Abstract
The Heart and kidney are adversely affected by arsenic toxicity, a highly toxic environmental pollutant.
The aim of this study was to evaluate cardio-renal protective effects of taurine against sodium arsenite–induced toxicity. Taurine (2-amino ethane sulphonic acid; TAU) is a major free intracellular amino acid in many mammalian tissues including the liver. Taurine supplementation has been documented to mitigate steatosis and hepatotoxicity in several animal models.
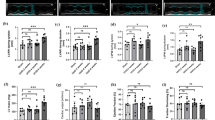
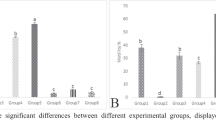
Sixty cockerel chicks (1-day old) of uniform weight were randomly assigned into six groups of ten cockerel chicks: Group A, Control; Group B, Sodium arsenite (SA) 75 mg/kg body weight; Group C, SA + 100 mg/kg taurine; Group D, SA + 200 mg/kg taurine; Group E, 100 mg/kg taurine and Group F, 200 mg/kg taurine. At the end of the experiment, blood pressure, electrocardiogram, electrolytes, and kidney function tests together with immunohistochemistry of cardiac troponin, gelatinase-associated lipocalin (NGAL), and podocin were assessed. Our findings revealed significant (P < 0.05) increases in blood pressure parameters, pulse rate, serum electrolytes (sodium, potassium, chloride, and bicarbonate) and markers of oxidative stress, prolonged QTc and PR interval with concurrent significant decrease in antioxidant status of cockerels intoxicated with SA. Immunohistochemistry revealed higher expressions of cardiac troponin, renal NGAL, and podocin in SA-intoxicated chicks relative to control and taurine-treated chicks.
Our study shows that taurine supplementation improved the electrolytes and electrocardiographic changes, attenuated oxidative stress biomarkers, high blood pressure, and lowered cardiac troponin, renal NGAL and podocin immuno-reactivity.












Similar content being viewed by others
Availability of data and materials
Data will be made available on request.
Code availability
All results from this research were expressed as mean ± standard deviation (SD) and the test of significance between two groups were estimated with Student’s t-test, The One-Way Analysis of Variance (ANOVA) with Tukey’s post hoc test. p-values < 0.05 considered statistically significant for all value. Graph pad prism 5.0 was used to analyze all values.
References
Adeyemo GO, Kabir AO, Tanimowo DA, Ologhobo AD (2018) Performance, blood chemistry, and serum electrolytes of broilers given water from different sources. Nig J Ani Sci 20(2):140–145
Adil M, Kandhare AD, Visnagri A, Bodhankar SL (2015) Naringin ameliorates sodium arsenite-induced renal and hepatic toxicity in rats: decisive role of KIM-1, Caspase-3, TGF-β, and TNF-α. Ren Fail 37(8):1396–1407
Adil M, Kandhare AD, Ghosh P, Bodhankar SL (2016) Sodium arsenite-induced myocardial bruise in rats: ameliorative effect of naringin via TGF-β/Smad and Nrf/HO pathways. Chemico-Biol Interact 253:66–77
Al-Brakati A, Alsharif KF, Alzahrani KJ, Kabrah S, Al-Amer O, Oyouni AA et al (2021) Using green biosynthesized lycopene-coated selenium nanoparticles to rescue renal damage in glycerol-induced acute kidney injury in rats. Inter J Nanomed 16:4335–4349
Buetler E, Duron O, Kelly MB (1963) Improved method for determination of blood glutathione reduced. J Lab Clin Meth 61:882–888
Bunderson M, Brooks DM, Walker DL, Rosenfeld ME, Coffin JD, Beall HD (2004) Arsenic exposure exacerbates atherosclerotic plaque formation and increases nitrotyrosine and leukotriene biosynthesis. Toxicol Appl Pharmacol 201(1):32–39. https://doi.org/10.1016/j.taap.2004.04.008
