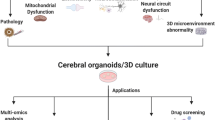
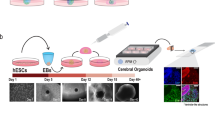
Abstract
Neurodegenerative diseases (NDs) are characterized by uncontrolled loss of neuronal cells leading to a progressive deterioration of brain functions. The transition rate of numerous neuroprotective drugs against Alzheimer’s disease, Parkinson’s disease, amyotrophic lateral sclerosis, and Huntington’s disease, leading to FDA approval, is only 8–14% in the last two decades. Thus, in spite of encouraging preclinical results, these drugs have failed in human clinical trials, demonstrating that traditional cell cultures and animal models cannot accurately replicate human pathophysiology. Hence, in vitro three-dimensional (3D) models have been developed to bridge the gap between human and animal studies. Such technological advancements in 3D culture systems, such as human-induced pluripotent stem cell (iPSC)-derived cells/organoids, organ-on-a-chip technique, and 3D bioprinting, have aided our understanding of the pathophysiology and underlying mechanisms of human NDs. Despite these recent advances, we still lack a 3D model that recapitulates all the key aspects of NDs, thus making it difficult to study the ND’s etiology in-depth. Hence in this review, we propose developing a combinatorial approach that allows the integration of patient-derived iPSCs/organoids with 3D bioprinting and organ-on-a-chip technique as it would encompass the neuronal cells along with their niche. Such a 3D combinatorial approach would characterize pathological processes thoroughly, making them better suited for high-throughput drug screening and developing effective novel therapeutics targeting NDs.



Similar content being viewed by others
Data availability
Not applicable
References
Abuirmeileh AN, Alzoubi KH, Rababa’h AM (2020) The effect of levosimendan on two distinct rodent models of Parkinson’s disease. Curr Alzheimer Res 17(11):1043–1051. https://doi.org/10.2174/1567205017666201218102724
Aguilar MA, Rodriguez-Arias M, Marí-Sanmillán MI, Miñarro J (1997) Effects of risperidone on conditioned avoidance responding in male mice. Behav Pharmacol 8(8):669–676. https://doi.org/10.1097/00008877-199712000-00001
Ahmad S, Khan A, Ali W, Jo MH, Park J, Ikram M, Kim MO (2021) Fisetin rescues the mice brains against D-galactose-induced oxidative stress, neuroinflammation and memory impairment. Front Pharmacol 12:612078–612078. https://doi.org/10.3389/FPHAR.2021.612078
Ahmed RM, Irish M, van Eersel J, Ittner A, Ke YD, Volkerling A, van der Hoven J, Tanaka K, Karl T, Kassiou M, Kril JJ, Piguet O, Götz J, Kiernan MC, Halliday GM, Hodges JR, Ittner LM (2017) Mouse models of frontotemporal dementia: a comparison of phenotypes with clinical symptomatology. Neurosci Biobehav Rev 74(Pt A):126–138. https://doi.org/10.1016/J.NEUBIOREV.2017.01.004
Akamatsu M, Yamashita T, Hirose N, Teramoto S, Kwak S (2016) The AMPA receptor antagonist perampanel robustly rescues amyotrophic lateral sclerosis (ALS) pathology in sporadic ALS model mice OPEN. https://doi.org/10.1038/srep28649
Allen PJ, Feigin A (2014) Gene-based therapies in Parkinson’s disease. Neurother : J Am Soc Exp NeuroTher 11(1):60–67. https://doi.org/10.1007/S13311-013-0233-2
Anderson RM, Hadjichrysanthou C, Evans S, Wong MM (2017) Why do so many clinical trials of therapies for Alzheimer’s disease fail? Lancet (london, England) 390(10110):2327–2329. https://doi.org/10.1016/S0140-6736(17)32399-1
Armstrong MJ, Okun MS (2020) Diagnosis and treatment of Parkinson disease: a review. JAMA 323(6):548–560. https://doi.org/10.1001/JAMA.2019.22360
Ayalon G, Lee SH, Adolfsson O, Foo-Atkins C, Atwal JK, Blendstrup M, Booler H, Bravo J, Brendza R, Brunstein F, Chan R, Chandra P, Couch JA, Datwani A, Demeule B, DiCara D, Erickson R, Ernst JA, Foreman O, … Kerchner GA (2021) Antibody semorinemab reduces tau pathology in a transgenic mouse model and engages tau in patients with Alzheimer’s disease. Sci Transl Med 13(593). https://doi.org/10.1126/SCITRANSLMED.ABB2639
Balon K, and Wiatrak B (2021) PC12 and THP-1 cell lines as neuronal and microglia model in neurobiological research. Appl Sci 11(9):3729. https://doi.org/10.3390/APP11093729
