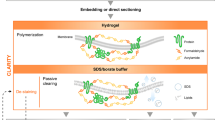
Abstract
Transcript labeling in intact tissues using in situ hybridization chain reaction has potential to provide vital spatiotemporal information for molecular characterization of heterogeneous neuronal populations. However, large tissue labeling in non-perfused or fresh-frozen rodent and postmortem human samples, which provide more flexible utilization than perfused tissues, is largely unexplored. In the present study, we optimized the combination of in situ hybridization chain reaction in fresh-frozen rodent brains and then evaluated the uniformity of neuronal labeling between two clearing methods, CLARITY and iDISCO+. We found that CLARITY yielded higher signal-to-noise ratios but more limited imaging depth and required longer clearing times, whereas, iDISCO+ resulted in better tissue clearing, greater imaging depth and a more uniform labeling of larger samples. Based on these results, we used iDISCO+-cleared fresh-frozen rodent brains to further validate this combination and map the expression of a few genes of interest pertaining to mood disorders. We then examined the potential of in situ hybridization chain reaction to label transcripts in cleared postmortem human brain tissues. The combination failed to produce adequate mRNA labeling in postmortem human cortical slices but produced visually adequate labeling in the cerebellum tissues. We next, investigated the multiplexing ability of in situ hybridization chain reaction in cleared tissues which revealed inconsistent fluorescence output depending upon the fluorophore conjugated to the hairpins. Finally, we applied our optimized protocol to assess the effect of glucocorticoid receptor overexpression on basal somatostatin expression in the mouse cortex. The constitutive glucocorticoid receptor overexpression resulted in lower number density of somatostatin-expressing neurons compared to wild type. Overall, the combination of in situ hybridization chain reaction with clearing methods, especially iDISCO+, may find broad application in the transcript analysis in rodent studies, but its limited use in postmortem human tissues can be improved by further optimizations.








Similar content being viewed by others

References
Azaripour A, Lagerweij T, Scharfbillig C, Jadczak AE, Willershausen B, Van Noorden CJF (2016) A survey of clearing techniques for 3D imaging of tissues with special reference to connective tissue. Prog Histochem Cytochem 51:9–23. https://doi.org/10.1016/J.PROGHI.2016.04.001
Biesemeier A, Schraermeyer U, Eibl O (2011) Chemical composition of melanosomes, lipofuscin and melanolipofuscin granules of human RPE tissues. Exp Eye Res 93:29–39. https://doi.org/10.1016/j.exer.2011.04.004
Bria A, Iannello G (2012) TeraStitcher: a tool for fast automatic 3D-stitching of teravoxel-sized microscopy images. BMC Bioinformatics 13:316. https://doi.org/10.1186/1471-2105-13-316
Choi HMT, Chang JY, Trinh LA, Padilla JE, Fraser SE, Pierce NA (2010) Programmable in situ amplification for multiplexed imaging of mRNA expression. Nat Biotechnol 28:1208–1212. https://doi.org/10.1038/nbt.1692
Choi HMT, Beck VA, Pierce NA (2014) Next-generation in situ hybridization chain reaction: higher gain, lower cost, greater durability. ACS Nano 8:4284–4294. https://doi.org/10.1021/nn405717p
Choi HMT, Schwarzkopf M, Fornace ME, Acharya A, Artavanis G, Stegmaier J, Cunha A, Pierce NA (2018) Third-generation in situ hybridization chain reaction: multiplexed, quantitative, sensitive, versatile, robust. Development 145:dev165753. https://doi.org/10.1242/dev.165753
