Abstract
Background
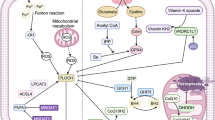
Photodynamic therapy (PDT) is an effective tumor treatment that involves the administration of a photosensitizer to generate cytotoxic 1O2 [reactive oxygen species (ROS)] from molecular oxygen that is produced from energy absorption following tumor irradiation at specific wavelengths. Ferroptosis is induced by the disruption of the glutathione peroxidase 4 (GPX4) antioxidant system, leading to lipid peroxidation. We hypothesized that talaporfin sodium-photodynamic therapy (TS-PDT)-generated ROS would lead to ferroptosis via accumulation of lipid peroxidation.
Methods
Cell viability assay in TS-PDT-treated cells in combination with a ferroptosis inhibitor (ferrostatin-1: Fer-1) or ferroptosis inducers (imidazole ketone erastin: IKE, Ras-selective lethal 3: RSL3) was performed. Accumulation of lipid peroxidation, GPX4 antioxidant system and cystine/glutamate antiporter (system xc−) activity in TS-PDT-treated cells was investigated. In xenograft mice, the antitumor effect of TS-PDT in combination with ferroptosis inducers (IKE or sorafenib) was examined.
Results
TS-PDT-induced cell death was partly suppressed by Fer-1 and accompanied by lipid peroxidation. TS-PDT combined with IKE or RSL3 enhanced the induction of cell death. TS-PDT inhibited cystine uptake activity via system xc−. In vivo, the combination of TS-PDT and ferroptosis inducers (IKE or sorafenib) reduced tumor volume.
Conclusion
This study found that the mechanism underlying TS-PDT-induced ferroptosis constitutes direct lipid peroxidation by the generated ROS, and the inhibition of system xc−, and that the combination of a ferroptosis inducer with TS-PDT enhances the antitumor effect of TS-PDT. Our findings suggest that ferroptosis-inducing therapies combined with PDT may benefit cancer patients.






Similar content being viewed by others
Abbreviations
- ATM:
-
Ataxia telangiectasia-mutated
- FBS:
-
Fetal bovine serum
- Fer-1:
-
Ferrostatin-1
- FSP1:
-
Ferroptosis suppressor protein 1
- GSH:
-
Reduced glutathione
- GPX4:
-
Glutathione peroxidase 4
- IKE:
-
Imidazole ketone erastin
- MDA:
-
Malondialdehyde
- NAC:
-
N-Acetylcysteine
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate hydrogen
- Nec-1s:
-
Necrostatin-1s
- PBS:
-
Phosphate-buffered saline
- PDT:
-
Photodynamic therapy
- PUFAs:
-
Polyunsaturated fatty acids
- RIPK:
-
Receptor-interacting protein kinase
- ROS:
-
Reactive oxygen species
- RSL3:
-
Ras-selective lethal 3
- SLC3A2:
-
Solute carrier family 3 member 2
- SLC7A11:
-
Solute carrier family 7 member 11
- TBARS:
-
Thiobarbituric acid-reactive substance
- TS:
-
Talaporfin sodium
References
Dolmans DE, Fukumura D, Jain RK. Photodynamic therapy for cancer. Nat Rev Cancer. 2003;3:380–7.
Agostinis P, Berg K, Cengel KA, et al. Photodynamic therapy of cancer: an update. CA A Cancer J Clin. 2011. https://doi.org/10.3322/caac.20114.
Brown SB, Brown EA, Walker I. The present and future role of photodynamic therapy in cancer treatment. Lancet Oncol. 2004;5:497–508.
Yano T, Minamide T, Takashima K, et al. Clinical practice of photodynamic therapy using talaporfin sodium for esophageal cancer. J Clin Med. 2021. https://doi.org/10.3390/jcm10132785.
Yano T, Kasai H, Horimatsu T, et al. A multicenter phase II study of salvage photodynamic therapy using talaporfin sodium (ME2906) and a diode laser (PNL6405EPG) for local failure after chemoradiotherapy or radiotherapy for esophageal cancer. Oncotarget. 2017;8:22135–44.
Dang J, He H, Chen D, et al. Manipulating tumor hypoxia toward enhanced photodynamic therapy (PDT). Biomater Sci. 2017;5:1500–11.
Li RQ, Zhang C, Xie BR, et al. A two-photon excited O(2)-evolving nanocomposite for efficient photodynamic therapy against hypoxic tumor. Biomaterials. 2019;194:84–93.
