Abstract
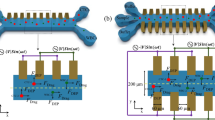
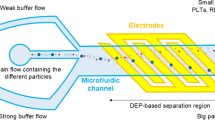
This study proposes a microfluidic device capable of separating monocytes from a type of cancer cell that is called T-cell acute lymphoblastic leukemia (RPMI-8402) in a continuous flow using negative and positive dielectrophoretic forces. The use of both the hydrodynamic and dielectrophoretic forces allows the separation of RPMI-8402 from monocytes based on differences in their intrinsic electrical properties and sizes. The specific crossover frequencies of monocytes and RPMI-8402 cells have been obtained experimentally. The optimum ranges of electrode pitch-to-channel height ratio at the cross sections with different electrode widths have been generally calculated by numerical simulations of the gradients of the electric field intensities and calculation their effective values (root-mean-square). In the device, the cell sorting has been conducted empirically, and then, the separation performance has been evaluated by analyzing the images before and after dielectrophoretic forces applied to the cells. In this work, the design of a chip with 77 μm gold–titanium electrode pitch was investigated to achieve high purity of monocytes of 95.2%. The proposed device can be used with relatively low applied voltages, as low as 16.5 V (peak to peak). Thus, the design can be used in biomedical diagnosis and chemical analysis applications as a lab-on-chip platform. Also, it can be used for the separation of biological cells such as bacteria, RNA, DNA, and blood cells.














Similar content being viewed by others
References
Whitesides GM (2006) The origins and the future of microfluidics. Nature 442(7101):368
Qian C, Huang H, Chen L, Li X, Ge Z, Chen T, Yang Z, Sun L (2014) Dielectrophoresis for bioparticle manipulation. Int J Mol Sci 15(10):18281–18309
Yan S, Tan SH, Li Y, Tang S, Teo AJ, Zhang J, Zhao Q, Yuan D, Sluyter R, Nguyen N-T (2018) A portable, hand-powered microfluidic device for sorting of biological particles. Microfluid Nanofluid 22(1):8
Bhagat AAS, Bow H, Hou HW, Tan SJ, Han J, Lim CT (2010) Microfluidics for cell separation. Med Biol Eng Comput 48(10):999–1014
Bagdi P, Bhardwaj P, Sen A (2012) Analysis and simulation of a micro hydrocyclone device for particle liquid separation. J Fluids Eng 134(2):021105
Yamada M, Seki M (2005) Hydrodynamic filtration for on-chip particle concentration and classification utilizing microfluidics. Lab Chip 5(11):1233–1239
Yamada M, Seki M (2006) Microfluidic particle sorter employing flow splitting and recombining. Anal Chem 78(4):1357–1362
Davis JA, Inglis DW, Morton KJ, Lawrence DA, Huang LR, Chou SY, Sturm JC, Austin RH (2006) Deterministic hydrodynamics: taking blood apart. Proc Natl Acad Sci 103(40):14779–14784
Hsu C-H, Di Carlo D, Chen C, Irimia D, Toner M (2008) Microvortex for focusing, guiding and sorting of particles. Lab Chip 8(12):2128–2134
Kuntaegowdanahalli SS, Bhagat AAS, Kumar G, Papautsky I (2009) Inertial microfluidics for continuous particle separation in spiral microchannels. Lab Chip 9:2973–2980
Takagi J, Yamada M, Yasuda M, Seki M (2005) Continuous particle separation in a microchannel having asymmetrically arranged multiple branches. Lab Chip 5(7):778–784
Ma Z, Collins DJ, Ai Y (2016) Detachable acoustofluidic system for particle separation via a traveling surface acoustic wave. Anal Chem 88(10):5316–5323
Huang S-B, Chen J, Wang J, Yang C-L, Wu M-H (2012) A new optically-induced dielectrophoretic (ODEP) force-based scheme for effective cell sorting. Int J Electrochem Sci 7:12656–12667
Inglis DW, Riehn R, Austin R, Sturm J (2004) Continuous microfluidic immunomagnetic cell separation. Appl Phys Lett 85(21):5093–5095
Rosales-Cruzaley E, Cota-Elizondo P, Sánchez D, Lapizco-Encinas BH (2013) Sperm cells manipulation employing dielectrophoresis. Bioprocess Biosyst Eng 36(10):1353–1362
Huang C, Smith JP, Saha TN, Rhim AD, Kirby BJ (2014) Characterization of microfluidic shear-dependent epithelial cell adhesion molecule immunocapture and enrichment of pancreatic cancer cells from blood cells with dielectrophoresis. Biomicrofluidics 8(4):044107
