Abstract
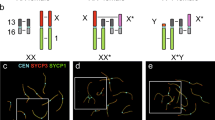
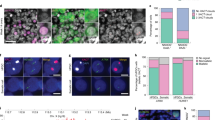
Although parthenogenesis is widespread in nature and known to have close relationships with bisexuality, the transitional mechanism is poorly understood. Artemia is an ideal model to address this issue because bisexuality and “contagious” obligate parthenogenesis independently exist in its congeneric members. In the present study, we first performed chromosome spreading and immunofluorescence to compare meiotic processes of Artemia adopting two distinct reproductive ways. The results showed that, unlike conventional meiosis in bisexual Artemia, meiosis II in parthenogenic Artemia is entirely absent and anaphase I is followed by a single mitosis-like equational division. Interspecific comparative transcriptomics showed that two central molecules in homologous recombination (HR), Dmc1 and Rad51, exhibited significantly higher expression in bisexual versus parthenogenetic Artemia. qRT-PCR indicated that the expression of both genes peaked at the early oogenesis and gradually decreased afterward. Knocking-down by RNAi of Dmc1 in unfertilized females of bisexual Artemia resulted in a severe deficiency of homologous chromosome pairing and produced univalents at the middle oogenesis stage, which was similar to that of parthenogenic Artemia, while in contrast, silencing Rad51 led to no significant chromosome morphological change. Our results indicated that Dmc1 is vital for HR in bisexual Artemia, and the deficiency of Dmc1 may be correlated with or even possibly one of core factors in the transition from bisexuality to parthenogenesis.





Similar content being viewed by others
Data availability
All data are included in this manuscript and in its supplementary materials, or submitted to public databases. The RNA-seq data are available in the NCBI Sequence Read Archive (SRA) database (SUB12209975). The nucleotide sequences of Dmc1 and Rad51 have been deposited in the GenBank/DDBJ/EMBL databases (OP773875 and OP773876).
References
Abatzopoulos TJ, Kastritsis CD, Triantaphyllidis CD (1986) A study of karyotypes and heterochromatic associations in Artemia, with special reference to 2n-Greek populations. Genetica 71:3–10. https://doi.org/10.1007/BF00123227
Abatzopoulos TJ, Beardmore JA, Clegg JS, Sorgeloos P (2002) Artemia: basic and applied biology. Kluwer Academic Publishers, Netherlands. https://doi.org/10.1007/978-94-017-0791-6
Archetti M (2010) Complementation, genetic conflict, and the evolution of sex and recombination. J Hered 101(Suppl 1):S21-33. https://doi.org/10.1093/jhered/esq009
Artom C (1907) La maturazione, la fecondazione, e i primi stadi di sviluppo dell’uovo della Artemia salina L. di Cagliari. Biologica Torino 1:495–515
Asem A, Sun SC (2016) Morphological differentiation of seven parthenogenetic Artemia (Crustacea: Branchiopoda) populations from China, with special emphasis on ploidy degrees. Microsc Res Tech 79:258–266. https://doi.org/10.1002/jemt.22625
Asher JH Jr (1970) Parthenogenesis and genetic variability. II. One-locus models for various diploid populations. Genetics 66:369–391. https://doi.org/10.1093/genetics/66.2.369
Barigozzi C (1941) I fenomeni cromosomici Delle cellule germinali in Artemia salina Leach. Chromosoma 2:549–575
Baudry E, Kryger P, Allsopp M, Koeniger N, Vautrin D, Mougel F, Cornuet JM, Solignac M (2004) Whole-genome scan in thelytokous-laying workers of the Cape honeybee (Apis mellifera capensis): central fusion, reduced recombination rates and centromere mapping using half-tetrad analysis. Genetics 167:243–252. https://doi.org/10.1534/genetics.167.1.243