Chung K, Wallace J, Kim S-Y, Kalyanasundaram S, Andalman AS, Davidson TJ, Mirzabekov JJ, Zalocusky KA, Mattis J, Denisin AK, Pak S, Bernstein H, Ramakrishnan C, Grosenick L, Gradinaru V, Deisseroth K (2013) Structural and molecular interrogation of intact biological systems. Nature. https://doi.org/10.1038/nature12107
Dirks RM, Pierce NA (2004) From the cover: triggered amplification by hybridization chain reaction. Proc Natl Acad Sci 101:15275–15278. https://doi.org/10.1073/pnas.0407024101
Fee C, Banasr M, Sibille E (2017) Somatostatin-positive GABA interneuron deficits in depression: cortical microcircuit and therapeutic perspectives. Biol Psychiatry 82:549. https://doi.org/10.1016/J.BIOPSYCH.2017.05.024
Fung SJ, Webster MJ, Sivagnanasundaram S, Duncan C, Elashoff M, Weickert CS (2010) Expression of interneuron markers in the dorsolateral prefrontal cortex of the developing human and in schizophrenia. Am J Psychiatry 167:1479–1488. https://doi.org/10.1176/appi.ajp.2010.09060784
Hebda-Bauer EK, Pletsch A, Darwish H, Fentress H, Simmons TA, Wei Q, Watson SJ, Akil H (2010) Forebrain glucocorticoid receptor overexpression increases environmental reactivity and produces a stress-induced spatial discrimination deficit. Neuroscience 169:645–653. https://doi.org/10.1016/j.neuroscience.2010.05.033
Iritani S, Satoh K (1991) Distribution of somatostatin-immunoreactive cell bodies and fibers in the neocortex ofMacaca fuscata. Synapse 9:50–59. https://doi.org/10.1002/syn.890090108
Jones EG, Hendry SHC, Liu X-B, Hodgins S, Potkin SG, Tourtellotte WW (1992) A method for fixation of previously fresh-frozen human adult and fetal brains that preserves histological quality and immunoreactivity. J Neurosci Methods 44:133–144. https://doi.org/10.1016/0165-0270(92)90006-Y
Kawaguchi Y, Kubota Y (1997) GABAergic cell subtypes and their synaptic connections in rat frontal cortex. Cereb Cortex 7:476–486. https://doi.org/10.1093/cercor/7.6.476
Kramer EE, Steadman PE, Epp JR, Frankland PW, Josselyn SA (2018) Assessing individual neuronal activity across the intact brain: using hybridization chain reaction (HCR) to detect arc mRNA localized to the nucleus in volumes of cleared brain tissue. Curr Protoc Neurosci. https://doi.org/10.1002/cpns.49
Krolewski DM, Kumar V, Martin B, Tomer R, Deisseroth K, Myers RM, Schatzberg AF, Lee FS, Barchas JD, Bunney WE, Akil H, Watson SJ (2018) Quantitative validation of immunofluorescence and lectin staining using reduced CLARITY acrylamide formulations. Brain Struct Funct 223:987–999. https://doi.org/10.1007/s00429-017-1583-z
Lee J, Vogt CE, McBrairty M, Al-Hashimi HM (2013) Influence of Dimethylsulfoxide on RNA Structure and Ligand Binding. Anal Chem 85:9692–9698. https://doi.org/10.1021/AC402038T
Lin LC, Sibille E (2013) Reduced brain somatostatin in mood disorders: a common pathophysiological substrate and drug target? Front Pharmacol 4:110. https://doi.org/10.3389/fphar.2013.00110
Lin LC, Sibille E (2015) Somatostatin, neuronal vulnerability and behavioral emotionality. Mol Psychiatry 20:377–387. https://doi.org/10.1038/mp.2014.184
Markram H, Toledo-Rodriguez M, Wang Y, Gupta A, Silberberg G, Wu C (2004) Interneurons of the neocortical inhibitory system. Nat Rev Neurosci 5:793–807. https://doi.org/10.1038/nrn1519
Papachristou DN, Liu JL, Patel YC (1994) Glucocorticoids regulate somatostatin peptide and steady state messenger ribonucleic acid levels in normal rat tissues and in a somatostatin-producing islet tumor cell line (1027B2). Endocrinology 134:2259–2266. https://doi.org/10.1210/endo.134.5.7908873