Berman T, Bayati A (2018) What are neurodegenerative diseases and how do they affect the brain? Frontiers for Young Minds. https://doi.org/10.3389/frym.2018.00070
Boland T, Mironov V, Gutowska A, Roth EA, Markwald RR (2003) Cell and organ printing 2: fusion of cell aggregates in three-dimensional gels. Anat Rec A Discov Mol Cell Evol Biol 272A(2):497–502. https://doi.org/10.1002/AR.A.10059
Bourque M, Morissette M, Conquet F, Charvin D, Di Paolo T (2023) Foliglurax a positive allosteric modulator of the metabotrophic glutamate receptor 4 protects dopaminergic neurons in MPTP-lesioned male mice. Brain Res 148349. https://doi.org/10.1016/J.BRAINRES.2023.148349
Brassard JA, Nikolaev M, Hübscher T, Hofer M, and Lutolf MP (2020) Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nature Mater 20(1):22–29. https://doi.org/10.1038/s41563-020-00803-5
Brown RC, Lockwood AH, Sonawane BR (2005) Neurodegenerative diseases: an overview of environmental risk factors. Environ Health Perspect 113(9):1250–1256. https://doi.org/10.1289/EHP.7567
Burkhardt MF, Martinez FJ, Wright S, Ramos C, Volfson D, Mason M, Garnes J, Dang V, Lievers J, Shoukat-Mumtaz U, Martinez R, Gai H, Blake R, Vaisberg E, Grskovic M, Johnson C, Irion S, Bright J, Cooper B, Javaherian A (2013) A cellular model for sporadic ALS using patient-derived induced pluripotent stem cells. Mol Cell Neurosci 56:355–364. https://doi.org/10.1016/J.MCN.2013.07.007
Chen SS, Yang C, Hao F, Li C, Lu T, Zhao LR, Duan WM (2014) Intrastriatal GDNF gene transfer by inducible lentivirus vectors protects dopaminergic neurons in a rat model of parkinsonism. Exp Neurol 261:87–96. https://doi.org/10.1016/J.EXPNEUROL.2014.06.022
Cope MB, Li X, Jumbo-Lucioni P, DiCostanzo CA, Jamison WG, Kesterson RA, Allison DB, Nagy TR (2009) Risperidone alters food intake core body temperature and locomotor activity in mice. Physiol Behav 96(3):457–463. https://doi.org/10.1016/J.PHYSBEH.2008.11.011
Cordero-Llana Ó, Houghton BC, Rinaldi F, Taylor H, Yáñez-Muñoz RJ, Uney JB, Wong LF, Caldwell MA (2015) Enhanced efficacy of the CDNF/MANF family by combined intranigral overexpression in the 6-OHDA rat model of Parkinson’s disease. Mol Ther : J Am Soc Gene Ther 23(2):244–254. https://doi.org/10.1038/MT.2014.206
Crouch B, Yeap JM, Pais B, Riedel G, Platt B (2019) Of mice and motion: Behavioural-EEG phenotyping of Alzheimer’s disease mouse models. J Neurosci Methods 319:89–98. https://doi.org/10.1016/J.JNEUMETH.2018.06.028
Csöbönyeiová M, Polák Š, Danišovič L (2016) Toxicity testing and drug screening using iPSC-derived hepatocytes, cardiomyocytes, and neural cells. Can J Physiol Pharmacol 94(7):687–694. https://doi.org/10.1139/CJPP-2015-0459
Cui X, Zuo P, Zhang Q, Li X, Hu Y, Long J, Packer L, Liu J (2006) Chronic systemic D-galactose exposure induces memory loss, neurodegeneration, and oxidative damage in mice: protective effects of R-alpha-lipoic acid. J Neurosci Res 84(3):647–654. https://doi.org/10.1002/JNR.20899
Cummings JL, Morstorf T, and Zhong K (2014. Alzheimer’s disease drug-development pipeline: few candidates, frequent failures. Alzheimer’s Res and Ther 6(4). https://doi.org/10.1186/ALZRT269
Curzer HJ, Perry G, Wallace MC, Perry D (2016) The three Rs of animal research: what they mean for the institutional animal care and use committee and why. Sci Eng Ethics 22(2):549–565. https://doi.org/10.1007/S11948-015-9659-8
Dawson TM, Golde TE, Lagier-Tourenne C (2018) Animal models of neurodegenerative diseases. Nat Neurosci 21(10):1370–1379. https://doi.org/10.1038/S41593-018-0236-8
Devos D, Lebouvier T, Lardeux B, Biraud M, Rouaud T, Pouclet H, Coron E, Bruley des Varannes S, Naveilhan P, Nguyen JM, Neunlist M, and Derkinderen P (2013) Colonic inflammation in Parkinson’s disease. Neurobiol Dis 50(1):42–48. https://doi.org/10.1016/J.NBD.2012.09.007
Durães F, Pinto M, Sousa E (2018) Old drugs as new treatments for neurodegenerative diseases. In Pharmaceuticals. https://doi.org/10.3390/ph11020044
Dutta S, Sengupta P (2016) Men and mice: relating their ages. Life Sci 152:244–248. https://doi.org/10.1016/J.LFS.2015.10.025
