Abstract
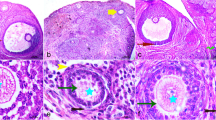
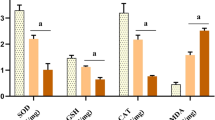
Exposure to inorganic arsenic is a notable health issue featuring several incidents of female reproductive dysfunctions. This study was designed to explore the extent of damages of sodium arsenite on female reproductive system of rats. Forty adult Wistar nulliparous rats having 3 months of age were grouped into four where the treated groups (Gr II, III, and IV) were gavaged with sodium arsenite (1, 3, and 5 mg/kg/day, respectively) for 30 consecutive days and a vehicle-fed group (Gr I) served as control. The treatment resulted in decrease of body weight (Gr III and IV) and weight of ovary and uterus (Gr II, III, and IV). Marked decrease of the pre-antrum, antral, and Graafian follicle pools with concomitant enhancement of follicular atresia (Gr III and IV) was noted but primordial and primary follicle pools remained unaltered. Compromised antioxidant defense in both the tissues was noted by altered enzymatic and non-enzymatic markers. The toxicity also resulted in the reduction of serum estradiol, LH, FSH and prolonged diestrus index. Alterations of uterine histoarchitecture with marked presence of shrunken endometrial glands, apoptotic bodies in the epithelium, and degenerated perimetrium were also noted. Elevated oxidative stress was further documented by increased presence of ROS in both the tissues. The subchronic exposure of sodium arsenite at higher doses caused progressive damages of folliculogenesis and uterine degeneration. This study demonstrates that the reproductive system of female rats is a target to sodium arsenite–induced ROS generation at higher doses.









Similar content being viewed by others
Availability of data and material
The data sets generated and/or analyzed during the study are included in the manuscript. The data that support this study will be shared upon reasonable request to the corresponding author.
Code availability
Not applicable.
References
Abercrombie M (1946) Estimation of nuclear population from microtome sections. Anat Rec 94:239–247. https://doi.org/10.1002/ar.1090940210
Agarwal A, Gupta S, Sharma RK (2005) Role of oxidative stress in female reproduction. Reprod Biol Endocrinol 3:28
Akram Z, Jalali S, Shami SA et al (2010) Adverse effects of arsenic exposure on uterine function and structure in female rat. Exp Toxicol Pathol 62(4):451–459. https://doi.org/10.1016/j.etp.2009.07.008
Akram Z, Jalali S, Shami SA et al (2009) Genotoxicity of sodium arsenite and DNA fragmentation in ovarian cells of rat. Toxicol Lett 190(1):81–85. https://doi.org/10.1016/j.toxlet.2009.07.003
Beers RF, Sizer IW (1952) A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem 195(1):133–140. https://doi.org/10.1093/jxb/48.2.181
Billig H, Furuta I, Hsueh AJW (1993) Estrogens inhibit and androgens enhance ovarian granulosa cell apoptosis. Endocrinology 133(5):2204–2212. https://doi.org/10.1210/endo.133.5.8404672
Biswas P, Mukhopadhyay A, Kabir SN, Mukhopadhyay PK (2019) High-protein diet ameliorates arsenic-induced oxidative stress and antagonizes uterine apoptosis in rats. Biol Trace Elem Res 192(2):222–233. https://doi.org/10.1007/s12011-019-1657-2
Chatterjee A, Chatterji U (2010) Arsenic abrogates the estrogen-signaling pathway in the rat uterus. Reprod Biol Endocrinol 8:80. https://doi.org/10.1186/1477-7827-8-80
Chattopadhyay S, Ghosh D (2010) Role of dietary GSH in the amelioration of sodium arsenite-induced ovarian and uterine disorders. Reprod Toxicol 30(3):481–488. https://doi.org/10.1016/j.reprotox.2010.05.002
Chattopadhyay S, Ghosh S, Chaki S et al (1999) Effect of sodium arsenite on plasma levels of gonadotrophins and ovarian steroidogenesis in mature albino rats: duration-dependent response. J Toxicol Sci 24(5):425–431. https://doi.org/10.2131/jts.24.5_425