Baxevanis AD, Kappas I, Abatzopoulos TJ (2006) Molecular phylogenetics and asexuality in the brine shrimp Artemia. Mol Phylogenet Evol 40:724–738. https://doi.org/10.1016/j.ympev.2006.04.010
Becks L, Agrawal AF (2012) The evolution of sex is favoured during adaptation to new environments. PLoS Biol 10:e1001317. https://doi.org/10.1371/journal.pbio.1001317
Bishop DK, Park D, Xu L, Kleckner N (1992) DMC1: a meiosis-specific yeast homolog of E. coli recA required for recombination, synaptonemal complex formation, and cell cycle progression. Cell 69:439–456. https://doi.org/10.1016/0092-8674(92)90446-J
Brown MS, Bishop DK (2014) DNA strand exchange and RecA homologs in meiosis. Cold Spring Harb Perspect Biol 7:a0116659. https://doi.org/10.1101/cshperspect.a016659
Browne RA (1992) Population genetics and ecology of Artemia: insights into parthenogenetic reproduction. Trends Ecol Evol 7:232–237. https://doi.org/10.1016/0169-5347(92)90051-C
Chan YL, Bishop DK (2018) Purification of Saccharomyces cerevisiae homologous recombination proteins Dmc1 and Rdh54/Tid1 and a fluorescent D-Loop assay. Meth Enzymol 600:307–320. https://doi.org/10.1016/bs.mie.2017.12.003
Chun J, Buechelmaier ES, Powell SN (2013) Rad51 paralog complexes BCDX2 and CX3 act at different stages in the BRCA1-BRCA2-dependent homologous recombination pathway. Mol Cell Biol 33:387–395. https://doi.org/10.1128/MCB.00465-12
Cloud V, Chan YL, Grubb J, Budke B, Bishop DK (2012) Rad51 is an accessory factor for Dmc1-mediated joint molecule formation during meiosis. Science 337:1222–1225. https://doi.org/10.1126/science.1219379
Cuellar O (1977) Animal parthenogenesis. Science 197:837–843. https://doi.org/10.1126/science.887925
Dai JQ, Zhu XJ, Liu FQ, Xiang JH, Nagasawa H, Yang WJ (2008) Involvement of p90 ribosomal S6 kinase in termination of cell cycle arrest during development of Artemia-encysted embryos. J Biol Chem 283:1705–1712. https://doi.org/10.1074/jbc.M707853200
Dale B, Marino M, Wilding M (1999) The ins and outs of meiosis. J Exp Biol 285:226–236. https://doi.org/10.1002/(SICI)1097-010X(19991015)285:3<226::AID-JEZ5>3.3.CO;2-Q
Decaestecker E, De Meester L, Mergeay J (2009) Cyclical parthenogenesis in Daphnia: sexual versus asexual reproduction. In: Schön I, Martens K, Dijk P (eds) Lost sex: the evolutionary biology of parthenogenesis. Springer Netherlands, Dordrecht, pp, 295–316. https://doi.org/10.1007/978-90-481-2770-2_15
D’Erfurth I, Jolivet S, Froger N, Catrice O, Novatchkova M, Mercier R (2009) Turning meiosis into mitosis. PLoS Biol 7:e1000124. https://doi.org/10.1371/journal.pbio.1000124
Domes K, Norton RA, Maraun M, Scheu S (2007) Reevolution of sexuality breaks Dollo’s law. Proc Natl Acad Sci USA 104:7139–7144. https://doi.org/10.1073/pnas.0700034104
Duncan EJ, Leask MP, Dearden PK (2013) The pea aphid (Acyrthosiphon pisum) genome encodes two divergent early developmental programs. Dev Biol 377:262–274. https://doi.org/10.1016/j.ydbio.2013.01.036
Eads BD, Tsuchiya D, Andrews J, Lynch M, Zolan ME (2012) The spread of a transposon insertion in Rec8 is associated with obligate asexuality in Daphnia. Proc Natl Acad Sci U S A 109:858–863. https://doi.org/10.1073/pnas.1119667109
Eimanifar A, Van Stappen G, Wink M (2015) Geographical distribution and evolutionary divergence times of Asian populations of the brine shrimp Artemia (Crustacea, Anostraca). Zool J Linn Soc 174:447–458. https://doi.org/10.1111/zoj.12242
Engelstadter J (2008) Constraints on the evolution of asexual reproduction. BioEssays 30:1138–1150. https://doi.org/10.1002/bies.20833
