Abstract
Background
This study aims to investigate the effect of PD-1/PD-L1 immunotherapy on cardiac-related adverse events in patients with advanced or metastatic lung cancer.
Methods
We conducted a detailed search in PubMed, Web of Science, Cochran, and Embase for articles on the application of immunotherapy for lung cancer and report cardiac-related adverse events with respect to myocardial ischemia, pericardial effusion, myocarditis, and electrophysiology. The dichotomous variables were assessed by relative risk (RR) and 95% confidence intervals (CI).
Results
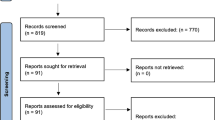
A total of 7132 subjects were included in 12 phase III randomized controlled trials (RCTs). The results showed that under the fixed effects model, the probability of cardiac-related adverse events in pericardial effusion was higher in the experimental group than in the control group (RR 2.30, 95% CI 1.01–5.21, P = 0.05). Under the random effects model, there was no statistical difference between the two groups (RR 2.03, 95% CI 0.81–5.12, P = 0.13). No statistical difference is observed between the experimental group and the control group (under the fixed effects model and the random effects model) for other cardiac-related adverse events, including myocarditis, acute coronary syndrome, myocardial infarction, acute myocardial infarction, myocardial ischemia, unstable angina, ventricular tachycardia, supraventricular tachycardia, tachycardia, bradycardia, atrial flutter, atrial fibrillation, cardiac failure, cardiac arrest, cardiopulmonary failure, acute heart failure, cardiac arrest (all P > 0.05).
Conclusions
PD-1/PD-L1 immunotherapy in advanced or metastatic lung cancer is generally safe for cardiac-related adverse events.



Similar content being viewed by others
Data availability
The datasets supporting the conclusions of this article are included within the article. If you want detailed data about this article, please contact the corresponding author.
Abbreviations
- RR :
-
Relative ratio
- CI :
-
Confidence interval
- PD-1 :
-
Programmed cell death-1
- PD-L1 :
-
Programmed cell death-ligand 1
- IL-1a :
-
Interleukin-1a
- IL-2 :
-
Interleukin-2
- IFNα2 :
-
Interferonα2
- IL-17 :
-
Interleukin-17
- MRI :
-
Magnetic resonance imaging
- CT :
-
Computed tomography
- ICI :
-
Immune checkpoint inhibitors
- CTLA-4 :
-
Cytotoxic T lymphocyte-associated antigen-4
- RCT :
-
Randomized clinical trial
References
Thai AA et al (2021) Lung cancer. Lancet 398(10299):535–554
Sung H et al (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Lahiri A et al (2023) Lung cancer immunotherapy: progress, pitfalls, and promises. Mol Cancer 22(1):40
Antonia SJ et al (2017) Durvalumab after chemoradiotherapy in stage III non–small-cell lung cancer. N Engl J Med 377(20):1919–1929
Garassino MC et al (2020) Patient-reported outcomes following pembrolizumab or placebo plus pemetrexed and platinum in patients with previously untreated, metastatic, non-squamous non-small-cell lung cancer (KEYNOTE-189): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol 21(3):387–397
Jotte R et al (2020) Atezolizumab in combination with carboplatin and nab-paclitaxel in advanced squamous NSCLC (IMpower131): results from a randomized phase III trial. J Thorac Oncol 15(8):1351–1360
Nishio M et al (2021) Atezolizumab plus chemotherapy for first-line treatment of nonsquamous NSCLC: results from the randomized phase 3 IMpower132 trial. J Thorac Oncol 16(4):653–664
Owonikoko TK et al (2021) Nivolumab and ipilimumab as maintenance therapy in extensive-disease small-cell lung cancer: CheckMate 451. J Clin Oncol 39(12):1349–1359
Paz-Ares L et al (2019) Durvalumab plus platinum–etoposide versus platinum–etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial. Lancet 394(10212):1929–1939
Rudin CM et al (2020) Pembrolizumab or placebo plus etoposide and platinum as first-line therapy for extensive-stage small-cell lung cancer: randomized, double-blind, phase III KEYNOTE-604 study. J Clin Oncol 38(21):2369–2379