Chattopadhyay S, Pal Ghosh S, Ghosh D, Debnath J (2003) Effect of dietary co-administration of sodium selenite on sodium arsenite-induced ovarian and uterine disorders in mature albino rats. Toxicol Sci 75(2):412–422
Ciani F, Cocchia N, d’Angelo D, Tafuri S (2015) Influence of ROS on ovarian functions. New Discoveries in Embryology. IntechOpen, London, pp 41–73
Denholm RB, More IA (1980) Atypical cilia of the human endometrial epithelium. J Anat 131(Pt2):309–315
Dezfouli MGZ, Eissazadeh S, Zade SMAS (2014) Histological and histometrical study of the protective role of α-tocopherol against sodium arsenite toxicity in rat ovaries. Microsc Microanal 20(4):1167–1179
Doroquez DB, Rebay I (2006) Signal integration during development: mechanisms of EGFR and notch pathway function and cross-talk. Crit Rev Biochem Mol Biol 41(6):339–385
Eruslanov E, Kusmartsev S (2010) Identification of ROS using oxidized DCFDA and flow-cytometry. Methods Mol Biol 594:57–72. https://doi.org/10.1007/978-1-60761-411-1_4
Guha Mazumder DN (2008) Chronic arsenic toxicity & human health. Indian J Med Res 128(4):436–447
Hirshfield AN, Midgley AR (1978) Morphometric analysis of follicular development in the rat. Biol Reprod 19(3):597–605. https://doi.org/10.1095/biolreprod19.3.597
Jana K, Jana S, Samanta PK (2006) Effects of chronic exposure to sodium arsenite on hypothalamo-pituitary-testicular activities in adult rats: possible an estrogenic mode of action. Reprod Biol Endocrinol 4:9. https://doi.org/10.1186/1477-7827-4-9
Jhala DD, Nair SB, Chinoy NJ (2004) Reversible toxicity of fluoride and arsenic in ovary of mice. Fluoride 37(2):71–79
Levine RL, Williams JA, Stadtman EP, Shacter E (1994) Carbonyl assays for determination of oxidatively modified proteins. Methods Enzymol 233:346–357. https://doi.org/10.1016/S0076-6879(94)33040-9
Lu J, Wang Z, Cao J et al (2018) A novel and compact review on the role of oxidative stress in female reproduction. Reprod Biol Endocrinol 16(1):80
Makanji Y, Tagler D, Pahnke J et al (2014) Hypoxia-mediated carbohydrate metabolism and transport promote early-stage murine follicle growth and survival. Am J Physiol - Endocrinol Metab 306(8):E893-903. https://doi.org/10.1152/ajpendo.00484.2013
Martin JP, Dailey M, Sugarman E (1987) Negative and positive assays of superoxide dismutase based on hematoxylin autoxidation. Arch Biochem Biophys 255(2):329–336. https://doi.org/10.1016/0003-9861(87)90400-0
Mondal R, Biswas S, Chatterjee A et al (2016) Protection against arsenic-induced hematological and hepatic anomalies by supplementation of Vitamin C and vitamin e in adult male rats. J Basic Clin Physiol Pharmacol 27(6):643–652. https://doi.org/10.1515/jbcpp-2016-0020
Navarro PAAS, Liu L, Keefe DL (2004) In vivo effects of arsenite on meiosis, preimplantation development, and apoptosis in the mouse. Biol Reprod 70(4):980–985. https://doi.org/10.1095/biolreprod.103.020586
Nemec MD, Holson JF, Farr CH, Hood RD (1998) Developmental toxicity assessment of arsenic acid in mice and rabbits. Reprod Toxicol 12(6):647–658. https://doi.org/10.1016/S0890-6238(98)00053-7
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Pachnanda R, Singh SP (2012) Histopathological alterations in testicular tissue of male rats exposed to arsenic. J Appl Nat Sci 4(2):247–251. https://doi.org/10.31018/jans.v4i2.258
Parhizkar S, Latiff LA, Rahman SA et al (2011) Assessing estrogenic activity of nigella sativa in ovariectomized rats using vaginal cornification assay. African J Pharm Pharmacol 5(2):137–142. https://doi.org/10.5897/ajpp10.276
