Abstract
Main conclusion
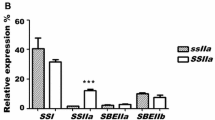
ZmSUS1 increases the amylose content of maize by regulating the expression of Shrunken2 (Sh2) and Brittle2 (Bt2) which encode the size subunits of endosperm ADP-glucose pyrophosphorylase, and Granule bound starchsynthase1 (GBSS1) and Starch synthase1 (SS1).
Abstract
Cereal crops accumulate starch in seeds as an energy reserve. Sucrose Synthase (SuSy) plays an important role in grain starch synthesis. In this study, ZmSUS1 was transformed into maize inbred line KN5585, and transgenic plants were obtained. Compared with the non-transgenic negative control, the content and activity of SuSy were significantly increased, the amylose content in mature seeds of transgenic maize increased by 41.1–69.2%, the total starch content increased by 5.0–13.5%, the 100-grain weight increased by 19.0–26.2% and the average diameter of starch granules increased by 10.8–17.2%. These results indicated that overexpression of ZmSUS1 can significantly improve the traits of maize seeds and obtain new lines with high amylose content. It was also found that the overexpression of ZmSUS1 may increase the amylose content by altering the expression of endosperm ADP-glucose pyrophosphorylase (AGPase) subunits Shrunken2 (Sh2) and Brittle2 (Bt2). Moreover, the ectopic expression of ZmSUS1 also affected the expression of Granule bound starch synthase1 (GBSS1) and Starch synthase1 (SS1) which encode starch synthase. This study proved the important role of ZmSUS1 in maize starch synthesis and provided a new technology strategy for improving maize starch content and yield.






Similar content being viewed by others
Data availability
All datasets generated for this study are included in the article/ Supplementary Materials.
Abbreviations
- ADPG:
-
ADP-glucose
- AGPase:
-
ADP-glucose pyrophosphorylase
- Bt2:
-
Brittle2
- DAP:
-
Days after pollination
- DEG:
-
Differentially expressed gene
- GBSS1:
-
Granule bound starchsynthase1
- Sh2:
-
Shrunken2
- SS1:
-
Starch synthase1
- SuSy:
-
Sucrose synthase
- UDPG:
-
UDP-glucose
References
Abt MR, Pfister B, Sharma M, Eicke S, Bürgy L, Neale I, Seung D, Zeeman SC (2020) STARCH SYNTHASE5, a noncanonical starch synthase-like protein, promotes starch granule initiation in Arabidopsis. Plant Cell 32(8):2543–2565. https://doi.org/10.1105/TPC.19.00946
Amor Y, Haigler CH, Johnson S, Wainscott M, Delmer DP (1995) A membrane-associated form of sucrose synthase and its potential role in the synthesis of cellulose and callose in plants. Proc Natl Acad Sci USA 92(20):9353–9357. https://doi.org/10.1073/pnas.92.20.9353
Baroja-Fernández E, Muñoz FJ, Saikusa T, Rodríguez-López M, Akazawa T, Pozueta-Romero JJP (2003) Sucrose synthase catalyzes the de novo production of ADP-glucose linked to starch biosynthesis in heterotrophic tissues of plants. Plant Cell Physiol 44(5):500–509. https://doi.org/10.1093/pcp/pcg062
Baroja-Fernández E, Muñoz FJ, Montero M, Etxeberria E, Sesma MT, Ovecka M, Bahaji A, Ezquer I, Li J, Prat S, Pozueta-Romero J (2009) Enhancing sucrose synthase activity in transgenic potato (Solanum tuberosum L.) tubers results in increased levels of starch, ADP-glucose, and UDP-glucose, and total yield. Plant Cell Physiol 50(9):1651–1662. https://doi.org/10.1093/pcp/pcp108