Capelli I, Vitali F, Zappulo F, Martini S, Donadei C, Cappuccilli M et al (2020) Biomarkers of kidney injury in very-low-birth-weight preterm infants: influence of maternal and neonatal factors. In Vivo 34(3):1333–1339
Chang L, Xu J, Yu F, Zhao J, Tang X, Tang C (2004) Taurine protected myocardialmitochondria injury induced by hyperhomocysteinemia in rats. Amino Acids 27:37–48
Chen Y, Factor-Litvak P, Howe GR (2011) Arsenic exposure from drinking water and mortality from cardiovascular disease in Bangladesh: prospective cohort study. Brit Med J 342:2431
Cohen SM, Arnold LL, Beck BD, Lewis AS, Eldan M (2013) Evaluation of the carcinogenicity of inorganic arsenic. Crit Rev Toxicol. 43(9):711–52
Das B, Rahman MM, Nayak B, Pal A, Chowdhury UK, Mukherjee SC et al (2009a) Groundwater arsenic contamination, its health effects and approach for mitigation in West Bengal, India and Bangladesh. Water Qual Exp Health 1(1):5–21
Das AK, Bag S, Sahu R, Dua TK, Sinha MK, Gangopadhyay M et al (2009b) Protective effect of Corchorus olitorius leaves on sodium arsenite-induced toxicity in experimental rats. Food Chem Toxicol 48(1):326–335
Das AK, Bag S, Sahu R, Dua TK, Sinha MK, Gangopadhyay M, Zaman K, Dewanjee S (2010) Protective effect of Corchorus olitorius leaves on sodium arsenite-induced toxicity in experimental rats. Food Chem Toxicol 48(2010):326–335
Drury RA, Wallington EA (1976) Editors Carlton’s histopathological techniques. 4th ed. London: Oxford University Press. 139–142
Ellman GL (1959) Tissue Sulfhydryl Groups. Arch Biochem Biophys 82:70–77
Foos TM, Wu JY (2002) The role of taurine in the central nervous system and the modulation of intracellular calcium homeostasis. Neurochem Res 27:21–26
Ghosh J, Das J, Manna P, Sil PC (2009) Taurine prevents arsenic induced cardiac oxidative stress and apoptotic damage: role of NF kappa B, p38 and JNK MAPK pathway. Toxicol Appl Pharmacol 240:73–87
Griffin BR, Faubel S, Edelstein CL (2019) Biomarkers of drug-induced kidney toxicity. Ther Drug Monit 41(2):213–226
Gross JL, De Azevedo MJ, Silveiro SP, Canani LH, Caramori ML, Zelmanovitz T (2005) Diabetic nephropathy: diagnosis, prevention, and treatment. Diabet Care 28:164–176
Guyton AC, Hall JE (2006) Textbook of medical physiology, 11th edn. Saunders WB Philadelphia 365–381
Habig WH, Pabst MJ, Jakpoby WB (1974) Glutathione transferase, a first enzymatic step in mercapturic acid formation. J Bio Chem 249:7130–7139
Haidara MA, Yassin HZ, Rateb M, Ammar H, Zorkani MA (2006) Role of oxidative stress in development of cardiovascular complications in diabetes mellitus. Curr Vasc Pharmacol 4:215–227
He W, Greenwell RJ, Brooks DM, Calderon-Garciduenas L, Beall HD, Coffin JD (2007) Arsenic exposure in pregnant mice disrupts placental vasculogenesis and causes spontaneous abortion. Toxicol Sci 99:244–253
Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133:1–16
Ito T, Kimura Y, Uozumi Y, Takai M, Muraoka S, Matsuda T et al (2008) Taurine depletion caused by knocking out the taurine transporter gene leads to cardiomyopathy with cardiac atrophy. J Mol Cell Cardiol 44:927–937
Ito T, Oishi S, Takai M, Kimura Y, Uozumi Y, Fujio Y et al (2010) Cardiac and skeletal muscle abnormality in taurine transporter-knockout mice. J Biomed Sci 17(1):20
