Abstract
Main conclusion
Several cis-elements including Myb-binding motifs together confer glandular trichome specificity as revealed from heterologous expression and analysis of menthol biosynthesis pathway gene promoters.
Abstract
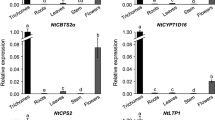
Glandular Trichomes (GTs) are result of division of epidermal cells that produce diverse metabolites. Species of mint family are important for their essential oil containing many high-value terpenoids, biosynthesized and stored in these GTs. Hence, GTs constitute attractive targets for metabolic engineering and GT-specific promoters are important. In this investigation, the upstream regions of the Mentha × piperita menthol biosynthetic pathway genes (-)-limonene synthase, (-)-P450 limonene-3- hydroxylase, (-)-trans-isopiperitenol dehydrogenase, (-)-Isopiperitenone reductase, ( +)-Pulegone reductase, (-)-Menthone reductase/ (-)-Menthol dehydrogenase and a branched pathway gene ( +)-menthofuran synthase were isolated and characterized. These fragments, fused to β-glucuronidase (GUS) reporter gene of pBI101 binary vector, are able to drive high level gene expression in transgenic tobacco trichomes with strong signals in GTs, except for (-)-Isopiperitenone reductase. The GT-enriched tissue from transformed plants were analysed for GUS enzyme activity and RNA expression which correlates the GUS staining. To characterize the cis-elements responsible for GT-specific expression, a series of 5’ deletion constructs for MpPLS and MpPMFS were cloned and analysed in stable transgenic tobacco lines. The specificity of trichome expression was located to − 797 to− 598 bp sequence for (-)-limonene synthase and− 629 to − 530 bp for ( +)-menthofuran synthase promoters containing specific Myb-binding motifs in addition to other unique motifs described for developmental regulation without any defined pattern. All other pathway promoters also recruits specific but different Myb factors as indicated by this analysis.







Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this article [and its supplementary information files] and nucleotide sequence information is available in GeneBank database [http://www.ncbi.nlm.nih.gov] under accession numbers mentioned in materials and methods.
References
Akhtar QM, Qamar N, Yadav P, Kulkarni P, Kumar A, Shasany AK (2017) Comparative glandular trichome transcriptome-based gene characterization reveals reasons for differential (−)-menthol biosynthesis in Mentha species. Physiol Plant. https://doi.org/10.1111/ppl.12550
Alcala G, Calo L, Gros F, Caissard JC, Gotor C, Romero LC (2005) A versatile promoter for the expression of proteins in glandular and non-glandular trichomes from a variety of plants. J Exp Bot 56(419):2487–2494
Alonso WR, Rajaonarivony JIM, Gershenzon J, Croteau R (1992) Purification of 4-S limonene synthase, a monoterpene cyclase from glandular trichomes of peppermint (M. × piperia) and spearmint (Mentha Χ spicata). J Biol Chem 267:7582–7587
Altschul SF, Madden TL, Schaffer AA, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Amelunxen F (1965) Elektronenmikroskopische Untersuchungen an den Dru¨ senschuppen von Mentha× piperita L. Plant Med 13:457–473
Amelunxen F, Wahlig T, Arbeiter H (1969) U¨ber den Nachweis des atherischen O¨ ls in isolierten Dru¨senhaaren und Dru¨senschuppen von Mentha × piperita L. Z Pflanzenphysiol 61:68–72
Bertea CM, Schalk M, Karp F, Maffei M, Croteau R (2001) Demonstration that menthofuran synthase of mint (Mentha) is a cytochrome P450 monooxygenase: cloning, functional expression and characterization of the responsible gene. Arch Biochem Biophys 390:279–286. https://doi.org/10.1006/abbi.2001.2378
Breathnach R, Chambon P (1981) Organization and expression of eukaryotic split genes coding for proteins. Annu Rev Biochem 50: 349–383. https://doi.org/10.1146/annurev.bi.50.070181.002025Buchanan
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buchanan BB, Gruissem W, Jones RL (2000) Biochemistry and molecular biology of plants. American Society of Plant Biologists, Rockville, Maryland