Perez GI, Tao XJ, Tilly JL (1999) Fragmentation and death (a.k.a. apoptosis) of ovulated oocytes. Mol Hum Reprod 5(5):414–20. https://doi.org/10.1093/molehr/5.5.414
Regan SLP, Knight PG, Yovich JL et al (2018) Granulosa cell apoptosis in the ovarian follicle-a changing view. Front Endocrinol (lausanne) 9:61
Sharma S, Kaur T, Sharma AK, et al (2021) Betaine attenuates sodium arsenite-induced renal dysfunction in rats. Drug Chem Toxicol 11:(1–8). https://doi.org/10.1080/01480545.2021.1959699
Shin J, Seol I, Son C (2010) Interpretation of animal dose and human equivalent dose for drug development. J Korean Orient Med 31(3):1–7
Simpson E, Lauber M, Demeter M et al (1991) Regulation of expression of the genes encoding steroidogenic enzymes in the ovary. J Steroid Biochem Mol Biol 40(1–3):45–52. https://doi.org/10.1016/0960-0760(92)90366-Q
Souza ACF, Ervilha LOG, Coimbra JLP et al (2020) Reproductive disorders in female rats after prenatal exposure to sodium arsenite. J Appl Toxicol 40(2):214–223. https://doi.org/10.1002/jat.3897
Stadtman ER (2006) Protein oxidation and aging. Free Radic Res 40(12):1250–1258. https://doi.org/10.1080/10715760600918142
Suzuki T, Sugino N, Fukaya T et al (1999) Superoxide dismutase in normal cycling human ovaries: Immunohistochemical localization and characterization. Fertil Steril 72(4):720–726. https://doi.org/10.1016/S0015-0282(99)00332-5
Thangapandiyan S, Ramesh M, Miltonprabu S et al (2019) Sulforaphane potentially attenuates arsenic-induced nephrotoxicity via the PI3K/Akt/Nrf2 pathway in albino Wistar rats. Environ Sci Pollut Res 26(12):12247–12263. https://doi.org/10.1007/s11356-019-04502-w
Vegetti W, Alagna F (2006) FSH and folliculogenesis: from physiology to ovarian stimulation. Reprod Biomed Online 12(6):684–694. https://doi.org/10.1016/S1472-6483(10)61080-2
Wang A, Holladay SD, Wolf DC et al (2006) Reproductive and developmental toxicity of arsenic in rodents: a review. Int J Toxicol 25(5):319–331. https://doi.org/10.1080/10915810600840776
Ye X (2020) Uterine luminal epithelium as the transient gateway for embryo implantation. trends Endocrinol. Metab 31(2):165–180
Acknowledgements
The authors would like to acknowledge Dr. Souryadeep Mukherjee, Department of Life Sciences, Presidency University, and Dr. Sumit Mandal, Department of Life Sciences, Presidency University, for their help.
Funding
This work is mainly supported by the grant received from the Department of Higher Education, Science & Technology and Biotechnology, Government of West Bengal (Memo No. 265(Sanc.) / ST/ P/ S&T/1G-44/2017 dated 25.03.2018). Apart from this, the authors also would like to acknowledge the DST-FIST grant of the Government of India and FRPDF grant of Presidency University.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: RM, AM, AC, AB, and PKM. Performed the experiments: RM. Analyzed the data: RM, AM, AC, AB, and PKM. Wrote the paper: RM. Overall manuscript checked: RM, AC, AB, AM, and PKM. Manuscript critically checked: RM, PKM. Final approval was given by all the authors.
Corresponding author
Ethics declarations
Ethics approval
The experiments were performed following the national guidelines implemented by the Committee for the Purpose of Control and Supervision of Experiments on Animals, India, with approval from the Animal Ethics Committee of Presidency University (Sanction number: PU/IAEC/PM/12).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mondal, R., Mukhopadhyay, A., Chattopadhyay, A. et al. Ovarian follicular atresia and uterine toxicity after subchronic oral exposure of postpubertal rats to sodium arsenite. Comp Clin Pathol 31, 597–612 (2022). https://doi.org/10.1007/s00580-022-03358-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-022-03358-w