Pardue ML, Gall JG (1969) Molecular hybridization of radioactive DNA to the DNA of cytological preparations. Proc Natl Acad Sci 64:600–604. https://doi.org/10.1073/pnas.64.2.600
Park Y-G, Sohn CH, Chen R, McCue M, Yun DH, Drummond GT, Ku T, Evans NB, Oak HC, Trieu W, Choi H, Jin X, Lilascharoen V, Wang J, Truttmann MC, Qi HW, Ploegh HL, Golub TR, Chen S-C, Frosch MP, Kulik HJ, Lim BK, Chung K (2018) Protection of tissue physicochemical properties using polyfunctional crosslinkers. Nat Biotechnol 37:73–83. https://doi.org/10.1038/nbt.4281
PubChem-AF488 5-Alexa Fluor 488 TFP ester | C27H12F4Li2N2O11S2 - PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/135564337. Accessed 13 Feb 2020
PubChem-AF594 Alexa Fluor 594 meta-isomer | C39H37N3O13S2 - PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/25137945. Accessed 13 Feb 2020
PubChem-AF647 Alexa Fluor 647 | C41H59N3O17PS4+ - PubChem. https://pubchem.ncbi.nlm.nih.gov/compound/102227060. Accessed 13 Feb 2020
Renier N, Wu Z, Simon DJ, Yang J, Ariel P, Tessier-Lavigne M (2014) iDISCO: a simple, rapid method to immunolabel large tissue samples for volume imaging. Cell 159:896–910. https://doi.org/10.1016/j.cell.2014.10.010
Silvestri L, Costantini I, Sacconi L, Pavone FS (2016) Clearing of fixed tissue: a review from a microscopist’s perspective. J Biomed Opt 21:081205. https://doi.org/10.1117/1.JBO.21.8.081205
Sonntag K-C, Tejada G, Subburaju S, Berretta S, Benes FM, Woo T-UW (2016) Limited predictability of postmortem human brain tissue quality by RNA integrity numbers. J Neurochem 138:53–59. https://doi.org/10.1111/jnc.13637
Sylwestrak EL, Rajasethupathy P, Wright MA, Jaffe A, Deisseroth K (2016) Multiplexed intact-tissue transcriptional analysis at cellular resolution. Cell. https://doi.org/10.1016/j.cell.2016.01.038
Tomer R, Ye L, Hsueh B, Deisseroth K (2014) Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nat Protoc. https://doi.org/10.1038/nprot.2014.123
Urban-Ciecko J, Barth AL (2016) Somatostatin-expressing neurons in cortical networks. Nat Rev Neurosci 17:401–409. https://doi.org/10.1038/nrn.2016.53
Wan P, Zhu J, Xu J, Li Y, Yu T, Zhu D (2018) Evaluation of seven optical clearing methods in mouse brain. Neurophotonics 5:1. https://doi.org/10.1117/1.NPh.5.3.035007
Wang L, Nie J, Sicotte H, Li Y, Eckel-Passow JE, Dasari S, Vedell PT, Barman P, Wang L, Weinshiboum R, Jen J, Huang H, Kohli M, Kocher J-PA (2016) Measure transcript integrity using RNA-seq data. BMC Bioinformatics 17:58. https://doi.org/10.1186/s12859-016-0922-z
Wei Q, Lu X-Y, Liu L, Schafer G, Shieh K-R, Burke S, Robinson TE, Watson SJ, Seasholtz AF, Akil H (2004) Glucocorticoid receptor overexpression in forebrain: a mouse model of increased emotional lability. Proc Natl Acad Sci USA 101:11851–11856. https://doi.org/10.1073/pnas.0402208101
Wei Q, Hebda-Bauer EK, Pletsch A, Luo J, Hoversten MT, Osetek AJ, Evans SJ, Watson SJ, Seasholtz AF, Akil H (2007) Overexpressing the glucocorticoid receptor in forebrain causes an aging-like neuroendocrine phenotype and mild cognitive dysfunction. J Neurosci 27:8836–8844. https://doi.org/10.1523/jneurosci.0910-07.2007
Wei Q, Fentress HM, Hoversten MT, Zhang L, Hebda-Bauer EK, Watson SJ, Seasholtz AF, Akil H (2012) Early-life forebrain glucocorticoid receptor overexpression increases anxiety behavior and cocaine sensitization. Biol Psychiatry 71:224–231. https://doi.org/10.1016/j.biopsych.2011.07.009