Burke C, Wildung MR, Croteau R (1999) Geranyl diphosphate synthase: cloning, expression, and characterization of this prenyltransferase as a heterodimer. Proc Natl Acad Sci USA 96:13062–13067. https://doi.org/10.1073/pnas.96.23.13062
Canevascini S, Caderas D, Mandel T, Fleming AJ, Dupuis I, Kuhlemeier C (1996) Tissue-specific expression and promoter analysis of the tobacco ltpl gene. Plant Physiol 2(51):3–524
Chambers HL, Hummer KE (1994) Chromosome counts in the Mentha collection at the USDAARS national germplasm repository. Taxon 43:423–432
Chow CN, Lee TY, Hung YC, Li GZ, Tseng KC, Liu YH, Kuo PL, Zheng HQ, Chang WC (2019) PlantPAN30: a new and updated resource for reconstructing transcriptional regulatory networks from ChIP-seq experiments in plants. Nucleic Acids Res 47:D1155–D1163. https://doi.org/10.1093/nar/gky1081
Colby SM, Alonso WR, Katahira E, McGarvey DJ, Croteau R (1993) 4S-Limonene synthase from the oil glands of spearmint (Mentha spicata): cDNA isolation, characterization and bacterial expression of the catalytically active monoterpene cyclase. J Biol Chem 268:23016–23024
Croteau RB, Davis EM, Ringer KL, Wildung MR (2005) (-)-Menthol biosynthesis and molecular genetics. Naturwissenschaften. https://doi.org/10.1007/s00114-005-0055-0
Davis EM, Ringer KL, McConkey ME, Croteau R (2005) Monoterpene metabolism. cloning, expression, and characterization of menthone reductases from peppermint. Plant Physiol 13:873–881. https://doi.org/10.1104/pp.104.053306
Dorman HJ, Kosar M, Kahlos K, Holm Y, Hiltune R (2003) Antioxidant prosperities and composition of aqueous extracts from Mentha species, hybrids, varieties and cultivars. J Agric Food Chem 51:4563–4569. https://doi.org/10.1021/jf034108k
Dubey VS (1999) Metabolism of monoterpenoids in palmarosa (Cymbopogon martini, Roxb. Vats. Var. motia) inflorescence. Ph.D. thesis. University of Lucknow, India
Fahn A (1979) Secretory Tissues in Plants. Academic Press, London, pp 162–164
Featherstone M (2002) Coactivators in transcription initiation. Curr Opin Genet Dev 12:149–155. https://doi.org/10.1016/S0959-437X(02)00280-0
Foster R, Izawa T, Chua NH (1994) Plant bZIP proteins gather at ACGT elements. FASEB J 8(2):192–200. https://doi.org/10.1096/fasebj.8.2.8119490
Gang DR, Lavid N, Zubieta C, Chen F, Beuerle T, Lewinsohn E, Noel JP, Pichersky E (2002) Characterization of phenylpropene O-methyltransferases from sweet basil: facile change of substrate specificity and convergent evolution within a plant O-methyltransferase family. Plant Cell 14:505–519
Gershenzon J, Dudareva N (2007) The function of terpene natural products in the natural world. Nat Chem Biol 3(7):408–414. https://doi.org/10.1038/nchembio.2007.5
Gershenzon J, McCaskill D, Rajaonarivony JIM, Mihaliak C, Karp F, Croteau R (1992) Isolation of secretory cells from plant glandular trichomes and their use in biosynthetic studies of monoterpenes and other gland products. Anal Biochem 200:130–213. https://doi.org/10.1016/0003-2697(92)90288-i
Goldberg ML (1979) Sequence analysis of Drosophila histone genes. Stanford University, California
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA-elements (PLACE). Nucl Acids Res 27:297–300. https://doi.org/10.1093/nar/27.1.297
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Hsu CY, Creech RG, Jenkins JN, Ma DP (1999) Analysis of promoter activity of cotton lipid transfer protein gene Ltp6 in transgenic tobacco plants. Plant Sci 143:63–70. https://doi.org/10.1016/S0168-9452(99)00026-6
Javahery R, Khachi A, Lo K, Zenzie-Gregory B, Smale ST (1994) DNA sequence requirements for transcriptional initiator activity in mammalian cells. Mol Cell Bio 14:116–127. https://doi.org/10.1128/mcb.14.1.116
Jefferson AR, Kavanagh AT, Bevan M (1987) GUS fusions:B-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. The EMBO J 6(13):3901–3907. https://doi.org/10.1002/j.1460-2075.198.tb02730.x
Jiang W, Lu X, Qiu B, Zhang F, Shen Q, Lv Z, Fu X, Yan T, Gao E, Zhu M, Chen L, Zhang L, Wang G, Sun X, Tang K (2014) Molecular cloning and characterization of a trichome-specific promoter of artemisinic aldehyde Δ11(13) reductase (DBR2) in Artemisia annua. Plant Mol Biol Rep 32:82–91
