nanosheets composite for hydrogen peroxide detection. Ecotox Environ Saf 190:110123. https://doi.org/10.1016/j.ecoenv.2019.110123
Zou J, Zhao GQ, Teng J, Liu Q, Jiang XY, Jiao FP, Yu JG (2019) Highly sensitive detection of bisphenol A in real water samples based on in-situ assembled graphene nanoplatelets and gold nanoparticles composite. Microchem J 145:693–702. https://doi.org/10.1016/j.microc.2018.11.040
Zhang FH, Yuan YW, Zheng YQ, Wang H, Liu TH, Hou SF (2017) A glassy carbon electrode modified with gold nanoparticle-encapsulated graphene oxide hollow microspheres for voltammetric sensing of nitrite. Microchim Acta 184(6):1565–1572. https://doi.org/10.1007/s00604-017-2264-6
Li RY, Liu Q, Jin Y, Li BX (2019) G-triplex/hemin DNAzyme: an ideal signal generator for isothermal exponential amplification reaction-based biosensing platform. Anal Chim Acta 1075:139–145. https://doi.org/10.1016/j.aca.2019.06.002
Wang X, Huang K, Zhang HX, Zeng LS, Zhou YK, Jing T (2019) Preparation of molecularly imprinted polymers on hemin-graphene surface for recognition of high molecular weight protein. Acs Biomater Sci Eng 105:110141. https://doi.org/10.1016/j.msec.2019.110141
Alizadeh N, Salimi A, Hallaj R (2020) Hemin/G-Quadruplex horseradish peroxidase-mimicking DNAzyme: principle and biosensing application. Adv Biochem Eng Biot 170:85–106. https://doi.org/10.1007/10_2017_37
Li S, Zhang LY, Jiang Y, Zhu SY, Lv XX, Duan ZQ, Wang H (2017) In-site encapsulating gold “nanowires” into hemin-coupled protein scaffolds through biomimetic assembly towards the nanocomposites with strong catalysis, electrocatalysis and fluorescence properties. Nanoscale 9(41):16005–16011. https://doi.org/10.1039/c7nr04945e
Yang ZT, Qian J, Yang XW, Jiang D, Du XJ, Wang K, Mao HP, Wang K (2015) A facile label-free colorimetric aptasensor for acetamiprid based on the peroxidase-like activity of hemin-functionalized reduced graphene oxide. Biosens Bioelectron 65:39–46. https://doi.org/10.1016/j.bios.2014.10.004
Khalil I, Yehye WA, Julkapli NM, Rahmati S, Ibn Sina A, Basirun WJ, Johan MR (2019) Graphene oxide and gold nanoparticle based dual platform with short DNA probe for the pcr free DNA biosensing using surface-enhanced raman scattering. Biosens Bioelectron 131:214–223. https://doi.org/10.1016/j.bios.2019.02.028
Bhatia A, Nandhakumar P, Kim G, Kim J, Lee NS, Yoon YH, Yang H (2019) Ultrasensitive detection of parathyroid hormone through fast silver deposition induced by enzymatic nitroso reduction and redox cycling. Acs Sensors 4(6):1641–1647. https://doi.org/10.1021/acssensors.9b00456
Conzuelo F, Grutzke S, Stratmann L, Pingarron JM, Schuhmann W (2016) Interrogation of immunoassay platforms by SERS and SECM after enzyme-catalyzed deposition of silver nanoparticles. Microchim Acta 183:281–287. https://doi.org/10.1007/s00604-015-1654-x
Huang Y, Tan J, Cui LJ, Zhou ZD, Zhou SF, Zhang ZH, Zheng R, Xue YW, Zhang MX, Li SS, Zhu NX, Liang JT, Li GY, Zhong LP, Zhao YX (2017) Graphene and Au NPs co-mediated enzymatic silver deposition for the ultrasensitive electrochemical detection of cholesterol. Biosens Bioelectron 102:560–567. https://doi.org/10.1016/j.bios.2017.11.037
Li R, Li S, Dong M, Zhang L, Qiao Y, Jiang Y, Qi W, Wang H (2015) A highly specific and sensitive electroanalytical strategy for microRNAs based on amplified silver deposition by the synergic TiO2 photocatalysis and guanine photoreduction using charge-neutral probes. Chem Commun (Camb) 51:16131–16134. https://doi.org/10.1039/c5cc07277h
Zhao M, Dong L, Liu Z, Yang S, Wu W, Lin J (2018) In vivo fluorescence imaging of hepatocellular carcinoma using a novel GPC3-specific aptamer probe. Quant Imag Med Surg 8(2):151–160. https://doi.org/10.21037/qims.2018.01.09
Zheng AX, Zhang XL, Gao J, Liu X, Liu JF (2016) Peroxidase-like catalytic activity of copper ions and its application for highly sensitive detection of glypican-3. Anal Chim Acta 941:87–93. https://doi.org/10.1016/j.aca.2016.08.036
Ma XY, Wang XL, Chen MF, Li FC (2017) Study on a new high affinity anti-glypicans-3 antibody in diagnosis of early hepatocellular carcinoma by differential pulse voltammetry. J Solid State Electr 21(6):31–1637. https://doi.org/10.1007/s10008-017-3535-1
Yuan GL, He JL, Li Y, Xu WL, Gao LL, Yu C (2015) A novel ultrasensitive electrochemical immunosensor based on carboxy-endcapped conductive polypyrrole for the detection of gypican-3 in human serum. Anal Mehods-Uk 7(5):1745–1750. https://doi.org/10.1039/c4ay02820a
Funding
This work was supported by the National Nature Science Foundation of China (Nos. 81760534 and 61941108), the National Science Foundation of Guangxi province of China (No. 2108GXNSFAA281231), and the Innovation Project of GUET Graduate Education (2018YJCX69).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The human serum samples used in this study were approved by the Guangxi Key Laboratory of Metabolic Diseases Research Ethics Committee in Guilin, China.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 839 kb)
Rights and permissions
About this article
Cite this article
Zhou, Z., Zhao, L., Li, W. et al. Glypican-3 electrochemical aptamer nanobiosensor based on hemin/graphene nanohybrids peroxidase-like catalytic silver deposition. Microchim Acta 187, 305 (2020). https://doi.org/10.1007/s00604-020-04284-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-04284-w