Betti M, Ciacci C, Abramovich S, Frontalini F (2021) Protein extractions from Amphistegina lessonii: protocol development and optimization. Life 11(5):418. https://doi.org/10.3390/life11050418
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Chourey PS, Nelson OE (1976) The enzymatic deficiency conditioned by the shrunken-1 mutations in maize. Biochem Genetics 14(11–12):1041–1055. https://doi.org/10.1007/bf00485135
Chourey PS, Taliercio EW (1994) Epistatic interaction and functional compensation between the two tissue- and cell-specific sucrose synthase genes in maize. Proc Natl Acad Sci USA 91(17):7917–7921. https://doi.org/10.1073/pnas.91.17.7917
Crofts N, Abe K, Aihara S, Itoh R, Nakamura Y, Itoh K, Fujita N (2012) Lack of starch synthase IIIa and high expression of granule-bound starch synthase I synergistically increase the apparent amylose content in rice endosperm. Plant Sci 193–194:62–69. https://doi.org/10.1016/j.plantsci.2012.05.006
Deng Y, Wang J, Zhang Z, Wu Y (2020) Transactivation of Sus1 and Sus2 by Opaque2 is an essential supplement to sucrose synthase-mediated endosperm filling in maize. Plant Biotechnol J 18(9):1897–1907. https://doi.org/10.1111/pbi.13349
Duncan KA, Hardin SC, Huber SC (2006) The three maize sucrose synthase isoforms differ in distribution, localization, and phosphorylation. Plant Cell Physiol 47(7):959–971. https://doi.org/10.1093/pcp/pcj068
Fan C, Feng S, Huang J, Wang Y, Wu L, Li X, Wang L, Tu Y, Xia T, Li J, Cai X, Peng L (2017) AtCesA8-driven OsSUS3 expression leads to largely enhanced biomass saccharification and lodging resistance by distinctively altering lignocellulose features in rice. Biotechnol Biofuels 10:221. https://doi.org/10.1186/s13068-017-0911-0
Fujita N, Satoh R, Hayashi A, Kodama M, Itoh R, Aihara S, Nakamura Y (2011) Starch biosynthesis in rice endosperm requires the presence of either starch synthase I or IIIa. J Exp Bot 62(14):4819–4831. https://doi.org/10.1093/job/err125
Haigler CH, Ivanova-Datcheva M, Hogan PS, Salnikov VV, Hwang S, Martin K, Delmer DP (2001) Carbon partitioning to cellulose synthesis. Plant Mol Biol 47(1–2):29–51. https://doi.org/10.1023/A:1010615027986
He S, Hao X, Wang S, Zhou W, Ma Q, Lu X, Chen L, Zhang P (2022) Starch synthase II plays a crucial role in starch biosynthesis and the formation of multienzyme complexes in cassava storage roots. J Exp Bot 73(8):2540–2557. https://doi.org/10.1093/jxb/erac022
Huang L, Tan H, Zhang C, Li Q, Liu Q (2021) Starch biosynthesis in cereal endosperms: an updated review over the last decade. Plant Commun 2(5):100237. https://doi.org/10.1016/j.xplc.2021.100237
Jiang Y, Guo W, Zhu H, Ruan YL, Zhang T (2012) Overexpression of GhSusA1 increases plant biomass and improves cotton fiber yield and quality. Plant Biotechnol J 10(3):301–312. https://doi.org/10.1111/j.1467-7652.2011.00662.x
Jiang L, Yu X, Qi X, Yu Q, Deng S, Bai B, Li N, Zhang A, Zhu C, Liu B, Pang J (2013) Multigene engineering of starch biosynthesis in maize endosperm increases the total starch content and the proportion of amylose. Transgenic Res 22(6):1133–1142. https://doi.org/10.1007/s11248-013-9717-4
Kim H-Y, Jane J-L, Lamsal B (2017) Hydroxypropylation improves film properties of high amylose corn starch. Ind Crops Prod 95:175–183. https://doi.org/10.1016/j.indcrop.2016.10.025
Li L, Jiang H, Campbell M, Blanco M, Jane J-l (2008) Characterization of maize amylose-extender (ae) mutant starches. Part I: relationship between resistant starch contents and molecular structures. Carbohydr Polymers 74(3):396–404. https://doi.org/10.1016/j.carbpol.2008.03.012