Jamil S, Jamil G, Mesameh H, Qureshi A, AlKaabi J, Sharma C et al (2021) Risk factor comparison in young patients presenting with acute coronary syndrome with atherosclerotic coronary artery disease vs. angiographically normal coronaries. Inter J Med Sci 18(15):3526–3532
Jomova K, Jenisova Z, Feszterova M, Baros S, Liska J, Hudecova D et al (2011) Arsenic: toxicity, oxidative stress and human disease. J Appl Toxicol 31(2):95–107
Junyent F, Romero R, De Lemos L, Ultrera J, Camins A, Pallas M et al (2010) Taurine treatment inhibits CaMKII activity and modulates the presence of calbindin D28k, calretinin and parvalbumin in the brain. J Neurosci Res 88:136–142. https://doi.org/10.1002/jnr.22192
Kagawa T, Zárybnický T, Omi T, Shirai Y, Toyokuni S, Oda S et al (2019) A scrutiny of circulating microRNA biomarkers for drug-induced tubular and glomerular injury in rats. Toxicol 415:26–36
Karaarslan K, Abud B, Karacelik MCB (2021) Cardioplegia application with a hand-squeezed cardioplegia bag. Is it safe? Heart Surg Forum 24(4):619–623
Kenyon EM, Del Razo LM, Hughes MF (2005) Tissue distribution and urinary excretion of inorganic arsenic and its methylated metabolites in mice following acute oral administration of arsenate. Toxicol Sci 85(1):468–475
Leary S, Pharmaceuticals F, Ridge H, Underwood W, Anthony R, Cartner S et al (2020) AVMA guidelines for the euthanasia of animals. 77
Le Hir M, Keller C, Eschmann V, Hähnel B, Hosser H, Kriz W (2001) Podocyte bridges between the tuft and Bowman’s capsule: an early event in experimental crescentic glomerulonephritis. J Ame Soc Nephrol 12:2060–2071
Lee MY, Jung BI, Chung SM, Bae ON, Lee JY, Park JD et al (2003) Arsenic-induced dysfunction in relaxation of blood vessels. Environ Health Perspect 111:513–517
Madden EF, Fowler BA (2000) Mechanisms of nephrotoxicity from metal combinations: a review. Drug Chem Toxicol 23:1–12
Manterys A, Francyk-Zarow M, Czyzynska-Cichon I, Drahun A, Kus E, Szymczyk B et al (2016) Haematological parameters, serum lipid profile, liver function, and fatty acid profile of broiler chickens fed on diets supplemented with pomegranate seed oil and linseed oil. Brit Poult Sci 57:771–779
Ma X, Dang C, Kang H, Dai Z, Lin S, Guan H et al (2015) Saikosaponin-D reduces cisplatin-induced nephrotoxicity by repressing ROS-mediated activation of MAPK and NF-kappaB signalling pathways. Inter Immunopharmacol 28(1):399–408
Ma ZN, Liu Z, Wang Z, Ren S, Tang S, Wang YP et al (2017) Supplementation of American ginseng berry extract mitigated cisplatin-evoked nephrotoxicity by suppressing ROS-mediated activation of MAPK and NF-kappaB signaling pathways. Food Chem Toxicol 110:62–73
Mehrzadi S (2018) Ellagic acid mitigates sodium arsenite-induced renal and hepatic toxicity in male Wistar rats. Pharmacol Rep 70(4):712–719
Miller WH, Schipper HM, Lee JS, Singer J, Waxman S (2002) Mechanisms of action of arsenic trioxide. Cancer Res 62:3893–3903
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 217:3170–3175
Most P, Papenbrock J (2015) Possible roles of plant sulfurtransferases in detoxification of cyanide, reactive oxygen species, selected heavy metals and arsenate. Molecules 20:1410–1423
Murakami S, Sakurai T, Tomoike H, Sakono M, Nasu T, Fukuda N (2021) Prevention of hypercholesterolemia and atherosclerosis in the hyperlipidemia- and atherosclerosis-prone Japanese (LAP) quail by taurine supplementation. Amino Acids 38:271–278
Nachman KE, Raber G, Francesconi KA, Navas-Acien A, Love D (2012) Arsenic species in poultry feather meal. Sci Total Environ 15:417–418
