Abstract
Purpose
To explore the role of choroidopathy in diabetic retinopathy (DR) by investigating the correlation between alterations of choroidal vessel and photoreceptors during the early stage of DR.
Methods
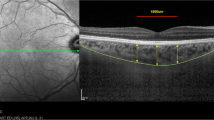
We performed a cross-sectional comparison of diabetic patients without DR (NDR group; n=16) and those with mild nonproliferative diabetic retinopathy (NPDR group; n=39). Optical coherence tomography (OCT) images of choroidal vessel alterations and photoreceptor structures were evaluated using the choroidal vascularity index (CVI) and adjusted ellipsoid zone (EZ) reflectivity, respectively. To evaluate the function of cone photoreceptors, the fundamental, harmonic amplitudes, the parameters S and Rmp3 were calculated from the electroretinogram (ERG). These factors were compared between groups. The correlation between the CVI and parameters describing the function and structure of the photoreceptors was evaluated.
Results
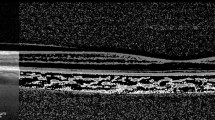
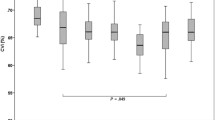
The significant decrease was observed in the CVI in the NPDR group compared to the NDR group (0.67 ± 0.04 vs. 0.70 ± 0.06; p = 0.028), but not in the adjusted EZ reflectivity or ERG parameters. In NPDR group and merging the 2 groups, CVI was moderately positively correlated with the fundamental amplitude obtained by the flicker ERG (NPDR only: r = 0.506; p = 0.001; merge the 2 groups: r = 0.423; p = 0.001), which was regulated by the response of the cone photoreceptors. The CVI was positively and moderately correlated with the logS (NPDR only: r = 0.462; p = 0.003; merge the 2 groups: r = 0.355; p = 0.008), indicating the sensitivity of cone cell light transduction.
Conclusion
Compared to eyes without DR, CVI decreased representing choroidal vascular changes in eyes with mild NPDR. These changes may be related to the functional impairment of cone photoreceptors, especially phototransduction sensitivity, as the DR develops.




Similar content being viewed by others
References
Assi L, Chamseddine F, Ibrahim P, Sabbagh H, Rosman L, Congdon N, Evans J, Ramke J, Kuper H, Burton MJ, Ehrlich JR, Swenor BK (2021) A global assessment of eye health and quality of life: a systematic review of systematic reviews. JAMA Ophthalmol 139:526–541. https://doi.org/10.1001/jamaophthalmol.2021.0146
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet (London, England) 376:124–136. https://doi.org/10.1016/s0140-6736(09)62124-3
Cao J, McLeod S, Merges CA, Lutty GA (1998) Choriocapillaris degeneration and related pathologic changes in human diabetic eyes. Arch Ophthalmol 116:589–597. https://doi.org/10.1001/archopht.116.5.589
Murakami T, Uji A, Suzuma K, Dodo Y, Yoshitake S, Ghashut R, Yoza R, Fujimoto M, Yoshimura N (2016) In vivo choroidal vascular lesions in diabetes on swept-source optical coherence tomography. PloS One 11:e0160317. https://doi.org/10.1371/journal.pone.0160317
Li L, Almansoob S, Zhang P, Zhou YD, Tan Y, Gao L (2019) Quantitative analysis of retinal and choroid capillary ischaemia using optical coherence tomography angiography in type 2 diabetes. Acta Ophthalmol 97:240–246. https://doi.org/10.1111/aos.14076
Betzler BK, Ding J, Wei X, Lee JM, Grewal DS, Fekrat S, Sadda SR, Zarbin MA, Agarwal A, Gupta V, Schmetterer L, Agrawal R (2022) Choroidal vascularity index: a step towards software as a medical device. Br J Ophthalmol 106:149–155. https://doi.org/10.1136/bjophthalmol-2021-318782
Gupta C, Tan R, Mishra C, Khandelwal N, Raman R, Kim R, Agrawal R, Sen P (2018) Choroidal structural analysis in eyes with diabetic retinopathy and diabetic macular edema-a novel OCT based imaging biomarker. PloS One 13:e0207435. https://doi.org/10.1371/journal.pone.0207435
Foo VHX, Gupta P, Nguyen QD, Chong CCY, Agrawal R, Cheng CY, Yanagi Y (2020) Decrease in choroidal vascularity index of Haller’s layer in diabetic eyes precedes retinopathy. BMJ Open Diabetes Res Care 8. https://doi.org/10.1136/bmjdrc-2020-001295
Hood DC, Birch DG (1995) Phototransduction in human cones measured using the alpha-wave of the ERG. Vision Res 35:2801–2810. https://doi.org/10.1016/0042-6989(95)00034-w
McAnany JJ, Chen YF, Liu K, Park JC (2019) Nonlinearities in the flicker electroretinogram: a tool for studying retinal dysfunction applied to early-stage diabetic retinopathy. Vision Res 161:1–11. https://doi.org/10.1016/j.visres.2019.05.005
Alexander KR, Barnes CS, Fishman GA (2001) Origin of deficits in the flicker electroretinogram of the cone system in X-linked retinoschisis as derived from response nonlinearities. J Opt Soc Am A Opt Image Sci Vis 18:747–754. https://doi.org/10.1364/josaa.18.000747