Liu LH, Zhang YH, Qiu WX, et al. Dual-stage light amplified photodynamic therapy against hypoxic tumor based on an O(2) self-sufficient nanoplatform. Small. 2017. https://doi.org/10.1002/smll.201701621.
Liu WL, Liu T, Zou MZ, et al. Aggressive man-made red blood cells for hypoxia-resistant photodynamic therapy. Adv Mater (Deerfield Beach, Fla). 2018;30:e1802006.
Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72.
Martin-Sanchez D, Ruiz-Andres O, Poveda J, et al. Ferroptosis, but not necroptosis, is important in nephrotoxic folic acid-induced AKI. J Am Soc Nephrol. 2017;28:218–29.
Carlson BA, Tobe R, Yefremova E, et al. Glutathione peroxidase 4 and vitamin E cooperatively prevent hepatocellular degeneration. Redox Biol. 2016;9:22–31.
Guiney SJ, Adlard PA, Bush AI, et al. Ferroptosis and cell death mechanisms in Parkinson’s disease. Neurochem Int. 2017;104:34–48.
Wu X, Li Y, Zhang S, et al. Ferroptosis as a novel therapeutic target for cardiovascular disease. Theranostics. 2021;11:3052–9.
Carneiro BA, El-Deiry WS. Targeting apoptosis in cancer therapy. Nat Rev Clin Oncol. 2020;17:395–417.
Viswanathan VS, Ryan MJ, Dhruv HD, et al. Dependency of a therapy-resistant state of cancer cells on a lipid peroxidase pathway. Nature. 2017;547:453–7.
Hangauer MJ, Viswanathan VS, Ryan MJ, et al. Drug-tolerant persister cancer cells are vulnerable to GPX4 inhibition. Nature. 2017;551:247–50.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82.
Yang WS, Stockwell BR. Ferroptosis: death by lipid peroxidation. Trends Cell Biol. 2016;26:165–76.
Zhu T, Shi L, Yu C, et al. Ferroptosis promotes photodynamic therapy: supramolecular photosensitizer-inducer nanodrug for enhanced cancer treatment. Theranostics. 2019;9:3293–307.
Xu T, Ma Y, Yuan Q, et al. enhanced ferroptosis by oxygen-boosted phototherapy based on a 2-in-1 nanoplatform of ferrous hemoglobin for tumor synergistic therapy. ACS Nano. 2020;14:3414–25.
Miotto G, Rossetto M, Di Paolo ML, et al. Insight into the mechanism of ferroptosis inhibition by ferrostatin-1. Redox Biol. 2020;28:101328.
Karuppagounder SS, Alin L, Chen Y, et al. N-acetylcysteine targets 5 lipoxygenase-derived, toxic lipids and can synergize with prostaglandin E(2) to inhibit ferroptosis and improve outcomes following hemorrhagic stroke in mice. Ann Neurol. 2018;84:854–72.
Slee EA, Zhu H, Chow SC, et al. Benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone (Z-VAD.FMK) inhibits apoptosis by blocking the processing of CPP32. Biochem J. 1996. https://doi.org/10.1042/bj3150021.
Mikuš P, Pecher D, Rauová D, et al. Determination of novel highly effective necrostatin Nec-1s in rat plasma by high performance liquid chromatography hyphenated with quadrupole-time-of-flight mass spectrometry. Molecules (Basel, Switzerland). 2018. https://doi.org/10.3390/molecules23081946.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods (San Diego, Calif). 2001;25:402–8.
Dixon SJ, Patel DN, Welsch M, et al. Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. Elife. 2014. https://doi.org/10.7554/eLife.02523.
Sengupta A, Lichti UF, Carlson BA, et al. Targeted disruption of glutathione peroxidase 4 in mouse skin epithelial cells impairs postnatal hair follicle morphogenesis that is partially rescued through inhibition of COX-2. J Invest Dermatol. 2013;133:1731–41.
Shimomura T, Hirakawa N, Ohuchi Y, et al. Simple fluorescence assay for cystine uptake via the xCT in cells using selenocystine and a fluorescent probe. ACS Sensors. 2021;6:2125–8.
Koppula P, Zhuang L, Gan B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. 2021;12:599–620.
Oleinick NL, Morris RL, Belichenko I. The role of apoptosis in response to photodynamic therapy: what, where, why, and how. Photochem Photobiol Sci Off J Eur Photochem Assoc Eur Soc Photobiol. 2002;1:1–21.
Aniogo EC, George BPA, Abrahamse H. Role of Bcl-2 family proteins in photodynamic therapy mediated cell survival and regulation. Molecules (Basel, Switzerland). 2020. https://doi.org/10.3390/molecules25225308.
