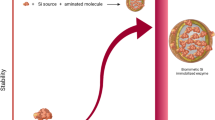
Abstract
High temperatures, harsh pH conditions, and toxic chemicals involved in the conventional synthesis and coating of silica limit the fabrication of new-generation hybrid materials immobilizing live cells and biomolecules such as enzymes and drugs. This hinders the application of inorganic–organic biohybrid materials in various fields, including bioelectronics, energy generation, and biomedicine. Silicatein, an enzyme found in siliceous sponges, catalyzes the polymerization of silica under mild conditions, that is, at room temperature and neutral pH. Silicatein was fused with a chitin-binding domain (ChBD) to selectively bind the fusion silicatein on the chitin material and with a small soluble tag called InakC, a hydrophilic protein from Pseudomonas syringae, to control the unfavorable aggregation of silicatein. The fusion silicatein was soluble in aqueous media and was successfully found to be adsorbed on the chitin material. The immobilized fusion silicatein acted as an interfacial catalyst to fabricate silica on chitin under ambient conditions. This technique can be used to fabricate inorganic–organic hybrid materials to immobilize biomolecules and can be applied to develop novel biocatalytic systems, biosensors, and tissue culture scaffolds.





Similar content being viewed by others
References
Sanchez C, Julián B, Belleville P, Popall M (2005) Applications of hybrid organic-inorganic nanocomposites. J Mater Chem 15:3559–3592
Aversa R, Petrescu RVV, Apicella A, Petrescu FIT (2017) Nano-diamond hybrid materials for structural biomedical application. Am J Biochem Biotechnol 13:34–41
Cai WY, Xu Q, Zhao XN et al (2006) Porous gold-nanoparticle-CaCO3 hybrid material: Preparation, characterization, and application for horseradish peroxidase assembly and direct electrochemistry. Chem Mater 18:279–284
Asefa T, Tao Z (2012) Biocompatibility of mesoporous silica nanoparticles. Chem Res Toxicol 25:2265–2284
Trewyn BG, Giri S, Slowing II, Lin VSY (2007) Mesoporous silica nanoparticle based controlled release, drug delivery, and biosensor systems. Chem Commun 38(31):3236–3245
Luckarift HR, Spain JC, Naik RR, Stone MO (2004) Enzyme immobilization in a biomimetic silica support. Nat Biotechnol 22:211–213
Rai A, Perry CC (2010) Facile fabrication of uniform silica films with tunable physical properties using silicatein protein from sponges. Langmuir 26:4152–4159
Povarova NV, Barinov NA, Baranov MS et al (2018) Efficient silica synthesis from tetra(glycerol)orthosilicate with cathepsin- and silicatein-like proteins. Sci Rep 8:16759
Müller WEG, Rothenberger M, Boreiko A et al (2005) Formation of siliceous spicules in the marine demosponge Suberites domuncula. Cell Tissue Res 321:285–297
Müller WEG, Schröder HC, Burghard Z et al (2013) Silicateins—a novel paradigm in bioinorganic chemistry: Enzymatic synthesis of inorganic polymeric silica. Chem A Eur J 19:5790–5804
Shkryl YN, Bulgakov VP, Veremeichik GN et al (2016) Bioinspired enzymatic synthesis of silica nanocrystals provided by recombinant silicatein from the marine sponge Latrunculia oparinae. Bioprocess Biosyst Eng 39:53–58
Wysokowski M, Jesionowski T, Ehrlich H (2018) Biosilica as a source for inspiration in biological materials science. Am Mineral 103:665–691
Ehrlich H, Deutzmann R, Brunner E et al (2010) Mineralization of the metre-long biosilica structures of glass sponges is templated on hydroxylated collagen. Nat Chem 2:1084–1088
Qin Z, Lin S, Qiu Y et al (2019) One-step immobilization-purification of enzymes by carbohydrate-binding module family 56 tag fusion. Food Chem 299:125037
Faustini M, Nicole L, Ruiz-Hitzky E, Sanchez C (2018) History of organic-inorganic hybrid materials: prehistory, art, science, and advanced applications. Adv Funct Mater 28:1704158
