Abstract
Objective
To analyze the prognostic significance of plasma total cholesterol (TC) and high-density lipoprotein (HDL) in patients with gastric cancer receiving oxaliplatin combination with S-1 (SOX) chemotherapy after radical resection and to establish models of relevant prognostic influencing factors.
Method
The clinicopathologic features of 301 patients treated with SOX after radical gastrectomy were retrospectively analyzed. Univariate analysis, multivariate analysis, and the Kaplan–Meier survival curve were used to analyze the prognostic value of TC and HDL in patients undergoing adjuvant SOX chemotherapy after curative gastric surgery. Based on the results of multivariate Cox regression, we constructed nomograms to predict 1 and 3-year CSS (cancer-specific survival time) and DFS (disease-free survival time) in patients with adjuvant chemotherapy after radical gastrectomy. We assessed the model's accuracy using the consistency index (C index) and calibration curve, and the ROC curve and DCA curve were applied to compare it with TNM staging.
Results
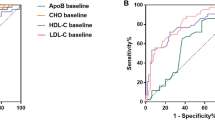
Multivariate analysis revealed that TC and HDL were independent influencing factors of CSS, while HDL was an independent factor unique to DFS. According to Kaplan–Meier curves, low TC and HDL suggested poor survival (P < 0.001). The relevant prognostic factors from the multivariate study were used to build nomograms for DFS and CSS. Both DFS and CSS had C index and AUC values that were higher than 0.71. The calibration curves demonstrated that the predicted results are consistent with the observed results. The AUC valves for DFS and CSS in our models were higher than TNM staging. The decision curve analysis indicated that net benefits were moderately positive. Significant differences in survival were seen between the high and low-risk groups according to the nomogram risk score.
Conclusions
TC and HDL have a certain significance for the prognosis of patients with gastric cancer after radical resection receiving adjuvant SOX chemotherapy. Lower TC and HDL suggested poor DFS and CSS. Both prediction models for CSS and DFS demonstrated good predictive ability and had a higher predictive value than the TNM staging system.








Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Asano K, Kubo M, Yonemoto K, Doi Y, Ninomiya T, Tanizaki Y, Arima H, Shirota T, Matsumoto T, Iida M et al (2008) Impact of serum total cholesterol on the incidence of gastric cancer in a population-based prospective study: the Hisayama study. Int J Cancer 122(4):909–914. https://doi.org/10.1002/ijc.23191
Bian X, Liu R, Meng Y, Xing D, Xu D, Lu Z (2021) Lipid metabolism and cancer. J Exp Med. https://doi.org/10.1084/jem.20201606
Cui MY, Yi X, Zhu DX, Wu J (2022) The role of lipid metabolism in gastric cancer. Front Oncol 12:916661. https://doi.org/10.3389/fonc.2022.916661
Espinosa G, López-Montero I, Monroy F, Langevin D (2011) Shear rheology of lipid monolayers and insights on membrane fluidity. Proc Natl Acad Sci U S A 108(15):6008–6013. https://doi.org/10.1073/pnas.1018572108
Fushida S, Kinoshita J, Oyama K, Fujimura T, Tsukada T, Yamaguchi T, Ninomiya I, Ohta T (2018) Multidisciplinary therapy for scirrhous gastric cancer: a retrospective analysis and proposal of new treatment strategy. Cancer Manag Res 10:3833–3839. https://doi.org/10.2147/cmar.S174950
Guo E, Chen L, Xie Q, Chen J, Tang Z, Wu Y (2007) Serum HDL-C as a potential biomarker for nodal stages in gastric cancer. Ann Surg Oncol 14(9):2528–2534. https://doi.org/10.1245/s10434-007-9401-0
