Abstract
Purposes
This study aimed to investigate the histopathological changes that occur within 2 weeks following spinal cord injury (SCI) in dogs.
Methods
Eight adult female Beagle dogs were included in this study, and SCI was induced using an epidural balloon catheter. Two dogs were killed at each of the following four time points: immediately after the procedure and 1 day, 1 week, and 2 weeks after the procedure. Neurological status was evaluated with five categories. Histopathological changes were visually observed for stained sections of formalin-fixed spinal cord to evaluate hemorrhage, spongiosis, necrosis, and gliosis morphologically.
Results
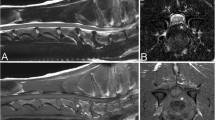
Along the 2 weeks post-injury, severe hemorrhage was observed at the primary injury site, the average diameter of which expanded quickly from 8 to 10 mm in 1 day and then decreased to 5 mm in 1 week. This indicates that the bleeding cavity expanded at the initial injury site to produce ascending and descending hemorrhage. The hemorrhage at the injury site resolved in 2 weeks. In contrast, spongiosis, parenchymal necrosis, and gliosis were first inconspicuous or mild and then became severe in 1 week or 2 weeks. Hemorrhage, hematoma, and other similar changes occurred at the regions approximately 20-mm rostral and caudal to the primary injury site. These changes were observed in both gray matter and white matter.
Conclusions
This study is the first to assess the sequential histopathological changes in the acute and intermediate phases following SCI in dogs. Our findings enhance the usefulness of the canine intervertebral disk disease model in the assessment of secondary spinal cord histopathology in human SCI.



Similar content being viewed by others
References
Shingu H, Ohama M, Ikata T, Katoh S, Akatsu T (1995) A nationwide epidemiological survey of spinal cord injuries in Japan from January 1990 to December 1992. Spinal Cord 33:183–188. https://doi.org/10.1038/sc.1995.42
Fluehmann G, Doherr MG, Jaggy A (2010) Canine neurological diseases in a referral hospital population between 1989 and 2000 in Switzerland. J Small Anim Pract 47:582–587. https://doi.org/10.1111/j.1748-5827.2006.00106.x
Brisson BA (2010) Intervertebral disc disease in dogs. Vet Clin North Am Small Anim Pract 40:829–858. https://doi.org/10.1016/j.cvsm.2010.06.001
Nakamoto Y, Nakamoto M, Ozawa T (2018) Survey of the incidence of neurological diseases in dogs at a secondary veterinary neurology facility [In Japanese]. J Jpn Vet Med Assoc 71:41–49
Besalti O, Can P, Akpinar E, Aktas Z, Elcin AE, Elcin YM (2015) Intraspinal transplantation of autologous neurogenically-induced bone marrow-derived mesenchymal stem cells in the treatment of paraplegic dogs without deep pain perception secondary to intervertebral disk disease. Turk Neurosurg 25:625–632. https://doi.org/10.5137/1019-5149.JTN.14502-15.2
Alisauskaite N, Spitzbarth I, Baumgartner W et al (2017) Chronic post-traumatic intramedullary lesions in dogs, a translational model. PLoS ONE 12:e0187746. https://doi.org/10.1371/journal.pone.0187746.eCollection2017
Henke D, Vandevelde M, Doherr MG, Stockli M, Foterre F (2013) Correlations between severity of clinical signs and histopathological changes in 60 dogs with spinal cord injury associated with acute thoracolumbar intervertebral disc disease. Vet J 198:70–75. https://doi.org/10.1016/j.tvjl.2013.04.003
Langerhuus L, Miles J (2017) Proportion recovery and times to ambulation for non-ambulatory dogs with thoracolumbar disc extrusions treated with hemilaminectomy or conservative treatment: A systematic review and meta-analysis of case-series studies. Vet J 220:7–16. https://doi.org/10.1016/j.tvjl.2016.12.008
Tamura K, Harada Y, Nagashima N (2012) Autotransplanting of bone marrow-derived mononuclear cells for complete cases of canine paraplegia and loss of pain perception secondary to intervertebral disc herniation. Exp Clin Transplant 3:263–272. https://doi.org/10.6002/ect.2011.0151
Cheriyan T, Ryan DJ, Weinreb JH et al (2014) Spinal cord injury models: a review. Spinal Cord 52:588–595. https://doi.org/10.1038/sc.2014.91
Purdy PD, Duong RT, White CL et al (2003) Percutaneous translumbar spinal cord compression injury in a dog model that uses angioplasty balloon: MR imaging and histopathologic findings. AJNR Am J Neuroradiol 24:177–184
Purdy PD, White CL 3rd, Baser DL et al (2004) Percutaneous translumbar spinal cord compression injury in dogs from an angioplasty balloon: MR and histopathologic changes with balloon sizes and compression times. AJNR Am J Neuroradiol 25:1435–1442
