Abstract
Purpose
It is unknown if future fertility is compromised by the administration of chemotherapy during pregnancy. The aim of this study was to identify if chemotherapy affects the maternal ovaries during pregnancy and whether these effects depend on type of chemotherapy and duration of exposure.
Methods
Pregnant 8-week-old female BL6 mice were exposed to 6 different single chemotherapeutic agents (carboplatin, cisplatin, paclitaxel, epirubicin, doxorubicin, or cyclophosphamide) or saline at gestational day (GD) 13.5. The mice were sacrificed at GD 15.5 or GD 18.5. Ovaries were assessed by histopathology and immunohistochemistry. Follicle count was determined per follicle stage and per treatment modality.
Results
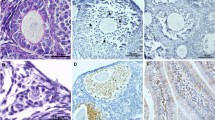
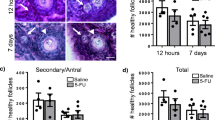
Maternal ovarian damage was demonstrated by the presence of apoptosis and necrosis in preantral follicles. The extent of this damage depends upon type of chemotherapy and duration of exposure (2 or 5 days). After short exposure, 81% of ovaries showed histopathologic signs of damage compared to 36% after long exposure, which might suggest a transient effect. Loss of primordial follicles (PMFs) was observed after both short and long exposure, with a reduction of more than 70%. Evidence of DNA damage, as demonstrated by phospho-H2AX expression, was present in 23% (range 0–89%) of PMFs exposed to chemotherapy, but only in the short exposure group. Overall, the least damage was seen after administration of paclitaxel.
Conclusion
Despite physiological ovarian function suppression during gestation, chemotherapy-induced damage of the ovaries occurs in pregnant mouse models, potentially affecting future fertility.








Similar content being viewed by others
References
de Haan J, Verheecke M, Van Calsteren K, Van Calster B, Shmakov RG, Mhallem Gziri M, Halaska MJ, Fruscio R, Lok CAR, Boere IA, Zola P, Ottevanger PB, de Groot CJM, Peccatori FA, Dahl Steffensen K, Cardonick EH, Polushkina E, Rob L, Ceppi L, Sukhikh GT, Han SN, Amant F, International Network on Cancer and Infertility Pregnancy (INCIP) (2018) Oncological management and obstetric and neonatal outcomes for women diagnosed with cancer during pregnancy: a 20-year international cohort study of 1170 patients. Lancet Oncol 19(3):337–346. https://doi.org/10.1016/S1470-2045(18)30059-7
Wolters V, Heimovaara J, Maggen C, Cardonick E, Boere I, Lenaerts L, Amant F (2021) Management of pregnancy in women with cancer. Int J Gynecol Cancer 31(3):314–322. https://doi.org/10.1136/ijgc-2020-001776
Hao X, Anastácio A, Liu K, Rodriguez-Wallberg KA (2019) Ovarian follicle depletion induced by chemotherapy and the investigational stages of potential fertility-protective treatments-a review. Int J Mol Sci 20(19):4720. https://doi.org/10.3390/ijms20194720
Kalich-Philosoph L, Roness H, Carmely A, Fishel-Bartal M, Ligumsky H, Paglin S, Wolf I, Kanety H, Sredni B, Meirow D (2013) Cyclophosphamide triggers follicle activation and “burnout”; AS101 prevents follicle loss and preserves fertility. Sci Transl Med 5(185):185ra62. https://doi.org/10.1126/scitranslmed.3005402
Chen H, Xiao L, Li J, Cui L, Huang W (2019) Adjuvant gonadotropin-releasing hormone analogues for the prevention of chemotherapy-induced premature ovarian failure in premenopausal women. Cochrane Database Syst Rev 3(3):CD008018. https://doi.org/10.1002/14651858.CD008018.pub3
Ma N, Chen G, Chen J, Cui M, Yin Y, Liao Q, Tang M, Feng X, Li X, Zhang S, Ma D, Chen G, Li K, Ai J (2020) Transient impact of paclitaxel on mouse fertility and protective effect of gonadotropin-releasing hormone agonist. Oncol Rep 44(5):1917–1928. https://doi.org/10.3892/or.2020.7740