Najafi L, Keshtkar RS, Pirsaheb S, Keyvani H, Khajavi A, Shati M et al (2021) Assessment of serum and urine neurophil gelatinase- associated lipocalin (s-NGAL and u-NGAL) level as a predictive factor of disease progression in diabetic nephropathy in type 2 DM. Iran J Kid Dis 15(4):270–278
Oyagbemi AA, Omobowale TO, Akinrinde AS, Saba AB, Ogunpolu BS, Daramola O (2015) Lack of reversal of oxidative damage in renal tissues of lead acetate-treated rats. Environ Toxicol 30:1235–1243
Oyagbemi AA, Omobowale TO, Awoyomi OV, Ajibade TO, Falayi OO, Ogunpolu BS, Okotie UJ, Asenuga ER, Adejumobi OA, Hassan FO, Ola-Davies OE, 1, Saba AB, Adedapo AA and MA Yakubu, (2019) Cobalt chloride toxicity elicited hypertension and cardiac complication via induction of oxidative stress and upregulation of COX-2/Bax signaling pathway. Hum Exp Toxicol 38(5):519–532
Omobowale TO, Esan OO, Adejumobi OA, Oladele OA (2017) Six-lead electrocardiographic studies of the pigeon (Columba livia) in Nigeria. Afri J Biomed Res 20(3):273–276
Oyagbemi AA, Omobowale TO, Asenuga ER, Abiola JO, Adedapo AA, Yakubu MA (2018) Kolaviron attenuated arsenic acid induced-cardiorenal dysfunction via regulation of ROS, C-reactive proteins (CRP), cardiac troponin I (CTnI) and BCL2. J Trad Comp Med 8(3):396–409
Pizzino G, Irrera N, Cucinotta M, Pallio G, Mannino F, Arcoraci V et al (2017) Oxidative stress: harms and benefits for human health. Oxid Med Cell Longev 2017:8416763
Prentice H, Modi JP, Wu JY (2015) Mechanisms of neuronal protection against excitotoxicity, endoplasmic reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative diseases. Oxid Med Cell Longev 2015:964518. https://doi.org/10.1155/2015/964518
Ramila KC, Jong CJ, Pastukh V, Ito T, Azuma J, Schaffer SW (2015) Role of protein phosphorylation in excitation-contraction coupling in taurine deficient hearts. Am J Physiol 308:H232–H239
Robles-Osorio ML, Sabath-Silva E, Sabath E (2015) Arsenic-mediated nephrotoxicity. Ren Fail 37(4):542–547
Saxena PN, Anand S, Saxena N, Bajaj P (2009) Effect of arsenic trioxide on renal functions and its modulation by Curcuma aromatica leaf extract in albino rat. J Environ Biol 30:527–531
Schaffer SW, Solodushko V, Kakhniashvili D (2002) Beneficial effect of taurine depletion on osmotic sodium and calcium loading during chemical hypoxia. Am J Physiol Cell Physiol 282(5):C1113–C1120. https://doi.org/10.1152/ajpcell.00485.2001
Seki E, Brenner DA, Karin M (2012) A liver full of JNK: signaling in regulation of cell function and disease pathogenesis, and clinical approaches. Gastroenterology 143 (2):307–320
Shenoy PS, Sen U, Kapoor S, Ranade AV, Chowdhury CR, Bose B (2019) Sodium fluoride induced skeletal muscle changes: degradation of proteins and signaling mechanism. Environ Pollut 244:534–548
Sheth SS, Deluna A, Allayee H, Lusis AJ (2002) Understanding atherosclerosis through mouse genetics. Curr Opin Lipidol 13:181–189
Singh AP, Goel RK, Kaur T (2011) Mechanisms pertaining to arsenic toxicity. Toxicol Inter 18(2):87–93
Singh R, Czaja MJ (2007) Regulation of hepatocyte apoptosis by oxidative stress. J Gastroenterol Hepatol 22(1):45–S48
Spickett CM (2013) The lipid peroxidation product 4-hydroxy-2- nonenal: advances in chemistry and analysis. Redox Biol 1:145–152
Susan A, Rajendran K, Sathyasivam K, Krishnan UM (2019) An overview of plant-based interventions to ameliorate arsenic toxicity. Biomed Pharmacoth 109:838–852