Jindal S, Longchar B, Singh A, Gupta V (2015) Promoters of AaGL2 and AaMIXTA-Like1 gene of Artemisia annua direct reporter gene expression in glandular and non-glandular trichomes. Plant Signal Behav. https://doi.org/10.1080/15592324.2015.1087629
Johnson HB (1975) Plant pubescence: an ecological perspective. Bot Rev 41:233–258
Khanuja SPS, Shasany AK, Darokar MP, Kumar S (1999) Rapid Isolation of DNA from dry and fresh samples of plants producing large amounts secondary metabolites and essential oils. Plant Mol Biol Rept 17:1–7. https://doi.org/10.1023/A:1007528101452
Kim S, Chang YJ, Kim S (2008) Tissue specificity and developmental pattern of amorpha-4,11-diene synthase (ADS) proved by ADS promoter-driven gus expression in the heterologous plant, Arabidopsis thaliana. Planta Med 74:188–193
Kollmar R, Famham PJ (1993) Site-specific initiation of transcription by RNA polymerase II. Proc Soc Exp Biol Med 203:127–139. https://doi.org/10.3181/00379727-203-43583
Larkin J, Oppenheimer D, Pollock S, Marks M (1993) Arabidopsis GLABROUS1 gene requires downstream sequences for function. Plant Cell 5:1739–1748
Larkin JC, Oppenheimer DG, Lloyd A, Paparozzi ET, Marks MD (1994) The roles of GLABROUS1 and TRANSPARENT TESTA GLABRA genes in Arabidopsis trichome development. Plant Cell 6:1065–1076
Lescot M, Déhais P, Thijs G (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucl Acids Res 30:325–327. https://doi.org/10.1093/nar/30.1.325
Liu HC, Creech RG, Jenkins JN, Ma DP (2000) Cloning and promoter analysis of the cotton lipid transfer protein gene Lpt3. Biochim Biophys Acta 1487:106–111. https://doi.org/10.1016/S1388-1981(00)00072-x
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)]. Method 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lupien S, Karp F, Wildung M, Croteau R (1999) Regiospecific cytochrome P450 limonene hydroxylases from mint (Mentha) species: cDNA isolation, characterization, and functional expression of (−)-4S-limonene-3-hydroxylase and (−)-4S-limonene-6-hydroxylase. Arch Biochem Biophys 368:181–192. https://doi.org/10.1006/abbi.1999.1298
Mahmoud SS, Croteau R (2002) Strategies for transgenic manipulation of monoterpene biosynthesis in plants. Trends Plant Sci 7:366–373. https://doi.org/10.1016/S1360-1385
Mauricio R, Rausher MD (1997) Experimental manipulation of putative selective agents provides evidence for the role of natural enemies in the evolution of plant defense. Evolution 51:1435–1444. https://doi.org/10.1111/j.1558-5646.1997.tb01467
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Olsson ME, Olofsson LM, Lindahl AL, Lundgren A, Brodelius M, Brodelius PE (2009) Localization of enzymes of artemisinin biosynthesis to the apical cells of glandular secretory trichomes of Artemisia annua L. Phytochemistry 70(9):1123–1128
Oppenheimer DG, Herman PL, Sivakumaran S, Esch J, Marks MD (1991) A myb gene required for leaf trichome differentiation in Arabidopsis is expressed in stipules. Cell 67(3):483–493
Payne T, Clement J, Arnold D, Lloyd A (1999) Heterologous Myb genes distinct from GL1 enhance trichome production when overexpressed in Nicotiana tabacum. Development 126:671–768
Pyee J, Kolattukudy PE (1995) The gene for the major cuticular wax-associated protein and three homologous genes from broccoli (Brassica oleracea) and their expression patterns. Plant J 71(1):49–59
Rerie WG, Feldmann KA, Marks MD (1994) The GLABRA2 gene encodes a homeo domain protein required for normal trichome development in Arabidopsis. Genes De 8:1388–1399
Ringer KL, McConkey ME, Davis EM, Rushing GW, Croteau R (2003) Monoterpene double-bond reductases of the (-)-menthol biosynthetic pathway: isolation and characterization of cDNAs encoding (-)-isopiperitenone reductase and (1)-pulegone reductase of peppermint. Arch Biochem Biophys 418:80–92. https://doi.org/10.1016/s0003-9861(03)00390-4
Ringer KL, Davis EM, Croteau R (2005) Monoterpene metabolism. cloning, expression, and characterization of (-)-Isopiperitenol/(-)-Carveol dehyrogenase of peppemint and spearmint. Plant Phys 137:863–872. https://doi.org/10.1104/pp.104.053298