Fitzgerald KA, Malhotra M, Curtin CM, O’Brien FJ, O’Driscoll CM (2015) Life in 3D is never flat: 3D models to optimise drug delivery. JCR : Official J Controlled Release Soc 215:39–54. https://doi.org/10.1016/J.JCONREL.2015.07.020
Franzen N, van Harten WH, Retèl VP, Loskill P, van den Eijnden-van Raaij J, IJzerman, M. (2019) Impact of organ-on-a-chip technology on pharmaceutical R&D costs. Drug Discov Today 24(9):1720–1724. https://doi.org/10.1016/J.DRUDIS.2019.06.003
Gitler AD, Dhillon P, Shorter J (2017) Neurodegenerative disease: models, mechanisms, and a new hope. Dis Model Mech 10(5):499–502. https://doi.org/10.1242/DMM.030205
Gonzalez-Barrios JA, Lindahl M, Bannon MJ, Anaya-Martínez V, Flores G, Navarro-Quiroga I, Trudeau LE, Aceves J, Martinez-Arguelles DB, Garcia-Villegas R, Jiménez I, Segovia J, Martinez-Fong D (2006) Neurotensin polyplex as an efficient carrier for delivering the human GDNF gene into nigral dopamine neurons of hemiparkinsonian rats. Mol Ther : J Am Soc Gene Ther 14(6):857–865. https://doi.org/10.1016/J.YMTHE.2006.09.001
Gordon K, Clouaire T, Bao XX, Kemp SE, Xenophontos M, de Las Heras JI, Stancheva I (2014) Immortality, but not oncogenic transformation, of primary human cells leads to epigenetic reprogramming of DNA methylation and gene expression. Nucleic Acids Res 42(6):3529. https://doi.org/10.1093/NAR/GKT1351
Gourie-Devi M (2018) Relevance of neuroepidemiology: burden of neurological disorders and public health issues. Annal Ind Acad Neurol. https://doi.org/10.4103/aian.AIAN-428-18
Grant LM, Kelm-Nelson CA, Hilby BL, Blue KV, Paul Rajamanickam ES, Pultorak JD, Fleming SM, Ciucci MR (2015) Evidence for early and progressive ultrasonic vocalization and oromotor deficits in a PINK1 gene knockout rat model of Parkinson’s disease. J Neurosci Res 93(11):1713–1727. https://doi.org/10.1002/JNR.23625
Grima JC, Daigle JG, Arbez N, Cunningham KC, Zhang K, Ochaba J, Geater C, Morozko E, Stocksdale J, Glatzer JC, Pham JT, Ahmed I, Peng Q, Wadhwa H, Pletnikova O, Troncoso JC, Duan W, Snyder SH, Ranum LPW, Rothstein JD (2017) Mutant Huntingtin disrupts the nuclear pore complex. Neuron 94(1):93–107.e6. https://doi.org/10.1016/J.NEURON.2017.03.023
Gross SK, Shim BS, Bartus RT, Deaver D, McEachin Z, Bétourné A, Boulis NM, and Maragakis NJ (2020) Focal and dose-dependent neuroprotection in ALS mice following AAV2-neurturin delivery. Exp Neurol 323. https://doi.org/10.1016/J.EXPNEUROL.2019.113091
Hashimoto M, Watanabe K, Miyoshi K, Koyanagi Y, Tadano J, Miyawaki I (2021) Multiplatform metabolomic analysis of the R6/2 mouse model of Huntington’s disease. FEBS Open Bio 11(10):2807–2818. https://doi.org/10.1002/2211-5463.13285
Hebb ALO, Robertson HA, Denovan-Wright EM (2004) Striatal phosphodiesterase mRNA and protein levels are reduced in Huntington’s disease transgenic mice prior to the onset of motor symptoms. Neuroscience 123(4):967–981. https://doi.org/10.1016/j.neuroscience.2003.11.009
Herrán E, Requejo C, Ruiz-Ortega JA, Aristieta A, Igartua M, Bengoetxea H, Ugedo L, Pedraz JL, Lafuente JV, Hernández RM (2014) Increased antiparkinson efficacy of the combined administration of VEGF- and GDNF-loaded nanospheres in a partial lesion model of Parkinson’s disease. Int J Nanomed 9(1):2677–2687. https://doi.org/10.2147/IJN.S61940
Israel MA, Yuan SH, Bardy C, Reyna SM, Mu Y, Herrera C, Hefferan MP, Van Gorp S, Nazor KL, Boscolo FS, Carson CT, Laurent LC, Marsala M, Gage FH, Remes AM, Koo EH, Goldstein LSB (2012) Probing sporadic and familial Alzheimer’s disease using induced pluripotent stem cells. Nature 482(7384):216–220. https://doi.org/10.1038/NATURE10821
Jankowsky JL, and Zheng H (2017) Practical considerations for choosing a mouse model of Alzheimer’s disease. Mol Neurodegener 12(1). https://doi.org/10.1186/S13024-017-0231-7
Jeon S, Lee SH, Ahmed SB, Han J, Heo SJ, Kang HW (2021) 3D cell aggregate printing technology and its applications. Essays Biochem 65(3):467–480. https://doi.org/10.1042/EBC20200128
Jucker M (2010) The benefits and limitations of animal models for translational research in neurodegenerative diseases. Nature Med 16(11):1210–1214. https://doi.org/10.1038/nm.2224