Jiang Z, Sun Y, Zhang W, Cui C, Yang L, Zhou A (2020) Comparison of S-1 plus oxaliplatin (SOX) and capecitabine plus oxaliplatin (XELOX) as adjuvant chemotherapies for stage II and III gastric cancer after D2 resection: a single-center retrospective study. Asia Pac J Clin Oncol 16(3):180–186. https://doi.org/10.1111/ajco.13321
Johnston FM, Beckman M (2019) Updates on management of gastric cancer. Curr Oncol Rep 21(8):67. https://doi.org/10.1007/s11912-019-0820-4
Kang R, Li P, Wang T, Li X, Wei Z, Zhang Z, Zhong L, Cao L, Heckman MG, Zhang YW et al (2016) Apolipoprotein E epsilon 2 allele and low serum cholesterol as risk factors for gastric cancer in a Chinese Han population. Sci Rep 6:19930. https://doi.org/10.1038/srep19930
Kattan MW, Karpeh MS, Mazumdar M, Brennan MF (2003) Postoperative nomogram for disease-specific survival after an R0 resection for gastric carcinoma. J Clin Oncol 21(19):3647–3650. https://doi.org/10.1200/jco.2003.01.240
Kinoshita T, Nashimoto A, Yamamura Y, Okamura T, Sasako M, Sakamoto J, Kojima H, Hiratsuka M, Arai K, Sairenji M et al (2004) Feasibility study of adjuvant chemotherapy with S-1 (TS-1; tegafur, gimeracil, oteracil potassium) for gastric cancer. Gastric Cancer 7(2):104–109. https://doi.org/10.1007/s10120-004-0278-3
Ma L, Chen G, Wang D, Zhang K, Zhao F, Tang J, Zhao J, Røe OD, He S, Liao D et al (2022) A nomogram to predict survival probability of gastric cancer patients undergoing radical surgery and adjuvant chemotherapy. Front Oncol 12:893998. https://doi.org/10.3389/fonc.2022.893998
Macdonald JS, Smalley SR, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, Haller DG, Ajani JA, Gunderson LL, Jessup JM et al (2001) Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med 345(10):725–730. https://doi.org/10.1056/NEJMoa010187
Noh SH, Park SR, Yang HK, Chung HC, Chung IJ, Kim SW, Kim HH, Choi JH, Kim HK, Yu W et al (2014) Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol 15(12):1389–1396. https://doi.org/10.1016/s1470-2045(14)70473-5
Park SH, Lim DH, Sohn TS, Lee J, Zang DY, Kim ST, Kang JH, Oh SY, Hwang IG, Ji JH et al (2021) A randomized phase III trial comparing adjuvant single-agent S1, S-1 with oxaliplatin, and postoperative chemoradiation with S-1 and oxaliplatin in patients with node-positive gastric cancer after D2 resection: the ARTIST 2 trial(☆). Ann Oncol 32(3):368–374. https://doi.org/10.1016/j.annonc.2020.11.017
Qin WH, Yang ZS, Li M, Chen Y, Zhao XF, Qin YY, Song JQ, Wang BB, Yuan B, Cui XL et al (2020) High serum levels of cholesterol increase antitumor functions of nature killer cells and reduce growth of liver tumors in mice. Gastroenterology 158(6):1713–1727. https://doi.org/10.1053/j.gastro.2020.01.028
Rawla P, Barsouk A (2019) Epidemiology of gastric cancer: global trends, risk factors and prevention. Prz Gastroenterol 14(1):26–38. https://doi.org/10.5114/pg.2018.80001
Sakuramoto S, Sasako M, Yamaguchi T, Kinoshita T, Fujii M, Nashimoto A, Furukawa H, Nakajima T, Ohashi Y, Imamura H et al (2007) Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med 357(18):1810–1820. https://doi.org/10.1056/NEJMoa072252
Sasako M, Sakuramoto S, Katai H, Kinoshita T, Furukawa H, Yamaguchi T, Nashimoto A, Fujii M, Nakajima T, Ohashi Y (2011) Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol 29(33):4387–4393. https://doi.org/10.1200/jco.2011.36.5908
Semenkovich TR, Yan Y, Subramanian M, Meyers BF, Kozower BD, Nava R, Patterson GA, Kreisel D, Puri V (2021) A clinical nomogram for predicting node-positive disease in esophageal cancer. Ann Surg 273(6):e214–e221. https://doi.org/10.1097/sla.0000000000003450