Li N, Zhang S, Zhao Y, Li B, Zhang J (2011) Over-expression of AGPase genes enhances seed weight and starch content in transgenic maize. Planta 233(2):241–250. https://doi.org/10.1007/s00425-010-1296-5
Li J, Baroja-Fernandez E, Bahaji A, Munoz FJ, Ovecka M, Montero M, Sesma MT, Alonso-Casajus N, Almagro G, Sanchez-Lopez AM, Hidalgo M, Zamarbide M, Pozueta-Romero J (2013) Enhancing sucrose synthase activity results in increased levels of starch and ADP-glucose in maize (Zea mays L.) seed endosperms. Plant Cell Physiol 54(2):282–294. https://doi.org/10.1093/pcp/pcs180
Li C, Qiao Z, Qi W, Wang Q, Yuan Y, Yang X, Tang Y, Mei B, Lv Y, Zhao H, Xiao H, Song R (2015) Genome-wide characterization of cis-acting DNA targets reveals the transcriptional regulatory framework of opaque2 in maize. Plant Cell 27(3):532–545. https://doi.org/10.1105/tpc.114.134858
Lloyd JR, Springer F, Buléon A, Müller-Röber B, Willmitzer L, Kossmann J (1999) The influence of alterations in ADP-glucose pyrophosphorylase activities on starch structure and composition in potato tubers. Planta 209(2):230–238. https://doi.org/10.1007/s004250050627
Martin C, Smith AM (1995) Starch biosynthesis. Plant Cell 7(7):971–985. https://doi.org/10.1105/tpc.7.7.971
Nelly Sandrine AM, Zhao H, Qin Y, Sun Q, Gong D, Pan Z, Qiu F (2020) 22KD zein content coordinates transcriptional activity during starch synthesis in maize endosperm. Agronomy 10(5):624. https://doi.org/10.3390/agronomy10050624
Okita TW (1992) Is there an alternative pathway for starch synthesis? Plant Physiol 100(2):560–564. https://doi.org/10.1104/pp.100.2.560
Raigond P, Ezekiel R, Raigond B (2015) Resistant starch in food: a review. J Sci Food Agric 95(10):1968–1978. https://doi.org/10.1002/jsfa.6966
Regassa A, Nyachoti CM (2018) Application of resistant starch in swine and poultry diets with particular reference to gut health and function. Animal Nutr 4(3):305–310. https://doi.org/10.1016/j.aninu.2018.04.001
Ren X, Zhang J (2013) Research progresses on the key enzymes involved in sucrose metabolism in maize. Carbohydr Res 368:29–34. https://doi.org/10.1016/j.carres.2012.10.016
Ross HA, Davies HV (1992) Sucrose metabolism in tubers of potato. Effects of sink removal and sucrose flux on sucrosedegrading enzymes. Plant Physiol 98(1):287–293. https://doi.org/10.1104/pp.98.1.287
Sakuma C, Sato T, Shibata T, Nakagawa M, Kurosawa Y, Okumura CJ, Maruyama T, Arakawa T, Akuta T (2021) Western blotting analysis of proteins separated by agarose native gel electrophoresis. Int J Biol Macromol 166:1106–1110. https://doi.org/10.1016/j.ijbiomac.2020.10.265
Schmölzer K, Gutmann A, Diricks M, Desmet T, Nidetzky B (2016) Sucrose synthase: a unique glycosyltransferase for biocatalytic glycosylation process development. Biotechnol Adv 34(2):88–111. https://doi.org/10.1016/j.biotechadv.2015.11.003
Shannon JC, Pien FM, Liu KC (1996) Nucleotides and nucleotide sugars in developing maize endosperms (synthesis of ADP-glucose in brittle-1). Plant Physiol 110(3):835–843. https://doi.org/10.1104/pp.110.3.835
Smith AM (2001) The biosynthesis of starch granules. Biomacromol 2(2):335–341. https://doi.org/10.1021/bm000133c
Stawski D (2008) New determination method of amylose content in potato starch. Food Chem 110(3):777–781. https://doi.org/10.1016/j.foodchem.2008.03.009