Fukuda S, Nakamura T, Kishigami Y et al (2005) New canine spinal cord injury model free from laminectomy. Brain Res Brain Res Protoc 14:171–180. https://doi.org/10.1016/j.brainresprot.2005.01.001
Lim JH, Jung CS, Byeon YE, Kim WH, Yoon JH, Kang KS, Kweon OK (2007) Establishment of a canine spinal cord injury model induced by epidural balloon compression. J Vet Sci 8:89–94. https://doi.org/10.4142/jvs.2007.8.1.89
Lee JH, Choi CB, Chung DJ et al (2008) Development of an improved canine model of percutaneous spinal cord compression injury by balloon catheter. J Neurosci Methods 167:310–316. https://doi.org/10.1016/j.jneumeth.2007.07.020
Nardone R, Florea C, Holler Y et al (2017) Rodent, large animal and non-human primate models of spinal cord injury. Zoology 123:101–114. https://doi.org/10.1016/j.zool.2017.06.004
Spitzbarth I, Moore SA, Stein VM et al (2020) Current insights into the pathology of canine intervertebral disc extrusion-induced spinal cord injury. Front Vet Sci 7:595796. https://doi.org/10.3389/fvets.2020.595796
Schulz KS, Walker M, Moon M, Waldron D, Slater M, McDonald DE (1998) Correlation of clinical, radiographic, and surgical localization of intervertebral disc extrusion in small-breed dogs: a prospective study of 50 cases. Vet Surg 27:105–111
Stevens JR, Casanova M, Poltorak M, Germain L, Buchan GC (1992) Comparison of immunocytochemical and Holzer’s methods for detection of acute and chronic gliosis in human postmortem material. J Neuropsychiatry Clin Neurosci 4:168–173. https://doi.org/10.1176/jnp.4.2.168
Arai K, Harada Y, Tomiyama H et al (2016) Evaluation of the survival of bone marrow-derived mononuclear cells and the growth factors produced upon intramedullary transplantation in rat models of acute spinal cord injury. Res Vet Sci 107:88–94. https://doi.org/10.1016/j.rvsc.2016.05.011
Fenn J, Drees R, Volk HA, De Decker S (2016) Comparison of clinical signs and outcomes between dogs with presumptive ischemic myelopathy and dogs with acute noncompressive nucleus pulposus extrusion. J Am Vet Med Assoc 249:767–775. https://doi.org/10.2460/javma.249.7.767
Webb AA, Ngan S, Fowler D (2010) Spinal cord injury II: Prognostic indicators, standards of care, and clinical trials. Can Vet J 51:598–604
Henke D, Vandevelde M, Doherr MG, Stöckli M, Forterre F (2013) Correlations between severity of clinical signs and histopathological changes in 60 dogs with spinal cord injury associated with acute thoracolumbar intervertebral disc disease. Vet J 198:70–75. https://doi.org/10.1016/j.tvjl.2013.04.003
Olby N, Levine J, Harris T, Muñana K, Skeen T, Sharp N (2003) Long-term functional outcome of dogs with severe injuries of the thoracolumbar spinal cord: 87 cases (1996–2001). J Am Vet Med Assoc 222:762–769. https://doi.org/10.2460/javma.2003.222.762
Tator CH, Koyanagi I (1997) Vascular mechanisms in the pathophysiology of human spinal cord injury. J Neurosurg 86:483–492. https://doi.org/10.3171/jns.1997.86.3.0483
Anwar MA, Al Shehab TS, Eid AH (2016) Inflammogenesis of secondary spinal cord injury. Front Cell Neurosci 10:98. https://doi.org/10.3389/fncel.2016.00098
Vandevelde M, Higgins RJ, Oevermann A (2002) Trauma. In: Veterinary neuropathology: essentials of theory and practice, 1st edn. Wiley-Blackwell, West Sussex, pp 81–91
Henke D, Gorgas D, Doherr MG, Howard J, Forterre F, Vandevelde M (2016) Longitudinal extension of myelomalacia by intramedullary and subdural haemorrhage in a canine model of spinal cord injury. Spine J 16:82–90. https://doi.org/10.1016/j.spinee.2015.09.018
Acknowledgments
We thank Jane Charbonneau, DVM, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Funding
This study did not receive any specific grants from funding agencies in the public, commercial, and government sources or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yuya Nakamoto, Gentarou Tsujimoto, Akito Ikemoto, Koichi Omori, and Tatsuo Nakamura declare that they have no conflict of interest.
Ethical approval
All animal experiments were performed in accordance with the Guidelines for Animal Experiments of Kyoto University (1989; approval number, R-16–28; approval date, 01 April, 2016) and the principles of laboratory animal care (NIH publication No. 86–23, revised 1985).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nakamoto, Y., Tsujimoto, G., Ikemoto, A. et al. Pathological changes within two weeks following spinal cord injury in a canine model. Eur Spine J 30, 3107–3114 (2021). https://doi.org/10.1007/s00586-021-06931-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-06931-z