Tang X, Xu Y, Dai X, Xing Y, Yang D, Huang Q et al (2021) The long-term effect of dobutamine on intrinsic myocardial function and myocardial injury in septic rats with myocardial dysfunction. Shock 56(4):582–592
Varshney R, Kale RK (1990) Effect of calmodulin antagonists on radiation induced lipid peroxidation in microsomes. Inter J Rad Biol 58:733–743
Veltman D, Wu M, Pokreisz P, Claus P, Gillijns H, Caluwé E et al (2021) Clec4e-receptor signaling in myocardial repair after ischemia-reperfusion injury. JACC: Bas Transl Sci 6(8):631–646
Wang CH, Jeng JS, Yip PK, Chen CL, Hsu LI, Hsueh YM et al (2002) Biological gradient between long-term arsenic exposure and carotid atherosclerosis. Circ 105:1804–1809
WHO (2010) Ten chemicals of major public health concern. WHO 1–4
Wolff SF (1994) Ferrous ion oxidation in the presence of ferric ion indicator xylenol orange for measurement of hydrogen peroxides. Meth Enzymol 233:182–189
Wu H, Jin Y, Wei J, Jin H, Sha D, Wu JY (2005) Mode of action of taurine as a neuroprotector. Brain Res 1038:123–131. https://doi.org/10.1016/j.brainres.2005.01.058
Yang Y, Zhang Y, Liu X, Zuo J, Wang K, Liu W et al (2013) Exogenous taurine attenuates mitochondrial oxidative stress and endoplasmic reticulum stress in rat cardiomyocytes. Acta Bioch Biophy Sin 45:359–367
Yousef MI, El-Demerdash FM, Radwan FM (2008) Sodium arsenite induced biochemical perturbations in rats: ameliorating effect of curcumin. Food Chem Toxicol 46:3506–3511
Funding
This work was funded by the African Union, through the Institute of Life and Earth Sciences (including Health and Agriculture) Pan African University, (PAULESI), Nigeria.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The authors, Esan Oluwaseun, Adamu Shafiu Maikifi, Leah Oluwaseyanu Esuola, Temitayo Olabisi Ajibade, Ademola Adetokunbo Oyagabemi and Temidayo Olutayo Omobowale designed the experiment. Esan Oluwaseun, Adamu Shafiu Maikifi, Esuola Leah Oluwaseyanu, performed the immunohistochemistry and biochemical assays. The blood pressure and electrocardiogram were performed by Oluwaseun Esan, Adamu Shafiu Maikifi, and Temidayo Olutayo Omobowale. Moses Olusola Adetona, Ademola Adetokunbo Oyagbemi, Temitayo Olabisi Ajibade, Olumuyiwa Abiola Adejumobi, Temidayo Olutayo Omobowale, Omolade Abodunrin Oladele, Oluwafemi Oguntibeju, Evaristus Nwulia, and Momoh Audu Yakubu supervised, proof-read and approved the submission.
Corresponding author
Ethics declarations
Ethical approval
All the cockerels were kept in spacious cages under normal environmental condition as stipulated by Animal Use and Care Research Ethics Committee (ACUREC) Ibadan. The study was conducted in accordance with the provision of the University of Ibadan ACUREC with approval code (UI-ACUREC/027–0722/7) and according to the NIH Guideline for the handling and care of experimental animals.
Consent to participate
For this type of study informed consent is not required.
Consent for publication
For this type of study consent for publication is not required.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Esan, O., Maikifi, A.S., Esuola, L.O. et al. Taurine mitigates sodium arsenite–induced cardiorenal dysfunction in cockerel chicks: from toxicological, biochemical, and immunohistochemical stand-points. Comp Clin Pathol 32, 769–782 (2023). https://doi.org/10.1007/s00580-023-03485-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-023-03485-y