Wu Y, Rivenson Y, Wang H, Luo Y, Ben-David E, Bentolila LA, Pritz C, Ozcan A (2019) Three-dimensional virtual refocusing of fluorescence microscopy images using deep learning. Nat Methods. https://doi.org/10.1038/s41592-019-0622-5
Yang B, Treweek JB, Kulkarni RP, Deverman BE, Chen C-K, Lubeck E, Shah S, Cai L, Gradinaru V (2014) Single-cell phenotyping within transparent intact tissue through whole-body clearing. Cell. https://doi.org/10.1016/j.cell.2014.07.017
Yin D (1996) Biochemical basis of lipofuscin, ceroid, and age pigment-like fluorophores. Free Radic Biol Med 21:871–888
Zheng H, Rinaman L (2016) Simplified CLARITY for visualizing immunofluorescence labeling in the developing rat brain. Brain Struct Funct 221:2375–2383. https://doi.org/10.1007/s00429-015-1020-0
Acknowledgements
This work was supported by the Pritzker Neuropsychiatric Research Consortium, the Hope for Depression Research Foundation, National Institute of Health R01MH104261, Office of Naval Research Grant N00014-12-1-0366 and National Institute on Drug Abuse U01DA043098. The authors have no conflicts of interest to declare. The authors would like to thank Ms. Jennifer Fitzpatrick, Mr. Evan Hughes, Ms. Claire Barcelo, and Mr. Hui Li for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
V.K., D.M.K., E.K.H., B.M., M.F., H.A., and S.J.W. are members of the Pritzker Neuropsychiatric Research Consortium, which is supported by the Pritzker Neuropsychiatric Disorders Research Fund, LLC (Fund). There exists a shared intellectual property agreement between the academic and philanthropic entities of the Consortium. The Fund has no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical approval
“All animal care and procedures performed in studies involving animals were in accordance with the ethical standards of the guide for the Care and Use of Laboratory Animals: Eighth Edition (revised in 2011, published by the National Academy of Sciences), and approved by the University of Michigan Committee on the Use and Care of Animals.“This article does not contain any studies with human participants performed by any of the authors.”
Informed consent
Postmortem human brains were collected by the Brain Donor Program at the University of California, Irvine with the consent of the relatives of the deceased.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (M4V 11080 KB) Supplemental movie ESM_1. 3D visualization of differences in the HCR-FISH labeled Sst signal between CLARITY (left panel) and iDISCO+ (right panel) processed mouse cortical slices. First half of this movie shows a series of xy-plane images moving in z-direction of confocal acquired representative stacks, followed by maximum intensity projection (mip) 3D pre-quantitation volume and post-quantitation surface rendering of Sst neurons. Second half of the movie shows a series of xz-plane images moving in y-direction, followed by mip 3D pre-quantitation volume and post-quantitation surface renderings of Sst neurons. This animation highlights the higher number density of detected Sst neurons and uniform labeling across the z-plane in iDISCO+ compared to CLARITY.
Supplementary file2 (M4V 43589 KB) Supplemental movie ESM_2. 3D visualization of HCR FISH-labeled Sst expressing neurons in a rat forebrain hemi-slice cleared using iDISCO+. This movie first shows a series of xy-plane images acquired on a COLM followed by a mip 3D volume rendering. The characteristic layer-specific expression pattern of Sst in rat cortex can be observed in this movie.