Ehrlich H, Janussen D, Simon P et al (2008) Nanostructural organization of naturally occurring composites-Part II: Silica-chitin-based biocomposites. J Nanomater 2008:1–8
Brunner E, Richthammer P, Ehrlich H et al (2009) Chitin-based organic networks: An integral part of cell wall biosilica in the diatom Thalassiosira pseudonana. Angew Chemie Int Ed 48:9724–9727
Ehrlich H, Simon P, Carrillo-Cabrera W et al (2010) Insights into chemistry of biological materials: Newly discovered silica-aragonite-chitin biocomposites in demosponges. Chem Mater 22:1462–1471
Ehrlich H, Krautter M, Hanke T et al (2007) First evidence of the presence of chitin in skeletons of marine sponges. Part II. Glass sponges (Hexactinellida: Porifera). J Exp Zool Part B Mol Dev Evol 308B:473–483
Ehrlich H, Maldonado M, Parker AR et al (2016) Supercontinuum generation in naturally occurring glass sponges spicules. Adv Opt Mater 4:1608–1613
Ehrlich H (2010) Chitin and collagen as universal and alternative templates in biomineralization. Int Geol Rev 52:661–699
Jayakumar R, Menon D, Manzoor K et al (2010) Biomedical applications of chitin and chitosan based nanomaterials—a short review. Carbohydr Polym 82:227–232
Madhumathi K, Binulal NS, Nagahama H et al (2009) Preparation and characterization of novel β-chitin-hydroxyapatite composite membranes for tissue engineering applications. Int J Biol Macromol 44:1–5
Borchard G (2001) Chitosans for gene delivery. Adv Drug Deliv Rev 52:145–150
Itoh Y, Kawase T, Nikaidou N et al (2002) Functional analysis of the chitin-binding domain of a family 19 chitinase from Streptomyces griseus HUT6037: substrate-binding affinity and cis-dominant increase of antifungal function. Biosci Biotechnol Biochem 66:1084–1092
Hashimoto M, Ikegami T, Seino S et al (2000) Expression and characterization of the chitin-binding domain of chitinase A1 from Bacillus circulans WL-12. J Bacteriol 182:3045–3054
Shoseyov O, Shani Z, Levy I (2006) Carbohydrate binding modules: biochemical properties and novel applications. Microbiol Mol Biol Rev 70:283–295
Nygaard S, Wendelbo R, Brown S (2002) Surface-specific zeolite-binding proteins. Adv Mater 14:1853–1856
Yang M, Choi BG, Park TJ et al (2011) Site-specific immobilization of gold binding polypeptide on gold nanoparticle-coated graphene sheet for biosensor application. Nanoscale 3:2950–2956
Care A, Bergquist PL, Sunna A (2015) Solid-binding peptides: smart tools for nanobiotechnology. Trends Biotechnol 33:259–268
Costa S, Almeida A, Castro A, Domingues L (2014) Fusion tags for protein solubility, purification, and immunogenicity in Escherichia coli: the novel Fh8 system. Front Microbiol 5:1–20
Kimple ME, Brill AL, Pasker RL (2013) Overview of affinity tags for protein purification. Curr Protoc Protein Sci 73:608–616
Godigamuwa K, Nakashima K, Okamoto J, Kawasaki S (2020) Biological route to fabricate silica on cellulose using immobilized silicatein fused with a carbohydrate-binding module. Biomacromol 21:2922–2928
Kobayashi H, Yoshida T, Inouye M (2009) Significant enhanced expression and solubility of human proteins in Escherichia coli by fusion with protein S from Myxococcus xanthus. Appl Environ Microbiol 75:5356–5362
Xu Y, Liu Q, Zhou L et al (2008) Surface display of GFP by Pseudomonas syringae truncated ice nucleation protein in attenuated Vibrio anguillarum strain. Mar Biotechnol 10:701–708
Ki MR, Yeo KB, Pack SP (2013) Surface immobilization of protein via biosilification catalyzed by silicatein fused to glutathione S-transferase (GST). Bioprocess Biosyst Eng 36:643–648
Schröder HC, Wang X, Manfrin A et al (2012) Acquisition of structure-guiding and structure-forming properties during maturation from the pro-silicatein to the silicatein form. J Biol Chem 287:22196–22205