Grelon M, Vezon D, Gendrot G, Pelletier G (2001) AtSPO11-1 is necessary for efficient meiotic recombination in plants. EMBO J 20:589–600. https://doi.org/10.1093/emboj/20.3.589
Haag CR, Theodosiou L, Zahab R, Lenormand T (2017) Low recombination rates in sexual species and sex-asex transitions. Philos Trans R Soc Lond B Biol Sci 372:20160461. https://doi.org/10.1098/rstb.2016.0461
Heethoff M, Domes K, Laumann M, Maraun M, Norton RA, Scheu S (2007) High genetic divergences indicate ancient separation of parthenogenetic lineages of the oribatid mite Platynothrus peltifer (Acari, Oribatida). J Evol Biol 20:392–402. https://doi.org/10.1111/j.1420-9101.2006.01183.x
Hiruta C, Nishida C, Tochinai S (2010) Abortive meiosis in the oogenesis of parthenogenetic Daphnia pulex. Chromosome Res 18:833–840. https://doi.org/10.1007/s10577-010-9159-2
Hodges CA, Hunt PA (2002) Simultaneous analysis of chromosomes and chromosome-associated proteins in mammalian oocytes and embryos. Chromosoma 111:165–169. https://doi.org/10.1007/s00412-002-0195-3
Howard-Till RA, Lukaszewicz A, Loidl J (2011) The recombinases Rad51 and Dmc1 play distinct roles in DNA break repair and recombination partner choice in the meiosis of Tetrahymena. PLoS Genet 7:e1001359. https://doi.org/10.1371/journal.pgen.1001359
Hunter N (2015) Meiotic recombination: the essence of heredity. Cold Spring Harb Perspect Biol 7:a016618. https://doi.org/10.1101/cshperspect.a016618
Huylmans AK, Macon A, Hontoria F, Vicoso B (2021) Transitions to asexuality and evolution of gene expression in Artemia brine shrimp. Proc Biol Sci 288:20211720. https://doi.org/10.1098/rspb.2021.1720
Hwang GH, Hopkins JL, Jordan PW (2018) Chromatin spread preparations for the analysis of mouse oocyte progression from prophase to metaphase II. J Vis Exp 132:e56736. https://doi.org/10.3791/56736
Kato M, Hiruta C, Tochinai S (2016) The behavior of chromosomes during parthenogenetic oogenesis in marmorkrebs Procambarus fallax f. virginalis. Zool Sci 33:426–430. https://doi.org/10.2108/zs160018
Lewis SC, Dyal LA, Hilburn CF, Weitz S, Liau WS, Lamunyon CW, Denver DR (2009) Molecular evolution in Panagrolaimus nematodes: origins of parthenogenesis, hermaphroditism and the Antarctic species P davidi. BMC Evol Biol 9:15. https://doi.org/10.1186/1471-2148-9-15
Liu Y-L, Zhao Y, Dai Z-M, Chen H-M, Yang W-J (2009) Formation of diapause cyst shell in brine shrimp, Artemia parthenogenetica, and its resistance role in environmental stresses. J Biol Chem 284:16931–16938. https://doi.org/10.1074/jbc.M109.004051
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Maccari M, Amat F, Gomez A (2013a) Origin and genetic diversity of diploid parthenogenetic Artemia in Eurasia. PLoS One 8:e83348. https://doi.org/10.1371/journal.pone.0083348
Maccari M, Gomez A, Hontoria F, Amat F (2013b) Functional rare males in diploid parthenogenetic Artemia. J Evol Biol 26:1934–1948. https://doi.org/10.1111/jeb.12191
Maccari M, Amat F, Hontoria F, Gomez A (2014) Laboratory generation of new parthenogenetic lineages supports contagious parthenogenesis in Artemia. PEERJ 2:e439. https://doi.org/10.7717/peerj.439
Maniatsi S, Baxevanis AD, Kappas I, Deligiannidis P, Triantafyllidis A, Papakostas S, Bougiouklis D, Abatzopoulos TJ (2011) Is polyploidy a persevering accident or an adaptive evolutionary pattern? The case of the brine shrimp Artemia. Mol Phylogenet Evol 58:353–364. https://doi.org/10.1016/j.ympev.2010.11.029
Martin P, Thonagel S, Scholtz G (2016) The parthenogenetic Marmorkrebs (Malacostraca: Decapoda: Cambaridae) is a triploid organism. J Zoolog Syst Evol Res 54:13–21. https://doi.org/10.1111/jzs.12114