Supplementary file3 (M4V 8041 KB) Supplemental movie ESM_3. Volume rendering of a rat hippocampus showing Sst expression pattern in the iDISCO+ cleared intact tissue and labeled using HCR FISH method. This movie shows a series of xy-plane images acquired on a COLM followed by 3D mip volume rendering.
Supplementary file4 (M4V 38281 KB) Supplemental movie ESM_4. 3D visualization of a mouse left hemisphere showing Sst expressing neurons across the entire rostro-caudal extent of the brain. Post HCR FISH labeling, brain tissue was cleared using the iDISCO+ method and imaging was performed on a COLM.
Supplementary file5 (MP4 108109 KB) Supplemental movie ESM_5. Sst expression in an iDISCO+ cleared sample of a mouse left hemi-brainstem. COLM-acquired images are first rendered as a series of xy-planes progressing in the medio-lateral direction and subsequently as mip 3D volume rendering.
Supplementary file6 (M4V 79471 KB) Supplemental movie ESM_6. Volume rendering of HCR FISH-labeled Pvalb expressing neurons in a rat forebrain hemi-slice. This representative COLM-acquired image stack of an iDISCO+-cleared brain shows the predominant cortical expression of Pvalb.
Supplementary file7 (MP4 94155 KB) Supplemental movie ESM_7. 3D visualization of HCR FISH-labeled Pvalb expressing neurons in a rat brainstem hemi-slice showing the expression pattern in cerebellum and several nuclei. iDISCO+-cleared and COLM-acquired image stacks are first projected as a series of xy-planes moving in a rostral-caudal-rostral direction, then as a mip 3D volume rendering.
Supplementary file8 (MP4 18243 KB) Supplemental movie ESM_8. 3D visualization of an iDISCO+ cleared rat mid-brain slice showing the expression of Th mRNA in the SNr/c, VTA, PBP and PAG regions. This movie first shows the rostro-caudal progression of a series of xy-plane images acquired on the COLM followed by a mip 3D volume rendering.
Supplementary file9 (M4V 22808 KB) Supplemental movie ESM_9. Dbh expressing neurons in the rat locus coeruleus are shown here first as a series of xy-plane images acquired on a confocal microscope followed by a mip 3D volume. This representative brainstem hemi-slice was processed through HCR FISH and cleared using the iDISCO+ method.
Supplementary file10 (M4V 6137 KB) Supplemental movie ESM_10. 3D visualization of HCR FISH-labeled CALB expressing neurons in the postmortem human cerebellum block. Confocal microscope-acquired image stacks are rendered first as a series of xy-plane images followed by a mip 3D volume rendering.
Supplementary file11 (M4V 13962 KB) Supplemental movie ESM_11. PVALB expressing neurons in the postmortem human cerebellum block are visualized in this movie as a series of xy-plane images then by a mip 3D volume. This representative block was cleared using iDISCO+ and images were acquired on a confocal microscope.
Supplementary file12 (MP4 10459 KB) Supplemental movie ESM_12. 3D visualization of COLM acquired dual HCR FISH labeling in iDISCO+ cleared mouse hemisphere. Movie shows labeling of Pvalb (green, AF-647) and Vglut1 (red, AF-594) mRNAs in a series of dorso-ventral planes from a down-sampled image volume. Left pane shows progression of z-planes from an intact hemisphere whereas right pane shows a zoomed-in view of ventral hippocampus and cortex (regions of lateral entorhinal cortex) whose z-plane progression is synced anatomically with the whole hemisphere in the left pane.
Rights and permissions
About this article
Cite this article
Kumar, V., Krolewski, D.M., Hebda-Bauer, E.K. et al. Optimization and evaluation of fluorescence in situ hybridization chain reaction in cleared fresh-frozen brain tissues. Brain Struct Funct 226, 481–499 (2021). https://doi.org/10.1007/s00429-020-02194-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-020-02194-4