Winter H, Huber SC (2000) Regulation of sucrose metabolism in higher plants: localization and regulation of activity of key enzymes. Crit Rev Biochem Mol Biol 35(4):253–289. https://doi.org/10.1080/10409230008984165
Winter H, Huber JL, Huber SC (1997) Membrane association of sucrose synthase: changes during the graviresponse and possible control by protein phosphorylation. FEBS Lett 420(2–3):151–155. https://doi.org/10.1016/s0014-5793(97)01506-8
Xu S-M, Brill E, Llewellyn DJ, Furbank RT, Ruan Y-L (2012) Overexpression of a potato sucrose synthase gene in cotton accelerates leaf expansion, reduces seed abortion, and enhances fiber production. Mol Plant 5(2):430–441. https://doi.org/10.1093/mp/ssr090
Zha K, Xie H, Ge M, Wang Z, Wang Y, Si W, Gu L (2019) Expression of maize MADS transcription factor ZmES22 negatively modulates starch accumulation in rice endosperm. Int J Mol Sci 20(3):483. https://doi.org/10.3390/ijms20030483
Zhan J, Li G, Ryu C-H, Ma C, Zhang S, Lloyd A, Hunter BG, Larkins BA, Drews GN, Wang X, Yadegari R (2018) Opaque-2 regulates a complex gene network associated with cell differentiation and storage functions of maize endosperm. Plant Cell 30(10):2425–2446. https://doi.org/10.1105/tpc.18.00392
Zhang Z, Zheng X, Yang J, Messing J, Wu Y (2016) Maize endosperm-specific transcription factors O2 and PBF network the regulation of protein and starch synthesis. Proc Natl Acad Sci USA 113(39):10842–10847. https://doi.org/10.1073/pnas.1613721113
Zhang X, Gao X, Li Z, Xu L, Li Y, Zhang R, Xue J, Guo D-W (2020) The effect of amylose on kernel phenotypic characteristics, starch-related gene expression and amylose inheritance in naturally mutated high-amylose maize. J Integr Agric 19(6):1554–1564. https://doi.org/10.1016/S2095-3119(19)62779-6
Zhao Y, Li N, Li B, Li Z, Xie G, Zhang J (2015) Reduced expression of starch branching enzyme IIa and IIb in maize endosperm by RNAi constructs greatly increases the amylose content in kernel with nearly normal morphology. Planta 241(2):449–461. https://doi.org/10.1007/s00425-014-2192-1
Zhu L, Gu M, Meng X, Cheung S, Yu H, Huang J, Sun Y, Shi Y, Liu Q (2012) High-amylose rice improves indices of animal health in normal and diabetic rats. Plant Biotechnol J 10(3):353–362. https://doi.org/10.1111/j.1467-7652.2011.00667.x
Zrenner R, Salanoubat M, Willmitzer L, Sonnewald U (1995) Evidence of the crucial role of sucrose synthase for sink strength using transgenic potato plants (Solanum tuberosum L.). Plant J 7(1):97–107. https://doi.org/10.1046/j.1365-313x.1995.07010097.x
Acknowledgements
We would like to thank BGI Tech Solutions Co., Ltd (Shenzhen, China) for Illumina sequencing and primary bioinformatics analysis. This study was financially supported by the National Key R&D Program of China (2018YFD1000500), Integration of Science and Education Program Foundation for the Talents by the Qilu University of Technology, Shandong Academy of Sciences (No.2018-81110268), and Shandong Natural Science Foundation (ZR2020MC099).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, P., Ma, H., Xiao, N. et al. Overexpression of the ZmSUS1 gene alters the content and composition of endosperm starch in maize (Zea mays L.). Planta 257, 97 (2023). https://doi.org/10.1007/s00425-023-04133-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04133-z