Van Kampen JM, Baranowski DC, Robertson HA, Shaw CA, Kay DG, and Lewis P (2015) The progressive BSSG rat model of Parkinson’s: recapitulating multiple key features of the human disease. PloS One 10(10). https://doi.org/10.1371/JOURNAL.PONE.0139694
Kasper C, Egger D, Senatov F, Raghunath M, Chariyev-Prinz F, Louit A, Galbraith T, and Berthod F (2023) In vitro 3D modeling of neurodegenerative diseases. Bioeng 10(1):93. https://doi.org/10.3390/BIOENGINEERING10010093
Kaur G, Dufour JM (2012) Cell lines: valuable tools or useless artifacts. Spermatogenesis 2(1):1–5. https://doi.org/10.4161/SPMG.19885
Kehoe EG, McNulty JP, Mullins PG, Bokde ALW (2014) Advances in MRI biomarkers for the diagnosis of Alzheimer’s disease. Biomark Med 8(9):1151–1169. https://doi.org/10.2217/BMM.14.42
Komuro Y, Xu G, Bhaskar K, Lamb BT (2015) Human tau expression reduces adult neurogenesis in a mouse model of tauopathy. Neurobiol Aging 36(6):2034–2042. https://doi.org/10.1016/J.NEUROBIOLAGING.2015.03.002
Kosel F, Hamilton JS, Harrison SL, Godin V, Franklin TB (2021) Reduced social investigation and increased injurious behavior in transgenic 5xFAD mice. J Neurosci Res 99(1):209–222. https://doi.org/10.1002/JNR.24578
Kulkarni AD, Vanjari YH, Sancheti KH, Belgamwar VS, Surana SJ, Pardeshi CV (2015) Nanotechnology-mediated nose to brain drug delivery for Parkinson’s disease: a mini review. J Drug Target 23(9):775–788. https://doi.org/10.3109/1061186X.2015.1020809
Kuttner-Hirshler Y, Venkatasubramanian PN, Apolinario J, Bonds J, Wyrwicz AM, Lazarov O (2017) Brain biomarkers in familial Alzheimer’s disease mouse models. J Alzheimer’s Dis: JAD 60(3):949–958. https://doi.org/10.3233/JAD-170269
Kwon D (2022) Guardians of the brain: how a special immune system protects our grey matter. Nature 606(7912):22–24. https://doi.org/10.1038/D41586-022-01502-8
Lee D, Kim N, Jeon SH, Gee MS, Ju YJ, Jung MJ, Cho JS, Lee Y, Lee S, Lee JK (2022) Hesperidin improves memory function by enhancing neurogenesis in a mouse model of Alzheimer’s disease. Nutrients 14(15):3125. https://doi.org/10.3390/NU14153125/S1
Lee DJ, Milosevic L, Gramer R, Sasikumar S, Al-Ozzi TM, De Vloo P, Dallapiazza RF, Elias GJB, Cohn M, Kalia SK, Hutchison WD, Fasano A, Lozano AM (2019) Nucleus basalis of Meynert neuronal activity in Parkinson’s disease. J Neurosurg 132(2):574–582. https://doi.org/10.3171/2018.11.JNS182386
Lee KH, Lee DW, and Kang BC (2020) The “R” principles in laboratory animal experiments. Lab Animal Res 36(1). https://doi.org/10.1186/S42826-020-00078-6
Lee W, Pinckney J, Lee V, Lee JH, Fischer K, Polio S, Park JK, Yoo SS (2009) Three-dimensional bioprinting of rat embryonic neural cells. NeuroReport 20(8):798–803. https://doi.org/10.1097/WNR.0B013E32832B8BE4
Lee YB, Polio S, Lee W, Dai G, Menon L, Carroll RS, Yoo SS (2010) Bio-printing of collagen and VEGF-releasing fibrin gel scaffolds for neural stem cell culture. Exp Neurol 223(2):645–652. https://doi.org/10.1016/J.EXPNEUROL.2010.02.014
Lin X, Parisiadou L, Sgobio C, Liu G, Yu J, Sun L, Shim H, Gu XL, Luo J, Long CX, Ding J, Mateo Y, Sullivan PH, Wu LG, Goldstein DS, Lovinger D, Cai H (2012) Conditional expression of Parkinson’s disease-related mutant α-synuclein in the midbrain dopaminergic neurons causes progressive neurodegeneration and degradation of transcription factor nuclear receptor related 1. J Neurosci 32(27):9248–9264. https://doi.org/10.1523/JNEUROSCI.1731-12.2012
Low LA, and Tagle DA (2017) Organs-on-chips: progress, challenges, and future directions. Exp Biol Med 242(16):1573–1578. https://doi.org/10.1177/1535370217700523
Lozano R, Stevens L, Thompson BC, Gilmore KJ, Gorkin R, Stewart EM, in het Panhuis, M., Romero-Ortega, M., & Wallace, G. G. (2015) 3D printing of layered brain-like structures using peptide modified gellan gum substrates. Biomaterials 67:264–273. https://doi.org/10.1016/J.BIOMATERIALS.2015.07.022
Lüesse HG, Schiefer J, Spruenken A, Puls C, Block F, Kosinski CM (2001) Evaluation of R6/2 HD transgenic mice for therapeutic studies in Huntington’s disease: behavioral testing and impact of diabetes mellitus. Behav Brain Res 126(1–2):185–195. https://doi.org/10.1016/S0166-4328(01)00261-3
