Abstract
Background
Vestibular migraine (VM) and Menière’s disease (MD) are two common causes of recurrent spontaneous vertigo. Using history, video-nystagmography and audiovestibular tests, we developed machine learning models to separate these two disorders.
Methods
We recruited patients with VM or MD from a neurology outpatient facility. One hundred features from six “feature subsets”: history, acute video-nystagmography and four laboratory tests (video head impulse test, vestibular-evoked myogenic potentials, caloric testing and audiogram) were used. We applied ten machine learning algorithms to develop classification models. Modelling was performed using three “tiers” of data availability to simulate three clinical settings. “Tier 1” used all available data to simulate the neuro-otology clinic, “Tier 2” used only history, audiogram and caloric test data, representing the general neurology clinic, and “Tier 3” used history alone as occurs in primary care. Model performance was evaluated using tenfold cross-validation.
Results
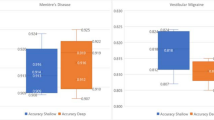
Data from 160 patients with VM and 114 with MD were used for model development. All models effectively separated the two disorders for all three tiers, with accuracies of 85.77–97.81%. The best performing algorithms (AdaBoost and Random Forest) yielded accuracies of 97.81% (95% CI 95.24–99.60), 94.53% (91.09–99.52%) and 92.34% (92.28–96.76%) for tiers 1, 2 and 3. The best feature subset combination was history, acute video-nystagmography, video head impulse test and caloric testing, and the best single feature subset was history.
Conclusions
Machine learning models can accurately differentiate between VM and MD and are promising tools to assist diagnosis by medical practitioners with diverse levels of expertise and resources.




Similar content being viewed by others
Data availability
The dataset used for this study contains patient health data and is not publicly available for privacy reasons. A deidentified version of dataset is available on reasonable request from the corresponding author M.W. The data will be shared through a data sharing agreement. With this mediated access, the data are FAIR compliant.
References
Australian Bureau of Statistics (2021) Population: Census. https://www.abs.gov.au/statistics/people/population/population-census/latest-release. Accessed 14 Aug 2022
Baier B, Stieber N, Dieterich M (2009) Vestibular-evoked myogenic potentials in vestibular migraine. J Neurol 256:1447–1454. https://doi.org/10.1007/s00415-009-5132-4
Breiman L (2001) Random forests. Mach Learn 45:5–32. https://doi.org/10.1023/A:1010933404324
Chen T, Guestrin C (2016) XGBoost: A scalable tree boosting system. In: Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining. Association for Computing Machinery, San Francisco, California, USA, pp 785–794
Colebatch JG, Halmagyi GM, Skuse NF (1994) Myogenic potentials generated by a click-evoked vestibulocollic reflex. J Neurol Neurosurg Psychiatry 57:190–197. https://doi.org/10.1136/jnnp.57.2.190
Cortes C, Vapnik V (1995) Support-vector networks. Mach Learn 20:273–297. https://doi.org/10.1007/BF00994018
Crammer K, Dekel O, Keshet J, Shalev-Shwartz S, Singer Y (2006) Online passive-aggressive algorithms. J Mach Learn Res 7:551–585
Dieterich M, Obermann M, Celebisoy N (2016) Vestibular migraine: the most frequent entity of episodic vertigo. J Neurol 263:82–89. https://doi.org/10.1007/s00415-015-7905-2
Exarchos TP, Rigas G, Bibas A, Kikidis D, Nikitas C, Wuyts FL, Ihtijarevic B, Maes L, Cenciarini M, Maurer C, Macdonald N, Bamiou DE, Luxon L, Prasinos M, Spanoudakis G, Koutsouris DD, Fotiadis DI (2016) Mining balance disorders’ data for the development of diagnostic decision support systems. Comput Biol Med 77:240–248. https://doi.org/10.1016/j.compbiomed.2016.08.016
Friedman JH (2001) Greedy function approximation: a gradient boosting machine. Ann Stat 29(1189–1232):1144
