Abstract
Purpose
The transmembrane serine protease 4 (TMPRSS4) gene is upregulated in various human cancers. However, its biological functions in pancreatic ductal adenocarcinoma remain unclear. We examined the expression of TMPRSS4 in pancreatic ductal adenocarcinoma tissues and its correlation with clinicopathological parameters in patients with pancreatic ductal adenocarcinoma who underwent surgery.
Methods
The TMPRSS4 expression was immunohistochemically examined in 81 PDAC patients with pancreatic ductal adenocarcinoma. We analyzed the association between the TMPRSS4 expression and clinicopathological factors, the recurrence-free survival (RFS), and the overall survival (OS) and examined the effect of TMPRSS4 expression on cell migration and sensitivity to 5-fluorouracil.
Results
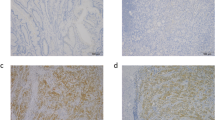
The expression rate of TMPRSS4 in the samples was 62.9% (51/81). The TMPRSS4 expression was not correlated with any clinicopathological feature. The five-year overall and recurrence-free survival rates were significantly lower in the TMPRSS4-positive group than in the TMPRSS4-negative group. On a multivariate analysis, TMPRSS4 positivity, poorly differentiated histology, and non-adjuvant chemotherapy predicted a poor OS, while TMPRSS4 positivity and poorly differentiated histology predicted a poor RFS. TMPRSS4-silenced pancreatic ductal adenocarcinoma cells showed higher sensitivity to 5- fluorouracil than did the control siRNA-transfected cells.
Conclusions
TMPRSS4 can be considered a prognostic factor and therapeutic target for pancreatic ductal adenocarcinoma.




Similar content being viewed by others
Abbreviations
- TMPRSS4:
-
Transmembrane serine protease 4
- PDAC:
-
In pancreatic ductal adenocarcinoma
- 5-FU:
-
5-Fluorouracil
- siControl:
-
Control siRNA
- FdUMP:
-
Fluorodeoxyuridine monophosphate
- TS:
-
Thymidylate synthase
- FBS:
-
Fetal bovine serum
- OS:
-
Overall survival
- RFS:
-
Recurrence-free survival
- NCCN:
-
National Comprehensive Cancer Network
- EMT:
-
Epithelial-to-mesenchymal transition
- SDS:
-
Sodium dodecyl sulfate
- PSM:
-
Propensity score matching
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Moon HJ, An JY, Heo JS, Choi SH, Joh JW, Kim YI. Predicting survival after surgical resection for pancreatic ductal adenocarcinoma. Pancreas. 2006;32:37–43.
Oettle H, Post S, Neuhaus P, Gellert K, Langrehr J, Ridwelski K, et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAM. 2007;297:267–77.
Uesaka K, Boku N, Fukutomi A, Okamura Y, Konishi M, Matsumoto I, et al. Adjuvant chemotherapy of S-1 versus gemcitabine for resected pancreatic cancer: a phase 3, open-label, randomised, non-inferiority trial (JASPAC 01). Lancet. 2016;388:248–57.
Neoptolemos JP, Palmer DH, Ghaneh P, Psarelli EE, Valle JW, Halloran CM, et al. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, phase 3 trial. Lancet. 2017;389:1011–24.
Conroy T, Hammel P, Hebbar M, Ben Abdelghani M, Wei AC, Raoul JL, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379:2395–406.
Bugge TH, Antalis TM, Wu Q. Type II transmembrane serine proteases. J Biol Chem. 2009;284:23177–81.
Wallrapp C, Hähnel S, Müller-Pillasch F, Burghardt B, Iwamura T, Ruthenbürger M, et al. A novel transmembrane serine protease (TMPRSS3) overexpressed in pancreatic cancer. Cancer Res. 2000;60:2602–6.
Huang A, Zhou H, Zhao H, Quan Y, Feng B, Zheng M. High expression level of TMPRSS4 predicts adverse outcomes of colorectal cancer patients. Med Oncol. 2013;30:712.
Liang B, Wu M, Bu Y, Zhao A, Xie F. Prognostic value of TMPRSS4 expression in patients with breast cancer. Med Oncol. 2013;30:497.
Wu XY, Zhang L, Zhang KM, Zhang MH, Ruan TY, Liu CY, et al. Clinical implication of TMPRSS4 expression in human gallbladder cancer. Tumour Biol. 2014;35:5481–6.
Wang CH, Guo ZY, Chen ZT, Zhi XT, Li DK, Dong ZR, et al. TMPRSS4 facilitates epithelial-mesenchymal transition of hepatocellular carcinoma and is a predictive marker for poor prognosis of patients after curative resection. Sci Rep. 2015;5:12366.
Chikaishi Y, Uramoto H, Koyanagi Y, Yamada S, Yano S, Tanaka F. TMPRSS4 expression as a marker of recurrence in patients with lung cancer. Anticancer Res. 2016;36:121–7.
Tazawa H, Suzuki T, Saito A, Ishikawa A, Komo T, Sada H, et al. Utility of TMPRSS4 as a prognostic biomarker and potential therapeutic target in patients with gastric cancer. J Gastrointest Surg. 2022;26:305–13.
THE HUMAN PROTEIN ATLAS. https://www.proteinatlas.org/ENSG00000137648-TMPRSS4 Accessed dd month yyyy.