Socinski MA et al (2018) Atezolizumab for first-line treatment of metastatic nonsquamous NSCLC. N Engl J Med 378(24):2288–2301
Sugawara S et al (2021) Nivolumab with carboplatin, paclitaxel, and bevacizumab for first-line treatment of advanced nonsquamous non-small-cell lung cancer. Ann Oncol 32(9):1137–1147
West H et al (2019) Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 20(7):924–937
Yang Y et al (2020) Efficacy and safety of sintilimab plus pemetrexed and platinum as first-line treatment for locally advanced or metastatic nonsquamous NSCLC: a randomized, double-blind, phase 3 study (Oncology pRogram by InnovENT anti-PD-1-11). J Thorac Oncol 15(10):1636–1646
Zhou C et al (2021) Sintilimab plus platinum and gemcitabine as first-line treatment for advanced or metastatic squamous NSCLC: results from a randomized, double-blind, phase 3 trial (ORIENT-12). J Thorac Oncol 16(9):1501–1511
Zhou YW et al (2019) Immune checkpoint inhibitor-associated cardiotoxicity: current understanding on its mechanism, diagnosis and management. Front Pharmacol 10:1350
Johnson DB et al (2016) Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med 375(18):1749–1755
Baik AH et al (2021) Mechanisms of cardiovascular toxicities associated with immunotherapies. Circ Res 128(11):1780–1801
Mahmood SS et al (2018) Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol 71(16):1755–1764
Heinzerling L et al (2016) Cardiotoxicity associated with CTLA4 and PD1 blocking immunotherapy. J Immunother Cancer 4:50
Cham J, Ng D, Nicholson L (2021) Durvalumab-induced myocarditis, myositis, and myasthenia gravis: a case report. J Med Case Reports 15(1):278
Nishimura T et al (2023) Fulminant myocarditis for non-small-cell carcinoma of the lung with nivolumab and ipilimumab plus chemotherapy. Intern Med 62(9):1319–1322
Sławiński G et al (2020) Immune checkpoint inhibitors and cardiac toxicity in patients treated for non-small lung cancer: a review. Int J Mol Sci 21(19):7195
Yun S et al (2015) Late onset ipilimumab-induced pericarditis and pericardial effusion: a rare but life threatening complication. Case Rep Oncol Med 2015:1–5
Gan L et al (2022) Cardiotoxicity associated with immune checkpoint inhibitors: current status and future challenges. Front Pharmacol 13:962596
Katsume Y et al (2018) Complete atrioventricular block associated with pembrolizumab-induced acute myocarditis: the need for close cardiac monitoring. Intern Med 57(21):3157–3162
Läubli H et al (2015) Acute heart failure due to autoimmune myocarditis under pembrolizumab treatment for metastatic melanoma. J Immunother Cancer 3:11
Wu T et al (2023) Case report: replacement of PD-1 inhibitors with PD-L1 inhibitors in the treatment of squamous non-small-cell lung carcinoma. Front Immunol 14:1243980
Funding
This study was supported by Ningbo Natural Science Foundation (No. 2022J039), The Medical and Health Research Project of Zhejiang (No.2022KY343), and The Science and Technology Project of Yinzhou (2022AS069).
Author information
Authors and Affiliations
Contributions
Each author contributed significantly to the conception and development of the present paper. Bangsheng Chen and Anyi Xu designed the research process. Yujing He, Yueming Zhang, and Yingying Fang searched the database for corresponding articles and extracted useful information from the articles above. Jiale Ruan, Dongying Cheng, and Min Dai used statistical software for analysis. Yu Wang and Jie Zhang drafted the meta-analysis. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable (this paper was provided based on researching in global databases).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, B., Xu, A., He, Y. et al. The cardiac-related adverse events of PD-1/PD-L1 immunotherapy in advanced or metastatic lung cancer: a RCT-based meta-analysis. Support Care Cancer 32, 140 (2024). https://doi.org/10.1007/s00520-024-08344-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08344-2