Matos J, West SC (2014) Holliday junction resolution: regulation in space and time. DNA Repair (amst) 19:176–181. https://doi.org/10.1016/j.dnarep.2014.03.013
Moreira MO, Fonseca C, Rojas D (2021) Parthenogenesis is self-destructive for scaled reptiles. Biol Lett 17:20210006. https://doi.org/10.1098/rsbl.2021.0006
Neale MJ, Keeney S (2006) Clarifying the mechanics of DNA strand exchange in meiotic recombination. Nature 442:153–158. https://doi.org/10.1038/nature04885
Neiman M, Sharbel TF, Schwander T (2014) Genetic causes of transitions from sexual reproduction to asexuality in plants and animals. J Evol Biol 27:1346–1359. https://doi.org/10.1111/jeb.12357
Nougue O, Rode NO, Jabbour-Zahab R, Segard A, Chevin LM, Haag CR, Lenormand T (2015) Automixis in Artemia: solving a century-old controversy. J Evol Biol 28:2337–2348. https://doi.org/10.1111/jeb.12757
Parker DJ, Bast J, Jalvingh K, Dumas Z, Robinson-Rechavi M, Schwander T (2019) Repeated evolution of asexuality involves convergent gene expression changes. Mol Biol Evol 36:350–364. https://doi.org/10.1093/molbev/msy217
Pfeiffer HH (1965) Les alterations de la meiose chez les animaux parthenogenetiques. Kolloid-Zeitschrift and Zeitschrift Fur Polymere 202:174
Philippe L, Tosca L, Zhang WL, Piquemal M, Ciapa B (2014) Different routes lead to apoptosis in unfertilized sea urchin eggs. Apoptosis 19:436–450. https://doi.org/10.1007/s10495-013-0950-3
Reatty RA (1967) CHAPTER 9 - parthenogenesis in vertebrates. In: Metz CB, Monroy A (eds) Fertilization. Academic Press, pp 413–440
Rey O, Loiseau A, Facon B, Foucaud J, Orivel J, Cornuet JM, Robert S, Dobigny G, Delabie JH, Mariano CS, Estoup A (2011) Meiotic recombination dramatically decreased in thelytokous queens of the little fire ant and their sexually produced workers. Mol Biol Evol 28:2591–2601. https://doi.org/10.1093/molbev/msr082
Riparbelli MG, Gottardo M, Callaini G (2017) Parthenogenesis in insects: the centriole renaissance. Results Probl Cell Differ 63:435–479. https://doi.org/10.1007/978-3-319-60855-6_19
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Schon IMKV (2009) Lost sex: the evolutionary biology of parthenogenesis. Dordrecht, Netherlands. https://doi.org/10.1007/978-90-481-2770-2
Schön I, Gandolfi A, di Masso E, Rossi V, Griffiths HI, Martens K, Butlin RK (2000) Persistence of asexuality through mixed reproduction in Eucypris virens (Crustacea, Ostracoda). Heredity 84:161–169. https://doi.org/10.1046/j.1365-2540.2000.00647.x
Schurko AM, Logsdon JM, Eads BD (2009) Meiosis genes in Daphnia pulex and the role of parthenogenesis in genome evolution. BMC Evol Biol 9:78. https://doi.org/10.1186/1471-2148-9-78
Schwander T, Crespi BJ (2009a) Multiple direct transitions from sexual reproduction to apomictic parthenogenesis in Timema stick insects. Evolution 63:84–103. https://doi.org/10.1111/j.1558-5646.2008.00524.x
Schwander T, Crespi BJ (2009b) Twigs on the tree of life? Neutral and selective models for integrating macroevolutionary patterns with microevolutionary processes in the analysis of asexuality. Mol Ecol 18:28–42. https://doi.org/10.1111/j.1365-294X.2008.03992.x
Seitz R, Vilpoux K, Hopp U, Harzsch S, Maier G (2005) Ontogeny of the Marmorkrebs (marbled crayfish): a parthenogenetic crayfish with unknown origin and phylogenetic position. J Exp Zool A Comp Exp Biol 303:393–405. https://doi.org/10.1002/jez.a.143
Simon JC, Delmotte F, Rispe C, Crease T (2003) Phylogenetic relationships between parthenogens and their sexual relatives: the possible routes to parthenogenesis in animals. Biol J Linn Soc 79:151–163. https://doi.org/10.1046/j.1095-8312.2003.00175.x
