Abstract
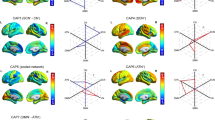
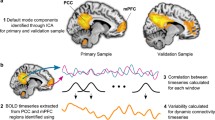
Accumulating evidence suggests that the brain is highly dynamic; thus, investigation of brain dynamics especially in brain connectivity would provide crucial information that stationary functional connectivity could miss. This study investigated temporal expressions of spatial modes within the default mode network (DMN), salience network (SN) and cognitive control network (CCN) using a reliable data-driven co-activation pattern (CAP) analysis in two independent data sets. We found enhanced CAP-to-CAP transitions of the SN in patients with MDD. Results suggested enhanced flexibility of this network in the patients. By contrast, we also found reduced spatial consistency and persistence of the DMN in the patients, indicating reduced variability and stability in individuals with MDD. In addition, the patients were characterized by prominent activation of mPFC. Moreover, further correlation analysis revealed that persistence and transitions of RCCN were associated with the severity of depression. Our findings suggest that functional connectivity in the patients may not be simply attenuated or potentiated, but just alternating faster or slower among more complex patterns. The aberrant temporal–spatial complexity of intrinsic fluctuations reflects functional diaschisis of resting-state networks as characteristic of patients with MDD.





Similar content being viewed by others
Availability of data and code
The data that support the findings of this study as well as code used during the current study are available from the corresponding author upon reasonable request.
References
Song Z, Zhang M, Huang P (2016) Aberrant emotion networks in early major depressive disorder patients: an eigenvector centrality mapping study. Transl Psychiatry 6:e819. https://doi.org/10.1038/tp.2016.81
Belmaker RH, Agam G (2008) Major depressive disorder. N Engl J Med 358:55–68
Fava M, Kendler KS (2000) Major depressive disorder. Neuron 28:335–341. https://doi.org/10.1016/S0896-6273(00)00112-4
Otte C, Gold SM, Penninx BW et al (2016) Major depressive disorder. Nat Rev Dis Prim 2:16065. https://doi.org/10.1038/nrdp.2016.65
Mulders PC, van Eijndhoven PF, Schene AH et al (2015) Resting-state functional connectivity in major depressive disorder: a review. Neurosci Biobehav Rev 56:330–344. https://doi.org/10.1016/j.neubiorev.2015.07.014
Ye M, Yang T, Qing P et al (2015) Changes of functional brain networks in major depressive disorder: a graph theoretical analysis of resting-state fMRI. PLoS One 10:e0133775. https://doi.org/10.1371/journal.pone.0133775
Dutta A, McKie S, Deakin JFW (2014) Resting state networks in major depressive disorder. Psychiatry Res 224:139–151. https://doi.org/10.1016/j.pscychresns.2014.10.003
Sheng J, Shen Y, Qin Y et al (2018) Spatiotemporal, metabolic, and therapeutic characterization of altered functional connectivity in major depressive disorder. Hum Brain Mapp 39:1957–1971. https://doi.org/10.1002/hbm.23976
Wang Y, Yang S, Sun W et al (2016) Altered functional interaction hub between affective network and cognitive control network in patients with major depressive disorder. Behav Brain Res 298:301–309. https://doi.org/10.1016/j.bbr.2015.10.040
Liu C-H, Guo J, Lu S-L et al (2018) Increased salience network activity in patients with insomnia complaints in major depressive disorder. Front Psychiatry 9:93. https://doi.org/10.3389/fpsyt.2018.00093
Li B, Liu L, Friston KJ et al (2013) A treatment-resistant default mode subnetwork in major depression. Biol Psychiatry 74:48–54. https://doi.org/10.1016/j.biopsych.2012.11.007
Ho TC, Connolly CG, Henje Blom E et al (2015) Emotion-dependent functional connectivity of the default mode network in adolescent depression. Biol Psychiatry 78:635–646. https://doi.org/10.1016/j.biopsych.2014.09.002
Bluhm R, Williamson P, Lanius R et al (2009) Resting state default-mode network connectivity in early depression using a seed region-of-interest analysis: decreased connectivity with caudate nucleus. Psychiatry Clin Neurosci 63:754–761. https://doi.org/10.1111/j.1440-1819.2009.02030.x
Yan CG, Chen X, Li L et al (2019) Reduced default mode network functional connectivity in patients with recurrent major depressive disorder. Proc Natl Acad Sci 116:201900390
Allen EA, Damaraju E, Plis SM et al (2014) Tracking whole-brain connectivity dynamics in the resting state. Cereb Cortex 24:663–676. https://doi.org/10.1093/cercor/bhs352