Sexton RE, Al Hallak MN, Diab M, Azmi AS (2020) Gastric cancer: a comprehensive review of current and future treatment strategies. Cancer Metastasis Rev 39(4):1179–1203. https://doi.org/10.1007/s10555-020-09925-3
Shi L, Wang Z, Wang L, Jia Y, Li J, Qin Y (2022) A prognostic nomogram and heat map to predict survival in stage II/III gastric cancer patients after curative gastrectomy followed by adjuvant chemotherapy. Cancer Manag Res 14:287–301. https://doi.org/10.2147/cmar.S348890
Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, Fukushima M (1996) Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 7(5):548–557. https://doi.org/10.1097/00001813-199607000-00010
Sjoquist KM, Renfro LA, Simes RJ, Tebbutt NC, Clarke S, Seymour MT, Adams R, Maughan TS, Saltz L, Goldberg RM et al (2018) Personalizing survival predictions in advanced colorectal cancer: the ARCAD nomogram project. J Natl Cancer Inst 110(6):638–648. https://doi.org/10.1093/jnci/djx253
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Tamura T, Inagawa S, Hisakura K, Enomoto T, Ohkohchi N (2012) Evaluation of serum high-density lipoprotein cholesterol levels as a prognostic factor in gastric cancer patients. J Gastroenterol Hepatol 27(10):1635–1640. https://doi.org/10.1111/j.1440-1746.2012.07189.x
Xu H, Zhou S, Tang Q, Xia H, Bi F (2020) Cholesterol metabolism: new functions and therapeutic approaches in cancer. Biochim Biophys Acta Rev Cancer 1874(1):188394. https://doi.org/10.1016/j.bbcan.2020.188394
Yamada Y, Higuchi K, Nishikawa K, Gotoh M, Fuse N, Sugimoto N, Nishina T, Amagai K, Chin K, Niwa Y et al (2015) Phase III study comparing oxaliplatin plus S-1 with cisplatin plus S-1 in chemotherapy-naïve patients with advanced gastric cancer. Ann Oncol 26(1):141–148. https://doi.org/10.1093/annonc/mdu472
Yu J, Gao Y, Chen L, Wu D, Shen Q, Zhao Z, Liu W, Yang H, Zhang Q, Wang X et al (2022) Effect of S-1 plus oxaliplatin compared with fluorouracil, leucovorin plus oxaliplatin as perioperative chemotherapy for locally advanced, resectable gastric cancer: a randomized clinical trial. JAMA Netw Open 5(2):e220426. https://doi.org/10.1001/jamanetworkopen.2022.0426
Zhang X, Liang H, Li Z, Xue Y, Wang Y, Zhou Z, Yu J, Bu Z, Chen L, Du Y et al (2021) Perioperative or postoperative adjuvant oxaliplatin with S-1 versus adjuvant oxaliplatin with capecitabine in patients with locally advanced gastric or gastro-oesophageal junction adenocarcinoma undergoing D2 gastrectomy (RESOLVE): an open-label, superiority and non-inferiority, phase 3 randomised controlled trial. Lancet Oncol 22(8):1081–1092. https://doi.org/10.1016/s1470-2045(21)00297-7
Funding
This study was supported by the National Natural Science Foundation of China (grant no.82273381).
Author information
Authors and Affiliations
Contributions
YQ and SW contributed to the study conception and design. Material preparation, data collection and analysis were performed by SW, CC, JZ and JL. The first draft of the manuscript was written by SW and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This is an observational study. The First Affiliated Hospital of Zhengzhou University Research Ethics Committee has confirmed that no ethical approval is required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, S., Chen, C., Zhang, J. et al. Prognostic value of blood lipids on patients treated with oxaliplatin combined with S-1 (SOX) after radical gastrectomy and establishment of prognostic nomogram. J Cancer Res Clin Oncol 149, 10293–10305 (2023). https://doi.org/10.1007/s00432-023-04942-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04942-5