Miki Y, Akimoto J, Moritake K, et al. Photodynamic therapy using talaporfin sodium induces concentration-dependent programmed necroptosis in human glioblastoma T98G cells. Lasers Med Sci. 2015;30:1739–45.
Song R, Li T, Ye J, et al. Acidity-activatable dynamic nanoparticles boosting ferroptotic cell death for immunotherapy of cancer. Adv Mater (Deerfield Beach, Fla). 2021;33:e2101155.
Zhou Y, Chen K, Lin WK, et al. Photo-enhanced synergistic induction of ferroptosis for anti-cancer immunotherapy. Adv Healthc Mater. 2023. https://doi.org/10.1002/adhm.202300994.
Chen Q, Ma X, Xie L, et al. Iron-based nanoparticles for MR imaging-guided ferroptosis in combination with photodynamic therapy to enhance cancer treatment. Nanoscale. 2021;13:4855–70.
Plaetzer K, Kiesslich T, Krammer B, et al. Characterization of the cell death modes and the associated changes in cellular energy supply in response to AlPcS4-PDT. Photochem Photobiol Sci Off J Eur Photochem Assoc Eur Soc Photobiol. 2002;1:172–7.
Hassannia B, Vandenabeele P, Vanden BT. Targeting ferroptosis to iron out cancer. Cancer Cell. 2019;35:830–49.
Ye LF, Chaudhary KR, Zandkarimi F, et al. Radiation-induced lipid peroxidation triggers ferroptosis and synergizes with ferroptosis inducers. ACS Chem Biol. 2020;15:469–84.
Lei G, Zhang Y, Koppula P, et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 2020;30:146–62.
Shui S, Zhao Z, Wang H, et al. Non-enzymatic lipid peroxidation initiated by photodynamic therapy drives a distinct ferroptosis-like cell death pathway. Redox Biol. 2021;45:102056.
Turubanova VD, Balalaeva IV, Mishchenko TA, et al. Immunogenic cell death induced by a new photodynamic therapy based on photosens and photodithazine. J Immunother Cancer. 2019;7:350.
Lang X, Green MD, Wang W, et al. Radiotherapy and Immunotherapy promote tumoral lipid oxidation and ferroptosis via synergistic repression of SLC7A11. Cancer Discov. 2019;9:1673–85.
Tanaka M, Sasaki M, Suzuki T, et al. Combination of talaporfin photodynamic therapy and Poly (ADP-Ribose) polymerase (PARP) inhibitor in gastric cancer. Biochem Biophys Res Commun. 2021;539:1–7.
Seibt TM, Proneth B, Conrad M. Role of GPX4 in ferroptosis and its pharmacological implication. Free Radical Biol Med. 2019;133:144–52.
Stockwell BR, Jiang X. The chemistry and biology of ferroptosis. Cell Chem Biol. 2020;27:365–75.
Sato H, Shiiya A, Kimata M, et al. Redox imbalance in cystine/glutamate transporter-deficient mice. J Biol Chem. 2005;280:37423–9.
Acknowledgements
We would like to thank Suzuka Asai and Yukimi Ito for their technical assistance. We are grateful for the assistance of the Research Equipment Sharing Center at the Nagoya City University.
Funding
This work was partially supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (2020–2022) (grant number 20K08361) (to M. Tanaka); JSPS KAKENHI (2023–2025) (grant number 23K07358) (to M. Tanaka); Toyoaki Scholarship Foundation (2022–2023) (grant number JOSE203179) (to M. Tanaka); Iketani Science and Technology Foundation (2022–2023) (grant number JOSE204007) (to M. Tanaka); JSPS KAKENHI (2022–2023) (grant number 22K20862) (to M. Sasaki); Grant from Bristol Myers Squibb (grant number JOSE202103) (to H. Kataoka) and JSPS KAKENHI (2023–2025) (grant number 23K07421) (to H. Kataoka); JSPS KAKENHI (2023–2024) (grant number 23K15019) (to M. Sasaki). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Study design was performed by YK and MT. Material preparation, data collection, and analysis were performed by YK, MS and MT. The first draft of the manuscript was written by YK. Funding was acquired by MS, MT and HK. All the authors commented on the final manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflicts of interest to disclose.
Ethics approval
Our study was approved by the Animal Research Committee of the Nagoya City University (project number: 21-014).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kojima, Y., Tanaka, M., Sasaki, M. et al. Induction of ferroptosis by photodynamic therapy and enhancement of antitumor effect with ferroptosis inducers. J Gastroenterol 59, 81–94 (2024). https://doi.org/10.1007/s00535-023-02054-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-023-02054-y