Chang C, Glover GH (2010) Time-frequency dynamics of resting-state brain connectivity measured with fMRI. Neuroimage 50:81–98. https://doi.org/10.1016/j.neuroimage.2009.12.011
Smith SM, Miller KL, Moeller S et al (2012) Temporally-independent functional modes of spontaneous brain activity. Proc Natl Acad Sci U S A 109:3131–3136. https://doi.org/10.1073/pnas.1121329109
Di X, Biswal BB (2020) Intersubject consistent dynamic connectivity during natural vision revealed by functional MRI. Neuroimage 216:116698. https://doi.org/10.1016/j.neuroimage.2020.116698
Kang J, Wang L, Yan C et al (2011) Characterizing dynamic functional connectivity in the resting brain using variable parameter regression and Kalman filtering approaches. Neuroimage 56:1222–1234. https://doi.org/10.1016/j.neuroimage.2011.03.033
Hutchison RM, Womelsdorf T, Gati JS et al (2013) Resting-state networks show dynamic functional connectivity in awake humans and anesthetized macaques. Hum Brain Mapp 34:2154–2177. https://doi.org/10.1002/hbm.22058
Fiorenzato E, Strafella AP, Kim J et al (2019) Dynamic functional connectivity changes associated with dementia in Parkinson’s disease. Brain 142:2860–2872. https://doi.org/10.1093/brain/awz192
Watanabe T, Rees G (2017) Brain network dynamics in high-functioning individuals with autism. Nat Commun 8:16048. https://doi.org/10.1038/ncomms16048
Wang J, Wang Y, Huang H et al (2020) Abnormal dynamic functional network connectivity in unmedicated bipolar and major depressive disorders based on the triple-network model. Psychol Med 50:465–474. https://doi.org/10.1017/S003329171900028X
Handwerker DA, Roopchansingh V, Gonzalez-Castillo J, Bandettini PA (2012) Periodic changes in fMRI connectivity. Neuroimage 63:1712–1719. https://doi.org/10.1016/j.neuroimage.2012.06.078
Jones DT, Vemuri P, Murphy MC et al (2012) Non-stationarity in the ‘resting brain’s’ modular architecture. PLoS One 7:e39731. https://doi.org/10.1371/journal.pone.0039731
Demirtaş M, Tornador C, Falcón C et al (2016) Dynamic functional connectivity reveals altered variability in functional connectivity among patients with major depressive disorder. Hum Brain Mapp 37:2918–2930. https://doi.org/10.1002/hbm.23215
Zhi D, Calhoun VD, Lv L et al (2018) Aberrant dynamic functional network connectivity and graph properties in major depressive disorder. Front Psychiatry 9:1–11. https://doi.org/10.3389/fpsyt.2018.00339
Liu X, Duyn JH (2013) Time-varying functional network information extracted from brief instances of spontaneous brain activity. Proc Natl Acad Sci U S A 110:4392–4397. https://doi.org/10.1073/pnas.1216856110
Marshall E, Nomi JS, Dirks B et al (2020) Coactivation pattern analysis reveals altered salience network dynamics in children with autism spectrum disorder. Netw Neurosci 4:1219–1234. https://doi.org/10.1162/netn_a_00163
Chen JE, Chang C, Greicius MD, Glover GH (2015) Introducing co-activation pattern metrics to quantify spontaneous brain network dynamics. Neuroimage 111:476–488. https://doi.org/10.1016/j.neuroimage.2015.01.057
Xue W, Kang J, Bowman FD et al (2014) Identifying functional co-activation patterns in neuroimaging studies via poisson graphical models. Biometrics 70:812–822. https://doi.org/10.1111/biom.12216
Messé A, Hütt M-T, Hilgetag CC (2018) Toward a theory of coactivation patterns in excitable neural networks. PLoS Comput Biol 14:e1006084. https://doi.org/10.1371/journal.pcbi.1006084
Kaiser RH, Whitfield-Gabrieli S, Dillon DG et al (2016) Dynamic resting-state functional connectivity in major depression. Neuropsychopharmacology 41:1822–1830. https://doi.org/10.1038/npp.2015.352
Kaiser RH, Kang MS, Lew Y et al (2019) Abnormal frontoinsular-default network dynamics in adolescent depression and rumination: a preliminary resting-state co-activation pattern analysis. Neuropsychopharmacology 44:1604–1612. https://doi.org/10.1038/s41386-019-0399-3
Smith SM, Fox PT, Miller KL et al (2009) Correspondence of the brain’s functional architecture during activation and rest. Proc Natl Acad Sci U S A 106:13040–13045. https://doi.org/10.1073/pnas.0905267106
Liu X, Chang C, Duyn JH (2013) Decomposition of spontaneous brain activity into distinct fMRI co-activation patterns. Front Syst Neurosci 7:101. https://doi.org/10.3389/fnsys.2013.00101
Xiang T, Gong S (2008) Spectral clustering with eigenvector selection. Pattern Recognit 41:1012–1029. https://doi.org/10.1016/j.patcog.2007.07.023