Alazzam A, Mathew B, Alhammadi F (2017) Novel microfluidic device for the continuous separation of cancer cells using dielectrophoresis. J Sep Sci 40(5):1193–1200
Pohl HA (1951) The motion and precipitation of suspensoids in divergent electric fields. J Appl Phys 22(7):869–871
Hu X, Bessette PH, Qian J, Meinhart CD, Daugherty PS, Soh HT (2005) Marker-specific sorting of rare cells using dielectrophoresis. Proc Natl Acad Sci 102(44):15757–15761
Pommer MS, Zhang Y, Keerthi N, Chen D, Thomson JA, Meinhart CD, Soh HT (2008) Dielectrophoretic separation of platelets from diluted whole blood in microfluidic channels. Electrophoresis 29(6):1213–1218
Piacentini N, Mernier G, Tornay R, Renaud P (2011) Separation of platelets from other blood cells in continuous-flow by dielectrophoresis field-flow-fractionation. Biomicrofluidics 5(3):034122
Alshareef M, Metrakos N, Juarez Perez E, Azer F, Yang F, Yang X, Wang G (2013) Separation of tumor cells with dielectrophoresis-based microfluidic chip. Biomicrofluidics 7(1):011803
Buyong MR, Larki F, Faiz MS, Hamzah AA, Yunas J, Majlis BY (2015) A tapered aluminium microelectrode array for improvement of dielectrophoresis-based particle manipulation. Sensors 15(5):10973–10990
Tedjo W, Nejad JE, Feeny R, Yang L, Henry CS, Tobet S, Chen T (2018) Electrochemical biosensor system using a CMOS microelectrode array provides high spatially and temporally resolved images. Biosens Bioelectron 114:78–88
Puydinger dos Santos MV, Béron F, Pirota KR, Diniz JA, Moshkalev S (2017) Electrical manipulation of a single nanowire by dielectrophoresis. In: Nanowires-new insights. InTech, Rijeka, Croatia, pp 41–58
Kale A, Patel S, Xuan X (2018) Three-dimensional reservoir-based dielectrophoresis (rDEP) for enhanced particle enrichment. Micromachines 9(3):123
Hirota Y, Hakoda M, Wakizaka Y (2010) Separation characteristics of animal cells using a dielectrophoretic filter. Bioprocess Biosyst Eng 33(5):607–612
Pethig RR (2017) Dielectrophoresis: theory, methodology and biological applications. Wiley, USA
Gascoyne PR, Shim S, Noshari J, Becker FF, Stemke-Hale K (2013) Correlations between the dielectric properties and exterior morphology of cells revealed by dielectrophoretic field-flow fractionation. Electrophoresis 34(7):1042–1050
Elitas M, Dhar N, Schneider K, Valero A, Braschler T, McKinney J, Renaud P (2017) Dielectrophoresis as a single cell characterization method for bacteria. Biomed Phys Eng Express 3(1):015005
Jones TB, Jones TB (2005) Electromechanics of particles. Cambridge University Press, Cambridge
Yang J, Huang Y, Wang X, Wang X-B, Becker FF, Gascoyne PR (1999) Dielectric properties of human leukocyte subpopulations determined by electrorotation as a cell separation criterion. Biophys J 76(6):3307–3314
Han S-I, Joo Y-D, Han K-H (2013) An electrorotation technique for measuring the dielectric properties of cells with simultaneous use of negative quadrupolar dielectrophoresis and electrorotation. Analyst 138(5):1529–1537
Kumar V, Gill KD (2018) Blood collection and preservation. Basic concepts in clinical biochemistry: a practical guide. Springer, Berlin, pp 5–7
Acknowledgements
This research was supported by the Tarbiat Modares University (TMU) Faculty of Mechanical Engineering and Faculty of Electrical & Computer Engineering. Also, we are thankful of Hematology Laboratory for access to the cleanroom facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest.
Ethical approval
Blood samples were collected from consenting donors, following a procedure approved by the Ethics Committee of the Tarbiat Modares University (TMU).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zahedi Siani, O., Zabetian Targhi, M., Sojoodi, M. et al. Dielectrophoretic separation of monocytes from cancer cells in a microfluidic chip using electrode pitch optimization. Bioprocess Biosyst Eng 43, 1573–1586 (2020). https://doi.org/10.1007/s00449-020-02349-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-020-02349-x