Stelzer CP, Schmidt J, Wiedlroither A, Riss S (2010) Loss of sexual reproduction and dwarfing in a small metazoan. PLoS One 5:e12854. https://doi.org/10.1371/journal.pone.0012854
Suomalainen E, Saura A, Lokki J (1987) Cytology and evolution in parthenogenesis. CRC Press Inc, Boca Raton
Swain JE, Ding J, Wu JW, Smith GD (2008) Regulation of spindle and chromatin dynamics during early and late stages of oocyte maturation by aurora kinases. Mol Hum Reprod 14:291–299. https://doi.org/10.1093/molehr/gan015
Taieb F, Thibier C, Jessus C (1997) On cyclins, oocytes, and eggs. Mol Reprod Dev 48:397–411. https://doi.org/10.1002/(SICI)1098-2795(199711)48:33.0.CO;2-T
Trudgill DL, Blok VC (2001) Apomictic, polyphagous root-knot nematodes: exceptionally successful and damaging biotrophic root pathogens. Annu Rev Phytopathol 39:53–77. https://doi.org/10.1146/annurev.phyto.39.1.53
Vavre F, de Jong JH, Stouthamer R (2004) Cytogenetic mechanism and genetic consequences of thelytoky in the wasp Trichogramma cacoeciae. Heredity (edinb) 93:592–596. https://doi.org/10.1038/sj.hdy.6800565
Vijverberg K, Ozias-Akins P, Schranz ME (2019) Identifying and engineering genes for parthenogenesis in plants. Front Plant Sci 10:128. https://doi.org/10.3389/fpls.2019.00128
Welch DM, Meselson M (2000) Evidence for the evolution of bdelloid rotifers without sexual reproduction or genetic exchange. Science 288:1211–1215. https://doi.org/10.1126/science.288.5469.1211
Xu S, Huynh TV, Snyman M (2022) The transcriptomic signature of obligate parthenogenesis. Heredity 128:132–138. https://doi.org/10.1038/s41437-022-00498-1
Yang HJ, Zhou C, Dhar A, Pavletich NP (2020) Mechanism of strand exchange from RecA-DNA synaptic and D-loop structures. Nature 586:801–806. https://doi.org/10.1038/s41586-020-2820-9
Yodmuang S, Tirasophon W, Roshorm Y, Chinnirunvong W, Panyim S (2006) YHV-protease dsRNA inhibits YHV replication in Penaeus monodon and prevents mortality. Biochem Biophys Res Commun 341:351–356. https://doi.org/10.1016/j.bbrc.2005.12.186
Zadorozhny K, Sannino V, Belan O, Mlcouskova J, Spirek M, Costanzo V, Krejci L (2017) Fanconi-anemia-associated mutations destabilize RAD51 filaments and impair replication fork protection. Cell Rep 21:333–340. https://doi.org/10.1016/j.celrep.2017.09.062
Zickler D, Kleckner N (2015) Recombination, pairing, and synapsis of homologs during meiosis. Cold Spring Harb Perspect Biol 7:a016626. https://doi.org/10.1101/cshperspect.a016626
Funding
This work was supported by the National Key R & D Program of China Grant 2018YFD0900603, Key Scientific and Technological Grant of Zhejiang for Breeding New Agricultural Varieties 2021C02069-4, National Natural Science Foundation of China Grant 32172945, and Natural Science Foundation of Zhejiang Province Grant LY23C040003.
Author information
Authors and Affiliations
Contributions
Wei-Jun Yang, Jin-Shu Yang, and Lian-Ying Xu conceived and designed the research. Lian-Ying Xu, Wen-Tao Wu, Ning Bi, Zhi-Jun Yan, and Fan Yang performed the experiments. Lian-Ying Xu and Jin-Shu Yang analyzed the experiment data. Jin-Shu Yang and Lian-Ying Xu wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors approved the final version of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, LY., Wu, WT., Bi, N. et al. A cytological revisit on parthenogenetic Artemia and the deficiency of a meiosis-specific recombinase DMC1 in the possible transition from bisexuality to parthenogenesis. Chromosoma 132, 89–103 (2023). https://doi.org/10.1007/s00412-023-00790-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-023-00790-x