Lukasik S, Kowalski PA, Charytanowicz M, Kulczycki, Vancouver, 24–29 July 2016, pp. 2724–2728 PBT-2016 IC on EC (2016) Clustering using flower pollination algorithm and Calinski–Harabasz index
Pang Y, Chen H, Wang Y et al (2018) Transdiagnostic and diagnosis-specific dynamic functional connectivity anchored in the right anterior insula in major depressive disorder and bipolar depression. Prog Neuropsychopharmacol Biol Psychiatry 85:7–15. https://doi.org/10.1016/j.pnpbp.2018.03.020
Tian S, Chattun MR, Zhang S et al (2019) Dynamic community structure in major depressive disorder: a resting-state MEG study. Prog Neuropsychopharmacol Biol Psychiatry 92:39–47. https://doi.org/10.1016/j.pnpbp.2018.12.006
Kaiser RH, Andrews-Hanna JR, Wager TD, Pizzagalli DA (2015) Large-scale network dysfunction in major depressive disorder: a meta-analysis of resting-state functional connectivity. JAMA Psychiat 72:603–611. https://doi.org/10.1001/jamapsychiatry.2015.0071
Bolton TAW, Wotruba D, Buechler R et al (2020) Triple network model dynamically revisited: lower salience network state switching in pre-psychosis. Front Physiol 11:66. https://doi.org/10.3389/fphys.2020.00066
Kupis L, Romero C, Dirks B et al (2020) Evoked and intrinsic brain network dynamics in children with autism spectrum disorder. NeuroImage Clin 28:102396. https://doi.org/10.1016/j.nicl.2020.102396
Kaiser RH, Whitfield-Gabrieli S, Dillon DG et al (2016) Dynamic resting-state functional connectivity in major depression. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 41:1822–1830. https://doi.org/10.1038/npp.2015.352
Broyd SJ, Demanuele C, Debener S et al (2009) Default-mode brain dysfunction in mental disorders: a systematic review. Neurosci Biobehav Rev 33:279–296. https://doi.org/10.1016/j.neubiorev.2008.09.002
Zamoscik V, Huffziger S, Ebner-Priemer U et al (2014) Increased involvement of the Parahippocampal gyri in a sad mood predicts future depressive symptoms. Soc Cogn Affect Neurosci 9:2034–2040. https://doi.org/10.1093/scan/nsu006
Nolen-hoeksema S, Wisco BE, Lyubomirsky S (2008) Rethinking rumination. Perspect Psychol Sci 3:400–424
Goodman ZT, Bainter SA, Kornfeld S et al (2021) Whole-brain functional dynamics track depressive symptom severity. Cereb Cortex 31:4867–4876. https://doi.org/10.1093/cercor/bhab047
Ho TC, Wu J, Shin D et al (2013) Altered cerebral perfusion in executive, affective, and motor networks during adolescent depression. J Am Acad Child Adolesc Psychiatry 52:1076-1091.e2
Chen H, Liu K, Zhang B et al (2019) More optimal but less regulated dorsal and ventral visual networks in patients with major depressive disorder. J Psychiatr Res 110:172–178
Ding YD, Yang R, Yan CG et al (2021) Disrupted hemispheric connectivity specialization in patients with major depressive disorder: evidence from the REST-meta-MDD Project. J Affect Disord 284:217–228
Cohen AD, Chang C, Wang Y (2021) Using multiband multi-echo imaging to improve the robustness and repeatability of co-activation pattern analysis for dynamic functional connectivity. Neuroimage 243:118555. https://doi.org/10.1016/j.neuroimage.2021.118555
Funding
This work was supported by the National Natural Science Foundation of China (62088102, U21A20485, 61976248, 91648208, 61976175, 81371478, 61503411).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection was performed by the LL, LL, JL and KZ. Clinical data were assessed by JL. The preprocessing analyses were performed by KZ and JQ. The first draft of the manuscript was written by KZ and BL. All authors commented on previous versions of the manuscript. KZ, BC and DH reviewed and revised the manuscript. All authors approved the final manuscript as submitted.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Ethics approval and consent to participants
This study was approved by the Ethics Committee in Zhumadian Psychiatric Hospital and Xi-jing Hospital. All subjects gave informed consent before participating in this study.
Research involving human participants and/or animals
Human and human samples were used in the current study.
Consent for publication
Last version of the manuscript was approved by the authors before the submission.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zheng, K., Li, B., Lu, H. et al. Aberrant temporal–spatial complexity of intrinsic fluctuations in major depression. Eur Arch Psychiatry Clin Neurosci 273, 169–181 (2023). https://doi.org/10.1007/s00406-022-01403-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-022-01403-x