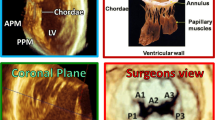
Abstract
Mitral valve prolapse (MVP) is the most common valve disease in the western world and recently emerged as a possible substrate for sudden cardiac death (SCD). It is estimated an annual risk of sudden cardiac death of 0.2 to 1.9% mostly caused by complex ventricular arrhythmias (VA). Several mechanisms have been recognized as potentially responsible for arrhythmia onset in MVP, resulting from the combination of morpho-functional abnormality of the mitral valve, structural substrates (regional myocardial hypertrophy, fibrosis, Purkinje fibers activity, inflammation), and mechanical stretch. Echocardiography plays a central role in MVP diagnosis and assessment of severity of regurgitation. Several abnormalities detectable by echocardiography can be prognostic for the occurrence of VA, from morphological alteration including leaflet redundancy and thickness, mitral annular dilatation, and mitral annulus disjunction (MAD), to motion abnormalities detectable with “Pickelhaube” sign. Additionally, speckle-tracking echocardiography may identify MVP patients at higher risk for VA by detection of increased mechanical dispersion. On the other hand, cardiac magnetic resonance (CMR) has the capability to provide a comprehensive risk stratification combining the identification of morphological and motion alteration with the detection of myocardial replacement and interstitial fibrosis, making CMR an ideal method for arrhythmia risk stratification in patients with MVP. Finally, recent studies have suggested a potential role in risk stratification of new techniques such as hybrid PET-MR and late contrast enhancement CT. The purpose of this review is to provide an overview of the mitral valve prolapse syndrome with a focus on the role of imaging in arrhythmic risk stratification.
Clinical relevance statement
Mitral valve prolapse is the most frequent valve condition potentially associated with arrhythmias. Imaging has a central role in the identification of anatomical, functional, mechanical, and structural alterations potentially associated with a higher risk of developing complex ventricular arrhythmia and sudden cardiac death.
Key Points
• Mitral valve prolapse is a common valve disease potentially associated with complex ventricular arrhythmia and sudden cardiac death.
• The mechanism of arrhythmogenesis in mitral valve prolapse is complex and multifactorial, due to the interplay among multiple conditions including valve morphological alteration, mechanical stretch, myocardial structure remodeling with fibrosis, and inflammation.
• Cardiac imaging, especially echocardiography and cardiac magnetic resonance, is crucial in the identification of several features associated with the potential risk of serious cardiac events. In particular, cardiac magnetic resonance has the advantage of being able to detect myocardial fibrosis which is currently the strongest prognosticator.
Graphical abstract







Similar content being viewed by others
Abbreviations
- AMVP:
-
Arrhythmic mitral valve prolapse
- BD:
-
Barlow’s disease
- CMR:
-
Cardiovascular MR
- cVA:
-
Complex ventricular arrhythmia
- FED:
-
Fibroelastic deficiency
- MAD:
-
Mitral annular disjunction
- MVP:
-
Mitral valve prolapse
- NSVT:
-
Non-sustained ventricular tachycardia
- PVC:
-
Premature ventricular contraction
- SCD:
-
Sudden cardiac death
- VF:
-
Ventricular fibrillation
- VT:
-
Ventricular tachycardia
References
Delling FN, Vasan RS (2014) Epidemiology and pathophysiology of mitral valve prolapse. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.113.006702
Adabifirouzjaei F, Hsiao A, DeMaria AN (2022) Mitral valve prolapse—the role of cardiac imaging modalities. Structural Heart. https://doi.org/10.1016/j.shj.2022.100024
Sabbag A, Essayagh B, Barrera JDR et al (2022) EHRA expert consensus statement on arrhythmic mitral valve prolapse and mitral annular disjunction complex in collaboration with the ESC Council on valvular heart disease and the European Association of Cardiovascular Imaging endorsed cby the Heart Rhythm Society, by the Asia Pacific Heart Rhythm Society, and by the Latin American Heart Rhythm Society. EP Europace. https://doi.org/10.1093/europace/euac125
Parwani P, Avierinos J-F, Levine RA, Delling FN (2017) Mitral valve prolapse: multimodality imaging and genetic insights. Prog Cardiovasc Dis. https://doi.org/10.1016/j.pcad.2017.10.007