Lutz C (2018) Mouse models of ALS: past, present and future. Brain Res 1693(Pt A):1–10. https://doi.org/10.1016/J.BRAINRES.2018.03.024
Martinez-Fong D, Bannon MJ, Trudeau LE, Gonzalez-Barrios JA, Arango-Rodriguez ML, Hernandez-Chan NG, Reyes-Corona D, Armendáriz-Borunda J, Navarro-Quiroga I (2012) NTS-polyplex: a potential nanocarrier for neurotrophic therapy of Parkinson’s disease. Nanomed Nanotechnol Biol Med 8(7):1052–1069. https://doi.org/10.1016/J.NANO.2012.02.009
Mehndiratta MM, Aggarwal V (2021) Neurological disorders in India: past, present, and next steps. Lancet Glob Health 9(8):e1043–e1044. https://doi.org/10.1016/S2214-109X(21)00214-X
Menalled LB, Patry M, Ragland N, Lowden PAS, Goodman J, Minnich J, Zahasky B, Park L, Leeds J, Howland D, Signer E, Tobin AJ, Brunner D (2010) Comprehensive behavioral testing in the R6/2 mouse model of Huntington’s disease shows no benefit from CoQ10 or minocycline. PLOS ONE 5(3):e9793. https://doi.org/10.1371/JOURNAL.PONE.0009793
Menalled LB, Sison JD, Dragatsis I, Zeitlin S, Chesselet MF (2003) Time course of early motor and neuropathological anomalies in a knock-in mouse model of Huntington’s disease with 140 CAG repeats. J Comp Neurol 465(1):11–26. https://doi.org/10.1002/CNE.10776
Mustapha M, and Taib CNM (2021) MPTP-induced mouse model of Parkinson’s disease: a promising direction of therapeutic strategies. Bosnian J Basic Med Sci 21(4):422–433. https://doi.org/10.17305/BJBMS.2020.5181
Nadella R, Voutilainen MH, Saarma M, Gonzalez-Barrios JA, Leon-Chavez BA, Jimnez, J DM, Jimnez SDH, Escobedo L, and Martinez-Fong D (2014) Transient transfection of human CDNF gene reduces the 6-hydroxydopamine-induced neuroinflammation in the rat substantia nigra. J Neuroinflammation 11(1). https://doi.org/10.1186/S12974-014-0209-0
Nagai K (2012) Bovine milk phospholipid fraction protects Neuro2a cells from endoplasmic reticulum stress via PKC activation and autophagy. J Biosci Bioeng 114(4):466–471. https://doi.org/10.1016/J.JBIOSC.2012.05.009
Nazari QA, Kume T, Takada-Takatori Y, Izumi Y, Akaike A (2013) Protective effect of luteolin on an oxidative-stress model induced by microinjection of sodium nitroprusside in mice. J Pharmacol Sci 122(2):109–117. https://doi.org/10.1254/JPHS.13019FP
Nazari QA, Mizuno K, Kume T, Takada-Takatori Y, Izumi Y, Akaike A (2012) In vivo brain oxidative stress model induced by microinjection of sodium nitroprusside in mice. J Pharmacol Sci 120(2):105–111. https://doi.org/10.1254/JPHS.12143FP
Nicklas WJ, Youngster SK, Kindt MV, Heikkila RE (1987) MPTP, MPP+ and mitochondrial function. Life Sci 40(8):721–729. https://doi.org/10.1016/0024-3205(87)90299-2
Niu M, Zhao F, Bondelid K, Siedlak S L, Torres S, Fujioka H, Wang W, Liu J, and Zhu X (2021) VPS35 D620N knockin mice recapitulate cardinal features of Parkinson’s disease. Aging Cell 20(5):e13347. https://doi.org/10.1111/ACEL.13347
Oddo S, Caccamo A, Shepherd JD, Murphy MP, Golde TE, Kayed R, Metherate R, Mattson MP, Akbari Y, LaFerla FM (2003) Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Aβ and synaptic dysfunction. Neuron 39(3):409–421. https://doi.org/10.1016/S0896-6273(03)00434-3
Olanow C W, Kieburtz K, and Schapira AHV (2008) Why have we failed to achieve neuroprotection in Parkinson’s disease? Annals Neurol 64(SUPPL. 2). https://doi.org/10.1002/ANA.21461
Osaki T, Uzel SGM, and Kamm RD (2018) Microphysiological 3D model of amyotrophic lateral sclerosis (ALS) from human iPS-derived muscle cells and optogenetic motor neurons. Sci Adv 4(10). https://doi.org/10.1126/SCIADV.AAT5847
Paldino E, Balducci C, La Vitola P, Artioli L, D’Angelo V, Giampà C, Artuso V, Forloni G, Fusco FR (2020) Neuroprotective effects of doxycycline in the R6/2 mouse model of Huntington’s disease. Mol Neurobiol 57(4):1889–1903. https://doi.org/10.1007/S12035-019-01847-8
Pardo J, Morel G, Astiz M, Schwerdt J, Leon M, Rodriguez S, Herenu C, Goya R (2014) Gene therapy and cell reprogramming for the aging brain: achievements and promise. Curr Gene Ther 14(1):24–34. https://doi.org/10.2174/1566523214666140120121733