European Society of Human Reproduction and Embryology (2020) ESHRE guideline female fertility preservation. https://www.eshre.eu/Guidelines-and-Legal/Guidelines/Female-fertility-preservation. Accessed 22 Mar 2022
Oktay K, Harvey BE, Partridge AH, Quinn GP, Reinecke J, Taylor HS, Wallace WH, Wang ET, Loren AW (2018) Fertility preservation in patients with cancer: ASCO clinical practice guideline update. J Clin Oncol 36(19):1994–2001. https://doi.org/10.1200/JCO.2018.78.1914
Practice Committee of the American Society for Reproductive Medicine (2019) Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril 112(6):1022–1033. https://doi.org/10.1016/j.fertnstert.2019.09.013
Van Calsteren K, Verbesselt R, Van Bree R, Heyns L, de Bruijn E, de Hoon J, Amant F (2011) Substantial variation in transplacental transfer of chemotherapeutic agents in a mouse model. Reprod Sci 18(1):57–63. https://doi.org/10.1177/1933719110379650
Ryniec JA, McGee EA (2020) Ovarian function during pregnancy and lactation. In: Kovacs CS, Deal CL (eds) Maternal-fetal and neonatal endocrinology, 1st edn. Academic Press, Cambridge, pp 117–127. https://doi.org/10.1016/B978-0-12-814823-5.00009-X
Spears N, Lopes F, Stefansdottir A, Rossi V, De Felici M, Anderson RA, Klinger FG (2019) Ovarian damage from chemotherapy and current approaches to its protection. Hum Reprod Update 25(6):673–693. https://doi.org/10.1093/humupd/dmz027
Morgan S, Anderson RA, Gourley C, Wallace WH, Spears N (2012) How do chemotherapeutic agents damage the ovary? Hum Reprod Update 18(5):525–535. https://doi.org/10.1093/humupd/dms022
Miller JJ 3rd, Williams GF, Leissring JC (1971) Multiple late complications of therapy with cyclophosphamide, including ovarian destruction. Am J Med 50:530–535. https://doi.org/10.1016/0002-9343(71)90341-x
Koyama H, Wada T, Nishizawa Y, Iwanaga T, Aoki Y (1977) Cyclophosphamide-induced ovarian failure and its therapeutic significance in patients with breast cancer. Cancer 39:1403–1409. https://doi.org/10.1002/1097-0142(197704)39:4%3c1403::aid-cncr2820390408%3e3.0.co;2-8
Stefansdottir A, Johnston ZC, Powles-Glover N, Anderson RA, Adams IR, Spears N (2016) Etoposide damages female germ cells in the developing ovary. BMC Cancer 16(1):482. https://doi.org/10.1186/s12885-016-2505-9
Acknowledgements
We would like to thank the people from the Preclinical Intervention Unit of the Mouse Clinic for Cancer and Ageing (MCCA) and the animal caretakers at the Laboratory Animal Centre of the NKI for performing the intervention studies.
Funding
Internal funding was used to purchase the mice.
Author information
Authors and Affiliations
Contributions
Study concept and design: CL, FA, and MV; material preparations and data collection: TS, JYS, and VW; data analysis and interpretation: TS, JYS, CL; writing—original draft: TS; critical revision: all authors; approval of final manuscript: all authors.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest. The authors have no relevant financial or non-financial interests to disclose.
Ethics approval and consent to participate
The animal experiments were approved by The Central Animal Testing Committee of The Netherlands (AVD3010020198564 EGP 21.1.8924).
Consent to publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Schuurman, T., Song, JY., Wolters, V. et al. Effects of chemotherapy on ovaries of pregnant mice. Arch Gynecol Obstet 307, 1163–1176 (2023). https://doi.org/10.1007/s00404-022-06793-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06793-w