Rombauts S, Déhais P, Montagu MV, Rouzé P (1999) PlantCARE, a plant cis-acting regulatory element database. Nucleic Acids Res 27:295–296
Shangguan X, Xu B, Yu ZX, Wang LJ, Chen XY (2008) Promoter of a cotton fibre Myb gene functional in trichomes of Arabidopsis and glandular trichomes of tobacco. J Exp Bot 59(13):3533–3542
Sharma S, Tyagi BR, Naqvi AA, Thakur RS (1992) Stability of essential oil yield and quality characters in japanese mint (Mentha arvensis L.) under varied environmental conditions. J Essent Oil Res 4:411–416
Siberil Y, Doireau P, Gantet P (2001) Plant bZIP Gbox binding factors. Modular structure and activation mechanisms. Eur J Biochem 268 (22): 5655–5666. https://doi.org/10.1046/j.0014-2956.2001.02552.x
Singh N, Luthra R, Sangwan RS, Thakur RS (1989) Metabolism of monoterpenoids in aromatic plants. Curr Res Med Arom Plants 11:174–197
Song YH, Yoo CM, Hong AP, Kim SH, Jeong HJ, Shin SY et al (2008) DNA binding study identifies C-Box and hybrid C/G-Box or C/A Box motifs as high affinity binding sites for STF1 and Long Hypocotyle5 proteins. Plant Physiol 146(4):862–1877. https://doi.org/10.1104/pp.107.113217
Stracke R, Werber M, Weisshaar B (2001) The R2R3-MYB gene family in Arabidopsis thaliana. Curr Opin Plant Biol 4:447–456. https://doi.org/10.1016/S1369-5266(00)00199-0
Turner GW, Croteau R (2004) Organization of monoterpene biosynthesis in Mentha: immunocytochemical localization of geranyl diphosphate synthase, limonene-6-hydroxylase, isopiperitenol dehydrogenase, and pulegone reductase. Plant Physiol 136:4215–4227. https://doi.org/10.1104/pp.104.050229
Vining KJ, Johnson SR, Ahkami A, Lange I, Parrish AN, Trapp SC, Croteau RB, Straub SCK, Pandelova I, Lange BM (2017) Draft genome sequence of Mentha longifolia and development of resources for mint cultivar improvement. Mol Plant 10:323–339
Wang E, Gan S, Wagner GJ (2002) Isolation and characterization of the CYP71D16 trichome specific promoter from Nicotiana tabaccum L. J Exp Bot 53(376):1891–1897. https://doi.org/10.1093/jxb/erf054
Wang H, Han J, Kanagarajan S, Lundgren A, Brodelius PE (2013) Trichome-specific expression of the amorpha-4,11-diene 12-hydroxylase (cyp71av1) gene, encoding a key enzyme of artemisinin biosynthesis in Artemisia annua, as reported by a promoter-GUS fusion. Plant Mol Biol 81:119–138
Wang C, Wang Y, Pan Q, Chen S, Feng C, Hai J, Li H (2019) Comparison of Trihelix transcription factors between wheat and Brachypodium distachyon at genome-wide. BMC Genomics. https://doi.org/10.1186/s12864-019-5494-7
Weis L, Reinberg D (1992) Transcription by RNA polymerase II: Initiator-directed formation of transcription-competent complexes. FASEB 6:3300–3309. https://doi.org/10.1096/fasebj.6.14.1426767
Yan D, Zhang X, Zhang L, Ye S, Zeng L, Liu J (2015) CURVED CHIMERIC PALEA 1 encoding an EMF1-like protein maintains epigenetic repression of OsMADS58 in rice palea development. Plant J 82(1):12–24. https://doi.org/10.1111/tpj.12784
Zimmermann G, Furlong EE, Suyama K, Scott MP (2006) Mes2, a MADF-containing transcription factor essential for Drosophila development. Dev Dyn 235(12):3387–3395. https://doi.org/10.1002/dvdy.20970
Acknowledgements
We thank Director, CSIR-CIMAP for providing necessary facilities, and Dr Vikrant Gupta for providing binary vector plasmid pBI101 for cloning. The research work was financially supported by the Department of Science and Technology (DST), Govt. of India, under project GAP252 and Council of Scientific and industrial research (CSIR), Govt. of India, under project (BSC0203). NQ received research fellowships from CSIR, Govt. of India, and her PhD degree from AcSIR, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest in this work and have nothing to disclose.
Additional information
Communicated by Anastasios Melis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qamar, N., Pandey, M., Vasudevan, M. et al. Glandular trichome specificity of menthol biosynthesis pathway gene promoters from Mentha × piperita. Planta 256, 110 (2022). https://doi.org/10.1007/s00425-022-04029-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-022-04029-4