Groezinger M, Huppert D, Strobl R, Grill E (2020) Development and validation of a classification algorithm to diagnose and differentiate spontaneous episodic vertigo syndromes: results from the DizzyReg patient registry. J Neurol 267:160–167. https://doi.org/10.1007/s00415-020-10061-9
Gürkov R, Jerin C, Flatz W, Maxwell R (2019) Clinical manifestations of hydropic ear disease (Menière’s). Eur Arch Otorhinolaryngol 276:27–40. https://doi.org/10.1007/s00405-018-5157-3
Huang CH, Wang SJ, Young YH (2011) Localization and prevalence of hydrops formation in Ménière’s disease using a test battery. Audiol Neurootol 16:41–48. https://doi.org/10.1159/000312199
Inoue A, Egami N, Fujimoto C, Kinoshita M, Yamasoba T, Iwasaki S (2016) Vestibular evoked myogenic potentials in vestibular migraine: do they help differentiating from Menière’s disease? Ann Otol Rhinol Laryngol 125:931–937. https://doi.org/10.1177/0003489416665192
Jongkees LB, Maas JP, Philipszoon AJ (1962) Clinical nystagmography. A detailed study of electro-nystagmography in 341 patients with vertigo. Pract Otorhinolaryngol (Basel) 24:65–93
Kabade V, Hooda R, Raj C, Awan Z, Young AS, Welgampola MS, Prasad M (2021) Machine learning techniques for differential diagnosis of vertigo and dizziness: a review. Sensors 21:7565
Lempert T, Olesen J, Furman J, Waterston J, Seemungal B, Carey J, Bisdorff A, Versino M, Evers S, Newman-Toker D (2012) Vestibular migraine: diagnostic criteria. J Vestib Res 22:167–172. https://doi.org/10.3233/VES-2012-0453
Lopez-Escamez JA, Carey J, Chung W-H, Goebel JA, Magnusson M, Mandalà M, Newman-Toker DE, Strupp M, Suzuki M, Trabalzini F, Bisdorff A (2015) Diagnostic criteria for Menière’s disease. J Vestib Res 25:1–7. https://doi.org/10.3233/VES-150549
Lopez-Escamez JA, Dlugaiczyk J, Jacobs J, Lempert T, Teggi R, von Brevern M, Bisdorff A (2014) Accompanying symptoms overlap during attacks in Menière’s disease and vestibular migraine. Front Neurol. https://doi.org/10.3389/fneur.2014.00265
MacDougall HG, McGarvie LA, Halmagyi GM, Curthoys IS, Weber KP (2013) The video head impulse test (vHIT) detects vertical semicircular canal dysfunction. PLoS ONE 8:e61488. https://doi.org/10.1371/journal.pone.0061488
MacDougall HG, Weber KP, McGarvie LA, Halmagyi GM, Curthoys IS (2009) The video head impulse test: diagnostic accuracy in peripheral vestibulopathy. Neurology 73:1134–1141. https://doi.org/10.1212/WNL.0b013e3181bacf85
Medical Board of Australia (2021) Medical Board of Australia Registrant Data—reporting Period: 01 October 2021 to 31 December 2021. Melbourne, Australia. https://www.ahpra.gov.au/documents/default.aspx?record=WD22%2f31646&dbid=AP&chksum=ccbe7hvigrjZ4H64z42Aaw%3d%3d. Accessed 14 Aug 2022
Muelleman T, Shew M, Subbarayan R, Shum A, Sykes K, Staecker H, Lin J (2017) Epidemiology of Dizzy patient population in a neurotology clinic and predictors of peripheral etiology. Otol Neurotol 38:870–875. https://doi.org/10.1097/mao.0000000000001429
Neff BA, Staab JP, Eggers SD, Carlson ML, Schmitt WR, Van Abel KM, Worthington DK, Beatty CW, Driscoll CL, Shepard NT (2012) Auditory and vestibular symptoms and chronic subjective dizziness in patients with Ménière’s disease, vestibular migraine, and Ménière’s disease with concomitant vestibular migraine. Otol Neurotol 33:1235–1244. https://doi.org/10.1097/MAO.0b013e31825d644a
Neuhauser HK, von Brevern M, Radtke A, Lezius F, Feldmann M, Ziese T, Lempert T (2005) Epidemiology of vestibular vertigo. A neurotologic survey of the general population 65:898–904. https://doi.org/10.1212/01.wnl.0000175987.59991.3d
Nham B, Reid N, Bein K, Bradshaw A, McGarvie L, Argaet E, Young A, Watson S, Halmagyi G, Black D, Welgampola M (2021) Capturing vertigo in the emergency room: three tools to double the rate of diagnosis. J Neurol. https://doi.org/10.1007/s00415-021-10627-1
Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, Blondel M, Prettenhofer P, Weiss R, Dubourg V, Vanderplas J, Passos A, Cournapeau D, Brucher M, Perrot M, Duchesnay É (2011) Scikit-learn: machine learning in Python. J Mach Learn Res 12:2825–2830