Zeng P, Zhang P, Zhou LN, Tang M, Shen YX, Jin J, et al. TMPRSS4 as an emerging potential poor prognostic factor for solid tumors: a systematic review and meta-analysis. Oncotarget. 2016;7:76327–36.
Kim S, Kang HY, Nam EH, Choi MS, Zhao XF, Hong CS, et al. TMPRSS4 induces invasion and epithelial-mesenchymal transition through upregulation of integrin alpha5 and its signaling pathways. Carcinogenesis. 2010;31:597–606.
Gu J, Huang W, Zhang J, Wang X, Tao T, Yang L, et al. TMPRSS4 promotes cell proliferation and inhibits apoptosis in pancreatic ductal adenocarcinoma by activating ERK1/2 signaling pathway. Front Oncol. 2021;11: 628353.
Brierley JD, Gospodarowicz MK, Wittekind C. International Union Against Cancer (UICC): TNM Classification of Malignant Tumors. Hoboken: John wiley & sons; 2017.
National Comprehensive Cancer Network NCCN Clinical Practice Guidelines in Oncology, 2021. https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.
Network NC. NCCN clinical practice guidelines in oncology. Pancreatic adenocarcinoma. Version 1, 2016. http://www.Vccn.Org/professionals/physician_gls/pdf/pancreatic.
Rubin DB. Estimating causal effects from large data sets using propensity scores. Ann Intern Med. 1997;127:757–63.
Safley DM, Amin AP, House JA, Baklanov D, Mills R, Giersiefen H, et al. Comparison of costs between transradial and transfemoral percutaneous coronary intervention: a cohort analysis from the Premier research database. Am Heart J. 2013;165:303-9.e2.
Ye H, Li T, Wang H, Wu J, Yi C, Shi J, et al. TSPAN1, TMPRSS4, SDR16C5, and CTSE as novel panel for pancreatic cancer: a bioinformatics analysis and experiments validation. Front Immunol. 2021;12: 649551.
Bhasin MK, Ndebele K, Bucur O, Yee EU, Otu HH, Plati J, et al. Meta-analysis of transcriptome data identifies a novel 5-gene pancreatic adenocarcinoma classifier. Oncotarget. 2016;7:23263–81.
Cheng Y, Wang K, Geng L, Sun J, Xu W, Liu D, et al. Identification of candidate diagnostic and prognostic biomarkers for pancreatic carcinoma. EBioMed. 2019;40:382–93.
Cheng H, Fukushima T, Takahashi N, Tanaka H, Kataoka H. Hepatocyte growth factor activator inhibitor type 1 regulates epithelial to mesenchymal transition through membrane-bound serine proteinases. Cancer Res. 2009;69:1828–35.
Exposito F, Villalba M, Redrado M, de Aberasturi AL, Cirauqui C, Redin E, et al. Targeting of TMPRSS4 sensitizes lung cancer cells to chemotherapy by impairing the proliferation machinery. Cancer Lett. 2019;453:21–33.
Maehara Y, Kusumoto T, Kusumoto H, Anai H, Sakaguchi Y, Kido Y, et al. 5-fluorouracil and UFT-sensitive gastric carcinoma has a high level of thymidylate synthase. Cancer. 1989;63:1693–6.
Kondo N, Murakami Y, Uemura K, Sudo T, Hashimoto Y, Nakashima A, et al. Prognostic impact of dihydropyrimidine dehydrogenase expression on pancreatic adenocarcinoma patients treated with S-1-based adjuvant chemotherapy after surgical resection. J Surg Oncol. 2011;104:146–54.
Oba A, Ban D, Kirimura S, Akahoshi K, Mitsunori Y, Matsumura S, et al. Clinical application of the biomarkers for the selection of adjuvant chemotherapy in pancreatic ductal adenocarcinoma. J Hepato Pancreat Sci. 2016;23:480–8.
Kurata N, Fujita H, Ohuchida K, Mizumoto K, Mahawithitwong P, Sakai H, et al. Predicting the chemosensitivity of pancreatic cancer cells by quantifying the expression levels of genes associated with the metabolism of gemcitabine and 5-fluorouracil. Int J Oncol. 2011;39:473–82.
Kurasaka C, Ogino Y, Sato A. Molecular mechanisms and tumor biological aspects of 5-fluorouracil resistance in HCT116 human colorectal cancer cells. Int J Mol Sci. 2021;22:2916.
Kurasaka C, Nishizawa N, Ogino Y, Sato A. Trapping of 5-Fluorodeoxyuridine monophosphate by thymidylate synthase confers resistance to 5-fluorouracil. ACS Omega. 2022;7:6046–52.
Mori R, Yoshida K, Futamura M, Suetsugu T, Shizu K, Tanahashi T, et al. The inhibition of thymidine phosphorylase can reverse acquired 5FU-resistance in gastric cancer cells. Gastric Cancer. 2019;22:497–505.
Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3:330–8.
Van Triest B, Pinedo HM, Giaccone G, Peters GJ. Downstream molecular determinants of response to 5-fluorouracil and antifolate thymidylate synthase inhibitors. Ann Oncol. 2000;11:385–91.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Sho Tazuma and other co-authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tazuma, S., Sudo, T., Ishikawa, A. et al. Effects of transmembrane serine protease 4 on the survival in patients with pancreatic ductal adenocarcinoma undergoing surgery followed by adjuvant chemotherapy. Surg Today (2024). https://doi.org/10.1007/s00595-024-02824-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00595-024-02824-y