Park IH, Arora N, Huo H, Maherali N, Ahfeldt T, Shimamura A, Lensch MW, Cowan C, Hochedlinger K, Daley GQ (2008a) Disease-specific induced pluripotent stem cells. Cell 134(5):877–886. https://doi.org/10.1016/J.CELL.2008.07.041
Park IH, Zhao R, West JA, Yabuuchi A, Huo H, Ince TA, Lerou PH, Lensch MW, Daley GQ (2008b) Reprogramming of human somatic cells to pluripotency with defined factors. Nature 451(7175):141–146. https://doi.org/10.1038/nature06534
Park J, Wetzel I, Marriott I, Dréau D, D’Avanzo C, Kim DY, Tanzi RE, and Cho H (2018) A 3D human triculture system modeling neurodegeneration and neuroinflammation in Alzheimer’s disease. Nature Neurosci 21(7):941–951. https://doi.org/10.1038/s41593-018-0175-4
Perlman RL (2016) Mouse models of human disease: an evolutionary perspective. Evolution, Medicine, and Public Health 2016(1):170. https://doi.org/10.1093/EMPH/EOW014
Pfeuffer S, Ruck T, Kleinschnitz C, Wiendl H, Meuth SG (2016) Failed, interrupted and inconclusive trials on relapsing multiple sclerosis treatment: update 2010–2015. Expert Rev Neurother 16(6):689–700. https://doi.org/10.1080/14737175.2016.1176531
Razgado-Hernandez LF, Espadas-Alvarez AJ, Reyna-Velazquez P, Sierra-Sanchez A, Anaya-Martinez V, Jimenez-Estrada I, Bannon MJ, Martinez-Fong D, and Aceves-Ruiz J (2015) The transfection of BDNF to dopamine neurons potentiates the effect of dopamine D3 receptor agonist recovering the striatal innervation, dendritic spines and motor behavior in an aged rat model of Parkinson’s disease. PloS One 10(2). https://doi.org/10.1371/JOURNAL.PONE.0117391
Renner M, Lancaster MA, Bian S, Choi H, Ku T, Peer A, Chung K, and Knoblich JA (2017) Self-organized developmental patterning and differentiation in cerebral organoids. The EMBO J 36(10):1316–1329. https://doi.org/10.15252/EMBJ.201694700
Ryskamp D, Wu J, Geva M, Kusko R, Grossman I, Hayden M, Bezprozvanny I (2017) The sigma-1 receptor mediates the beneficial effects of pridopidine in a mouse model of Huntington disease. Neurobiol Dis 97(Pt A):46–59. https://doi.org/10.1016/J.NBD.2016.10.006
Sampson TR, Challis C, Jain N, Moiseyenko A, Ladinsky MS, Shastri GG, Thron T, Needham BD, Horvath I, Debelius JW, Janssen S, Knight R, Wittung-Stafshede P, Gradinaru V, Chapman M, Mazmanian SK (2020) A gut bacterial amyloid promotes α-synuclein aggregation and motor impairment in mice. ELife 9. https://doi.org/10.7554/ELIFE.53111
Schneider LS, Mangialasche F, Andreasen N, Feldman H, Giacobini E, Jones R, Mantua V, Mecocci P, Pani L, Winblad B, Kivipelto M (2014) Clinical trials and late-stage drug development for Alzheimer’s disease: an appraisal from 1984 to 2014. J Intern Med 275(3):251–283. https://doi.org/10.1111/JOIM.12191
Semple BD, Blomgren K, Gimlin K, Ferriero DM, Noble-Haeusslein LJ (2013) Brain development in rodents and humans: identifying benchmarks of maturation and vulnerability to injury across species. Prog Neurobiol 106–107:1–16. https://doi.org/10.1016/J.PNEUROBIO.2013.04.001
Sheikh MA, Malik YS, Xing Z, Guo Z, Tian H, Zhu X, Chen X (2017) Polylysine-modified polyethylenimine (PEI-PLL) mediated VEGF gene delivery protects dopaminergic neurons in cell culture and in rat models of Parkinson’s disease (PD). Acta Biomater 54:58–68. https://doi.org/10.1016/J.ACTBIO.2016.12.048
Sheikh S, Ejazul H, Mir SS (2013) Neurodegenerative diseases: multifactorial conformational diseases and their therapeutic interventions. J Neurodegener Dis 2013:1–8. https://doi.org/10.1155/2013/563481
Shimoke K, Sasaya H, and Ikeuchi T (2011) Analysis of the role of nerve growth factor in promoting cell survival during endoplasmic reticulum stress in PC12 cells. Meth Enzymol 490(C)53–70. https://doi.org/10.1016/B978-0-12-385114-7.00003-9
Shin Y, Choi SH, Kim E, Bylykbashi E, Kim JA, Chung S, Kim DY, Kamm RD, Tanzi RE (2019) Blood–brain barrier dysfunction in a 3d in vitro model of Alzheimer’s disease. Adv Sci 6(20):1900962. https://doi.org/10.1002/ADVS.201900962
Skardal A, Atala A (2015) Biomaterials for integration with 3-D bioprinting. Ann Biomed Eng 43(3):730–746. https://doi.org/10.1007/S10439-014-1207-1