Jungbauer A, Kaar W (2007) Current status of technical protein refolding. J Biotechnol 128:587–596
Schloßmacher U, Wiens M, Der Schrö HC et al (2011) Silintaphin-1—interaction with silicatein during structure-guiding bio-silica formation. FEBS J Authors J Compil 278:1145–1155
Compton SJ, Jones CG (1985) Mechanism of dye response and interference in the Bradford protein assay. Anal Biochem 151:369–374
Shimizu K, Cha J, Stucky GD, Morse DE (1998) Silicatein α: Cathepsin L-like protein in sponge biosilica. Proc Natl Acad Sci U S A 95:6234–6238
Yamaguchi H, Miyazaki M (2014) Refolding techniques for recovering biologically active recombinant proteins from inclusion bodies. Biomolecules 4:235–251
Kanarjov P, Reedo V, Acik IO et al (2008) Luminescent materials based on thin metal oxide films doped with rare earth ions. Phys Solid State 50:1727–1730
Mizutani T, Nagase H, Ogoshi H (1998) Silicic acid polymerization catalyzed by amines and polyamines. Chem Lett 27(2):133–134
Elkhooly TA, Müller WEG, Wang X et al (2014) Bioinspired self-assembly of tyrosinase-modified silicatein and fluorescent core-shell silica spheres. Bioinspir Biomim 9:044001
Müller WEG, Engel S, Wang X et al (2008) Bioencapsulation of living bacteria (Escherichia coli) with poly(silicate) after transformation with silicatein-α gene. Biomaterials 29:771–779
Jung HC, Park JH, Park SH et al (1998) Expression of carboxymethylcellulase on the surface of Escherichia coli using Pseudomonas syringae ice nucleation protein. Enzyme Microb Technol 22:348–354
Wang X, Schloßmacher U, Wiens M et al (2012) Silicateins, silicatein interactors and cellular interplay in sponge skeletogenesis: formation of glass fiber-like spicules. FEBS J 279:1721–1736
Oliveira C, Carvalho V, Domingues L, Gama FM (2015) Recombinant CBM-fusion technology—applications overview. Biotechnol Adv 33:358–369
Murr MM, Morse DE (2005) Fractal intermediates in the self-assembly of silicatein filaments. Proc Natl Acad Sci USA 102:11657–11662
Schröder HC, Grebenjuk VA, Wang X, Müller WEG (2016) Hierarchical architecture of sponge spicules: Biocatalytic and structure-directing activity of silicatein proteins as model for bioinspired applications. Bioinspir Biomim 11:041002
Garde J, Mu WEG (2016) Self-assembly and photocatalytic activity of branched silicatein / silintaphin filaments decorated with silicatein-synthesized TiO2 nanoparticles. Bioprocess Biosyst Eng 39:1477–1486
Belton DJ, Deschaume O, Perry CC (2012) An overview of the fundamentals of the chemistry of silica with relevance to biosilicification and technological advances. FEBS J 279:1710–1720
Kamathewatta NJB, Deay DO, Karaca BT et al (2020) Self-immobilized putrescine oxidase biocatalyst system engineered with a metal binding peptide. Langmuir 36:11908–11917
Togo Y, Nakashima K, Mwandira W, Kawasaki S (2020) A novel metal adsorbent composed of a hexa-histidine tag and a carbohydrate-binding module on cellulose. Anal Sci 36:459–464
Mwandira W, Nakashima K, Togo Y et al (2020) Cellulose-metallothionein biosorbent for removal of Pb(II) and Zn(II) from polluted water. Chemosphere 246:125733
Acknowledgments
This work is partly supported by Tonen General Sekiyu research foundation. K. Godigamuwa would like to thank the Uehara Memorial Foundation for their financial support. The authors thank Shuhei Shimoda (Technical Division, Institute for Catalysis, Hokkaido University) for his technical support in surface area measurement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Godigamuwa, K., Nakashima, K., Tsujitani, S. et al. Fabrication of silica on chitin in ambient conditions using silicatein fused with a chitin-binding domain. Bioprocess Biosyst Eng 44, 1883–1890 (2021). https://doi.org/10.1007/s00449-021-02568-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-021-02568-w