Palmisano A, Nicoletti V, Colantoni C et al (2021) Dynamic changes of mitral valve annulus geometry at preprocedural CT: relationship with functional classes of regurgitation. Eur Radiol Exp. https://doi.org/10.1186/s41747-021-00231-3
Nalliah CJ, Mahajan R, Elliott AD et al (2019) Mitral valve prolapse and sudden cardiac death: a systematic review and meta-analysis. Heart. https://doi.org/10.1136/heartjnl-2017-312932
Tseng ZH, Olgin JE, Vittinghoff E et al (2018) Prospective countywide surveillance and autopsy characterization of sudden cardiac death. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.117.033427
Basso C, Perazzolo Marra M, Rizzo S et al (2015) Arrhythmic mitral valve prolapse and sudden cardiac death. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.115.016291
Basso C, Iliceto S, Thiene G, Perazzolo Marra M (2019) Mitral valve prolapse, ventricular arrhythmias, and sudden death. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.118.034075
Düren DR, Becker AE, Dunning AJ (1988) Long-term follow-up of idiopathic mitral valve prolapse in 300 patients: a prospective study. J Am Coll Cardiol. https://doi.org/10.1016/0735-1097(88)90164-7
Nishimura RA, McGoon MD, Shub C et al (1985) Echocardiographically documented mitral-valve prolapse. N Engl J Med. https://doi.org/10.1056/NEJM198511213132101
Aabel EW, Chivulescu M, Lie ØH et al (2023) Ventricular arrhythmias in arrhythmic mitral valve syndrome—a prospective continuous long-term cardiac monitoring study. EP Europace. https://doi.org/10.1093/europace/euac182
Virani SS, Alonso A, Aparicio HJ et al (2021) Heart disease and stroke statistics—2021 Update. Circulation. https://doi.org/10.1161/CIR.0000000000000950
Grigioni F, Enriquez-Sarano M, Ling LH et al (1999) Sudden death in mitral regurgitation due to flail leaflet. J Am Coll Cardiol. https://doi.org/10.1016/S0735-1097(99)00474-X
Savage DD, Devereux RB, Garrison RJ et al (1983) Mitral valve prolapse in the general population. 2. Clinical features: the Framingham Study. Am Heart J 106(3):577–581. https://doi.org/10.1016/0002-8703(83)90705-6
Han H, Ha FJ, Teh AW et al (2018) Mitral valve prolapse and sudden cardiac death: a systematic review. J Am Heart Assoc. https://doi.org/10.1161/JAHA.118.010584
Freed LA, Levy D, Levine RA et al (1999) Prevalence and Clinical Outcome of Mitral-Valve Prolapse. N Engl J Med. https://doi.org/10.1056/NEJM199907013410101
Bhutto ZR, Barron JT, Liebson PR et al (1992) Electrocardiographic abnormalities in mitral valve prolapse. Am J Cardiol. https://doi.org/10.1016/0002-9149(92)91287-E
Bekheit SG, Ali AA, Deglin SM, Jain AC (1982) Analysis of QT interval in patients with idiopathic mitral valve prolapse. Chest. https://doi.org/10.1378/chest.81.5.620
Kaya Ü, Eren H (2020) Fragmented QRS may be associated with complex ventricular arrhythmias in mitral valve prolapse. Minerva Cardioangiol 68(6):577–585. https://doi.org/10.23736/S0026-4725.20.05123-3
Basso C, Calabrese F, Corrado D, Thiene G (2001) Postmortem diagnosis in sudden cardiac death victims: macroscopic, microscopic and molecular findings. Cardiovasc Res. https://doi.org/10.1016/S0008-6363(01)00261-9
Miller MA, Dukkipati SR, Turagam M et al (2018) Arrhythmic mitral valve prolapse. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2018.09.048
Han H, Parsons SA, Teh AW et al (2020) Characteristic histopathological findings and cardiac arrest rhythm in isolated mitral valve prolapse and sudden cardiac death. J Am Heart Assoc. https://doi.org/10.1161/JAHA.119.015587
Han H-C, Parsons SA, Curl CL et al (2021) Systematic quantification of histologic ventricular fibrosis in isolated mitral valve prolapse and sudden cardiac death. Heart Rhythm. https://doi.org/10.1016/j.hrthm.2020.12.021
Morningstar JE, Gensemer C, Moore R et al (2021) Mitral Valve Prolapse Induces Regionalized Myocardial Fibrosis. J Am Heart Assoc. https://doi.org/10.1161/JAHA.121.022332
Hinderer S, Schenke-Layland K (2019) Cardiac fibrosis – a short review of causes and therapeutic strategies. Adv Drug Deliv Rev. https://doi.org/10.1016/j.addr.2019.05.011
Rizzo S, Perazzolo Marra M, De Gaspari M, Basso C (2021) The missing pieces in the puzzle of arrhythmic mitral valve prolapse: papillary muscles, mitral annulus dysjunction, and myocardial scarring. Heart Rhythm. https://doi.org/10.1016/j.hrthm.2021.01.004