Slanzi A, Iannoto G, Rossi B, Zenaro E, Constantin G (2020) In vitro models of neurodegenerative diseases. Front Cell Dev Biol 8:328. https://doi.org/10.3389/FCELL.2020.00328/BIBTEX
Son MY, Sim H, Son YS, Jung KB, Lee MO, Oh JH, Chung SK, Jung CR, Kim J (2017) Distinctive genomic signature of neural and intestinal organoids from familial Parkinson’s disease patient-derived induced pluripotent stem cells. Neuropathol Appl Neurobiol 43(7):584–603. https://doi.org/10.1111/NAN.12396
Song L, Yuan X, Jones Z, Griffin K, Zhou Y, Ma T, and Li Y (2019) Assembly of human stem cell-derived cortical spheroids and vascular spheroids to model 3-D brain-like tissues. Sci Rep 9(1). https://doi.org/10.1038/S41598-019-42439-9
Sullivan MA, Lane SD, Volkerling A, Engel M, Werry EL, Kassiou M, and Werry E (2022) 3D bioprinting of stem cell-derived central nervous system cells enables astrocyte growth, vasculogenesis and enhances neural differentiation/function. BioRxiv 11(13):516338. https://doi.org/10.1101/2022.11.13.516338
Swarup V, Phaneuf D, Bareil C, Robertson J, Rouleau GA, Kriz J, Julien JP (2011) Pathological hallmarks of amyotrophic lateral sclerosis/frontotemporal lobar degeneration in transgenic mice produced with TDP-43 genomic fragments. Brain J Neurol 134(Pt 9):2610–2626. https://doi.org/10.1093/BRAIN/AWR159
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131(5):861–872. https://doi.org/10.1016/J.CELL.2007.11.019
Tan EK, Srivastava AK, Arnold WD, Singh MP, Zhang Y (2015) Neurodegeneration: etiologies and new therapies. BioMed Res Int. https://doi.org/10.1155/2015/272630
Tang M, Rich JN, and Chen S (2021) Biomaterials and 3D bioprinting strategies to model glioblastoma and the blood-brain barrier. Adv Mater 33(5). https://doi.org/10.1002/ADMA.202004776
Tankersley CG, Haenggeli C, Rothstein JD (2007) Respiratory impairment in a mouse model of amyotrophic lateral sclerosis. J Appl Physiol (Bethesda Md.: 1985) 102(3):926–932. https://doi.org/10.1152/JAPPLPHYSIOL.00193.2006
Telesford QK, Simpson SL, Burdette JH, Hayasaka S, Laurienti PJ (2011) The brain as a complex system: using network science as a tool for understanding the brain. Brain Connect 1(4):295–308. https://doi.org/10.1089/BRAIN.2011.0055
Teli, P., Kale, V., & Vaidya, A. (2022). Mesenchymal stromal cells-derived secretome protects Neuro-2a cells from oxidative stress-induced loss of neurogenesis. Exp Neurol 354:114107. https://doi.org/10.1016/J.EXPNEUROL.2022.114107
Teli P, Nachanekar A, Kale V, Vaidya A (2023) Priming mesenchymal stromal cells with neurotrophic factors boosts the neuro-regenerative potential of their secretome. Regen Med 18(4):329–346. https://doi.org/10.2217/RME-2022-0201
Thomas, M., & Willerth, S. M. (2017). 3-D bioprinting of neural tissue for applications in cell therapy and drug screening. Front Bioeng Biotechnol 5(NOV):69. https://doi.org/10.3389/FBIOE.2017.00069/BIBTEX
Tu PH, Raju P, Robinson KA, Gurney ME, Trojanowski JQ, Lee VMY (1996) Transgenic mice carrying a human mutant superoxide dismutase transgene develop neuronal cytoskeletal pathology resembling human amyotrophic lateral sclerosis lesions. Proc Natl Acad Sci USA 93(7):3155–3160. https://doi.org/10.1073/PNAS.93.7.3155
Uhl EW, Warner NJ (2015) Mouse models as predictors of human responses: evolutionary medicine. Curr Pathobiol Rep 3(3):219–223. https://doi.org/10.1007/S40139-015-0086-Y
Wareham LK, Liddelow SA, Temple S, Benowitz LI, di Polo A, Wellington C, Goldberg JL, He Z, Duan X, Bu G, Davis AA, Shekhar K, Torre, A la, Chan DC, Canto-Soler MV, Flanagan JG, Subramanian P, Rossi S, Brunner T, Calkins DJ (2022) Solving neurodegeneration: common mechanisms and strategies for new treatments. Mol Neurodegener 17(1). https://doi.org/10.1186/S13024-022-00524-0
Waterston RH, Lindblad-Toh K, Birney E, Rogers J, Abril JF, Agarwal P, Agarwala R, Ainscough R, Alexandersson M, An P, Antonarakis SE, Attwood J, Baertsch R, Bailey J, Barlow K, Beck S, Berry E, Birren B, Bloom T, Lander ES (2002) Initial sequencing and comparative analysis of the mouse genome. Nature 420(6915):520–562. https://doi.org/10.1038/NATURE01262