Sonoda S, Sakamoto T, Yamashita T, Uchino E, Kawano H, Yoshihara N, Terasaki H, Shirasawa M, Tomita M, Ishibashi T (2015) Luminal and stromal areas of choroid determined by binarization method of optical coherence tomographic images. Am J Ophthalmol 159:1123–1131.e1121. https://doi.org/10.1016/j.ajo.2015.03.005
Zeng Y, Cao D, Yang D, Zhuang X, Yu H, Hu Y, Zhang Y, Yang C, He M, Zhang L (2020) Screening for diabetic retinopathy in diabetic patients with a mydriasis-free, full-field flicker electroretinogram recording device. Doc Ophthalmol 140:211–220. https://doi.org/10.1007/s10633-019-09734-2
Değirmenci MFK, Demirel S, Batıoğlu F, Özmert E (2018) Role of a mydriasis-free, full-field flicker ERG device in the detection of diabetic retinopathy. Doc Ophthalmol 137:131–141. https://doi.org/10.1007/s10633-018-9656-8
McAnany JJ, Park JC (2019) Cone photoreceptor dysfunction in early-stage diabetic retinopathy: association between the activation phase of cone phototransduction and the flicker electroretinogram. Invest Ophthalmol Vis Sci 60:64–72. https://doi.org/10.1167/iovs.18-25946
Agrawal R, Gupta P, Tan KA, Cheung CM, Wong TY, Cheng CY (2016) Choroidal vascularity index as a measure of vascular status of the choroid: measurements in healthy eyes from a population-based study. Sci Rep 6:21090. https://doi.org/10.1038/srep21090
Cuenca N, Ortuño-Lizarán I, Sánchez-Sáez X, Kutsyr O, Albertos-Arranz H, Fernández-Sánchez L, Martínez-Gil N, Noailles A, López-Garrido JA, López-Gálvez M, Lax P, Maneu V, Pinilla I (2020) Interpretation of OCT and OCTA images from a histological approach: clinical and experimental implications. Prog Retin Eye Res 77:100828. https://doi.org/10.1016/j.preteyeres.2019.100828
Flores M, Debellemanière G, Bully A, Meillat M, Tumahai P, Delbosc B, Saleh M (2015) Reflectivity of the outer retina on spectral-domain optical coherence tomography as a predictor of photoreceptor cone density. Am J Ophthalmol 160:588–595.e582. https://doi.org/10.1016/j.ajo.2015.06.008
Toprak I, Yildirim C, Yaylali V (2015) Impaired photoreceptor inner segment ellipsoid layer reflectivity in mild diabetic retinopathy. Can J Ophthalmol 50:438–441. https://doi.org/10.1016/j.jcjo.2015.07.009
Guyon B, Elphege E, Flores M, Gauthier AS, Delbosc B, Saleh M (2017) Retinal reflectivity measurement for cone impairment estimation and visual assessment after diabetic macular edema resolution (RECOVER-DME). Invest Ophthalmol Vis Sci 58:6241–6247. https://doi.org/10.1167/iovs.17-22380
Kessler LJ, Auffarth GU, Bagautdinov D, Khoramnia R (2021) Ellipsoid zone integrity and visual acuity changes during diabetic macular edema therapy: a longitudinal study. J Diabetes Res 2021:8117650. https://doi.org/10.1155/2021/8117650
Ro-Mase T, Ishiko S, Omae T, Ishibazawa A, Shimouchi A, Yoshida A (2020) Association between alterations of the choriocapillaris microcirculation and visual function and cone photoreceptors in patients with diabetes. Invest Ophthalmol Vis Sci 61:1. https://doi.org/10.1167/iovs.61.6.1
McAnany JJ, Park JC (2018) Temporal frequency abnormalities in early-stage diabetic retinopathy assessed by electroretinography. Invest Ophthalmol Vis Sci 59:4871–4879. https://doi.org/10.1167/iovs.18-25199
Xu F, Li Z, Yang X, Gao Y, Li Z, Li G, Wang S, Ning X, Li J (2022) Assessment of choroidal structural changes in patients with pre- and early-stage clinical diabetic retinopathy using wide-field SS-OCTA. Front Endocrinol 13:1036625. https://doi.org/10.3389/fendo.2022.1036625
Author information
Authors and Affiliations
Contributions
D.N.X. designed the work, performed data analysis, and written the manuscript. L.G.Y. maintained research data and was a major contributor in data analysis. F.D. was responsible for project administration and data acquisition. Z.S.C. was responsible for supervision of the study. L.X.L. was responsible for supervision of the study and funding acquisition. Y.S.S. was responsible for validation and supervision of the study. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethics Committee of the Zhongshan Ophthalmic Center, Sun Yat-sen University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dou, N., Li, G., Fang, D. et al. Association between choroidopathy and photoreceptors during the early stage of diabetic retinopathy: a cross-sectional study. Graefes Arch Clin Exp Ophthalmol 262, 1121–1129 (2024). https://doi.org/10.1007/s00417-023-06282-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-023-06282-z