Peterson LE (2009) K-nearest neighbor. Scholarpedia 4:1883. https://doi.org/10.4249/scholarpedia.188
Polensek SH, Tusa RJ (2010) Nystagmus during attacks of vestibular migraine: an aid in diagnosis. Audiol Neurotol 15:241–246. https://doi.org/10.1159/000255440
Quinlan JR (1996) Learning decision tree classifiers. ACM Comput Surv 28:71–72. https://doi.org/10.1145/234313.234346
Radtke A, Lempert T, Gresty MA, Brookes GB, Bronstein AM, Neuhauser H (2002) Migraine and Ménière’s disease: is there a link? Neurology 59:1700–1704. https://doi.org/10.1212/01.wnl.0000036903.22461.39
Radtke A, Neuhauser H, von Brevern M, Hottenrott T, Lempert T (2011) Vestibular migraine–validity of clinical diagnostic criteria. Cephalalgia 31:906–913. https://doi.org/10.1177/0333102411405228
Rauch SD, Zhou G, Kujawa SG, Guinan JJ, Herrmann BS (2004) Vestibular evoked myogenic potentials show altered tuning in patients with Ménière’s disease. Otol Neurotol 25:333–338. https://doi.org/10.1097/00129492-200405000-00022
Rosenblatt F (1961) Principles of neurodynamics. perceptrons and the theory of brain mechanisms. In:Cornell Aeronautical Lab Inc Buffalo NY
Rosengren SM, McAngus Todd NP, Colebatch JG (2005) Vestibular-evoked extraocular potentials produced by stimulation with bone-conducted sound. Clin Neurophysiol 116:1938–1948. https://doi.org/10.1016/j.clinph.2005.03.019
Schapire RE (2013) Explaining adaboost. Empirical inference. Springer, Berlin, pp 37–52
Taylor RL, Zagami AS, Gibson WPR, Black DA, Watson SRD, Halmagyi MG, Welgampola MS (2012) Vestibular evoked myogenic potentials to sound and vibration: characteristics in vestibular migraine that enable separation from Menière’s disease. Cephalalgia 32:213–225. https://doi.org/10.1177/0333102411434166
Vivar G, Strobl R, Grill E, Navab N, Zwergal A, Ahmadi SA (2021) Using base-ml to learn classification of common vestibular disorders on DizzyReg Registry data. Front Neurol 12:681140. https://doi.org/10.3389/fneur.2021.681140
von Brevern M, Zeise D, Neuhauser H, Clarke AH, Lempert T (2004) Acute migrainous vertigo: clinical and oculographic findings. Brain 128:365–374. https://doi.org/10.1093/brain/awh351
Wright RE (1995) Logistic regression. Reading and understanding multivariate statistics. American Psychological Association, Washington, DC, pp 217–244
Young AS, Lechner C, Bradshaw AP, MacDougall HG, Black DA, Halmagyi GM, Welgampola MS (2019) Capturing acute vertigo. A vestibular event monitor 92:e2743–e2753. https://doi.org/10.1212/wnl.0000000000007644
Young AS, Nham B, Bradshaw AP, Calic Z, Pogson JM, D’Souza M, Halmagyi GM, Welgampola MS (2021) Clinical, oculographic, and vestibular test characteristics of vestibular migraine. Cephalalgia 41:1039–1052. https://doi.org/10.1177/03331024211006042
Young AS, Nham B, Bradshaw AP, Calic Z, Pogson JM, Gibson WP, Halmagyi GM, Welgampola MS (2021) Clinical, oculographic and vestibular test characteristics of Ménière’s disease. J Neurol. https://doi.org/10.1007/s00415-021-10699-z
Zhang Y, Kong Q, Chen J, Li L, Wang D, Zhou J (2016) International Classification of Headache Disorders 3rd edition beta-based field testing of vestibular migraine in China: Demographic, clinical characteristics, audiometric findings and diagnosis statues. Cephalalgia 36:240–248. https://doi.org/10.1177/0333102415587704
Funding
This work was supported by funding from the Garnett Passe and Rodney Williams Memorial Foundation (Grant 2021_RS_Wang).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests.
Ethics approval
This study was approved by the Sydney Local Health District Ethics Committee (Protocol No X21-0295) and performed in accordance with the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Written informed consent was obtained from all participants.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, C., Young, A.S., Raj, C. et al. Machine learning models help differentiate between causes of recurrent spontaneous vertigo. J Neurol (2024). https://doi.org/10.1007/s00415-023-11997-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00415-023-11997-4