West T, Hu Y, Verghese PB, Bateman RJ, Braunstein JB, Fogelman I, Budur K, Florian H, Mendonca N, Holtzman DM (2017) Preclinical and clinical development of ABBV-8E12 a humanized anti-tau antibody for treatment of Alzheimer’s disease and other tauopathies. J Prev Alzheimer’s Dis 4(4):236–241. https://doi.org/10.14283/JPAD.2017.36
Who. (2008) Global burden of neurological disorders estimates and projections. Public Health Challenges, Neurol Disord https://doi.org/10.1001/archneurol.2007.19
Xiao B, Hui Ng H, Takahashi R, Tan EK (2016) Induced pluripotent stem cells in Parkinson’s disease: scientific and clinical challenges. J Neurol Neurosurg Psychiatry 87(7):697–702. https://doi.org/10.1136/JNNP-2015-312036
Xicoy H, Wieringa B, Martens GJM (2017) The SH-SY5Y cell line in Parkinson’s disease research: a systematic review. Mol Neurodegener 12(1):1–11. https://doi.org/10.1186/S13024-017-0149-0/TABLES/4
Yagi T, Ito D, Okada Y, Akamatsu W, Nihei Y, Yoshizaki T, Yamanaka S, Okano H, Suzuki N (2011) Modeling familial Alzheimer’s disease with induced pluripotent stem cells. Hum Mol Genet 20(23):4530–4539. https://doi.org/10.1093/HMG/DDR394
Yanamandra K, Jiang H, Mahan TE, Maloney SE, Wozniak DF, Diamond MI, Holtzman DM (2015) Anti-tau antibody reduces insoluble tau and decreases brain atrophy. Ann Clin Transl Neurol 2(3):278–288. https://doi.org/10.1002/ACN3.176
Yanamandra K, Kfoury N, Jiang H, Mahan TE, Ma S, Maloney SE, Wozniak DF, Diamond MI, Holtzman DM (2013) Anti-tau antibodies that block tau aggregate seeding in vitro markedly decrease pathology and improve cognition in vivo. Neuron 80(2):402–414. https://doi.org/10.1016/J.NEURON.2013.07.046
Yang Q, Jiao B, Shen L (2020) The development of C9orf72-related amyotrophic lateral sclerosis and frontotemporal dementia disorders. Front Genet 11:1045. https://doi.org/10.3389/FGENE.2020.562758/BIBTEX
Zeng H, Sanes JR (2017) Neuronal cell-type classification: challenges, opportunities and the path forward. Nat Rev Neurosci 18(9):530–546. https://doi.org/10.1038/NRN.2017.85
Zhang J, Cai Q, Jiang M, Liu Y, Gu H, Guo J, Sun H, Fang J, Jin L (2017) Mesencephalic astrocyte-derived neurotrophic factor alleviated 6-OHDA-induced cell damage via ROS-AMPK/mTOR mediated autophagic inhibition. Exp Gerontol 89:45–56. https://doi.org/10.1016/J.EXGER.2017.01.010
Zhang X, Feng ZJ, Chergui K (2014) Allosteric modulation of GluN2C/GluN2D-containing NMDA receptors bidirectionally modulates dopamine release: implication for Parkinson’s disease. Br J Pharmacol 171(16):3938–3945. https://doi.org/10.1111/BPH.12758
Zhang Y, Chen H, Long X, Xu T (2022) Three-dimensional-engineered bioprinted in vitro human neural stem cell self-assembling culture model constructs of Alzheimer’s disease. Bioactive Materials 11:192–205. https://doi.org/10.1016/j.bioactmat.2021.09.023
Zhao Z, Nelson AR, Betsholtz C, Zlokovic BV (2015) Establishment and dysfunction of the blood-brain barrier. Cell 163(5):1064–1078. https://doi.org/10.1016/j.cell.2015.10.067
Zhu F, Nair RR, Fisher EMC, and Cunningham TJ (2019) Humanising the mouse genome piece by piece. Nature Commun 10(1). https://doi.org/10.1038/S41467-019-09716-7
Acknowledgements
The authors would like to thank the Symbiosis Centre for Research & Innovation (SCRI), Symbiosis International (Deemed University) (SIU), Pune, India, for providing Senior Research Fellowship (SRF) to PT.
Funding
The financial support was provided by the Symbiosis Centre for Research & Innovation (SCRI), Symbiosis International (Deemed University) (SIU), Pune, India, under Major Research Project (MJRP), Grant no: SIU/SCRI/MJRP/19–20/1516-C.
Author information
Authors and Affiliations
Contributions
Prajakta Teli: writing, original draft preparation; Vaijayanti Kale: reviewing and editing; Anuradha Vaidya: conceptualization, visualization, resources, writing, reviewing and editing, supervision and approval of the final draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Teli, P., Kale, V. & Vaidya, A. Beyond animal models: revolutionizing neurodegenerative disease modeling using 3D in vitro organoids, microfluidic chips, and bioprinting. Cell Tissue Res 394, 75–91 (2023). https://doi.org/10.1007/s00441-023-03821-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-023-03821-2