Garbi M, Lancellotti P, Sheppard MN (2018) Mitral valve and left ventricular features in malignant mitral valve prolapse. Open Heart. https://doi.org/10.1136/openhrt-2018-000925
SantoroBIASE FLDI, HRANITZKY P et al (2014) Ventricular fibrillation triggered by PVCs from papillary muscles: clinical features and ablation. J Cardiovasc Electrophysiol. https://doi.org/10.1111/jce.12478
Alhede C, Higuchi S, Hadjis A et al (2022) Premature ventricular contractions are presaged by a mechanically abnormal sinus beat. JACC Clin Electrophysiol. https://doi.org/10.1016/j.jacep.2022.05.005
Sriram CS, Syed FF, Ferguson ME et al (2013) Malignant bileaflet mitral valve prolapse syndrome in patients with otherwise idiopathic out-of-hospital cardiac arrest. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2013.02.060
Villatore A, Sala S, Stella S et al (2021) Autoimmune myocarditis and arrhythmogenic mitral valve prolapse: an unexpected overlap syndrome. J Cardiovasc Dev Dis. https://doi.org/10.3390/jcdd8110151
Jiang W, Xiong Y, Li X, Yang Y (2021) Cardiac fibrosis: cellular effectors, molecular pathways, and exosomal roles. Front Cardiovasc Med. https://doi.org/10.3389/fcvm.2021.715258
Salvador AM, Nevers T, Velázquez F et al (2016) Intercellular adhesion molecule 1 regulates left ventricular leukocyte infiltration, cardiac remodeling, and function in pressure overload–induced heart failure. J Am Heart Assoc. https://doi.org/10.1161/JAHA.115.003126
Śniez̊ek-Maciejewska M, Dubiel JP, Piwowarska W et al (1992) Ventricular arrhythmias and the autonomic tone in patients with mitral valve prolapse. Clin Cardiol. https://doi.org/10.1002/clc.4960151029
Myles RC, Wang L, Kang C et al (2012) Local β-adrenergic stimulation overcomes source-sink mismatch to generate focal arrhythmia. Circ Res. https://doi.org/10.1161/CIRCRESAHA.111.262345
Mason JW, Koch FH, Billingham ME, Winkle RA (1978) Cardiac biopsy evidence for a cardiomyopathy associated with symptomatic mitral valve prolapse. Am J Cardiol. https://doi.org/10.1016/0002-9149(78)90623-9
La VL, Ometto R, Centofante P et al (1998) Arrhythmic profile, ventricular function, and histomorphometric findings in patients with idiopathic ventricular tachycardia and mitral valve prolapse: clinical and prognostic evaluation. Clin Cardiol. https://doi.org/10.1002/clc.4960211007
Bains S, Tester DJ, Asirvatham SJ et al (2019) A novel truncating variant in FLNC-encoded filamin C may serve as a proarrhythmic genetic substrate for arrhythmogenic bileaflet mitral valve prolapse syndrome. Mayo Clin Proc. https://doi.org/10.1016/j.mayocp.2018.11.028
Guglielmo M, Arangalage D, Bonino MA et al (2023) Additional value of cardiac magnetic resonance feature tracking parameters for the evaluation of the arrhythmic risk in patients with mitral valve prolapse. J Cardiovasc Magn Reson. https://doi.org/10.1186/s12968-023-00944-x
Nagata Y, Bertrand PB, Baliyan V et al (2023) Abnormal mechanics relate to myocardial fibrosis and ventricular arrhythmias in patients with mitral valve prolapse. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.122.014963
Chivulescu M, Aabel EW, Gjertsen E et al (2022) Electrical markers and arrhythmic risk associated with myocardial fibrosis in mitral valve prolapse. Europace. https://doi.org/10.1093/europace/euac017
Constant Dit Beaufils AL, Huttin O, Jobbe-Duval A et al (2021) Replacement myocardial fibrosis in patients with mitral valve prolapse: relation to mitral regurgitation, Ventricular Remodeling, and Arrhythmia. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.120.050214
Pavon AG, Arangalage D, Pascale P et al (2021) Myocardial extracellular volume by T1 mapping: a new marker of arrhythmia in mitral valve prolapse. J Cardiovasc Magn Reson. https://doi.org/10.1186/s12968-021-00797-2
Gatti M, Palmisano A, Esposito A et al (2021) Feature tracking myocardial strain analysis in patients with bileaflet mitral valve prolapse: relationship with LGE and arrhythmias. Eur Radiol. https://doi.org/10.1007/s00330-021-07876-z
Pradella S, Grazzini G, Brandani M et al (2019) Cardiac magnetic resonance in patients with mitral valve prolapse: focus on late gadolinium enhancement and T1 mapping. Eur Radiol. https://doi.org/10.1007/s00330-018-5634-5
Kitkungvan D, Nabi F, Kim RJ et al (2018) Myocardial fibrosis in patients with primary mitral regurgitation with and without prolapse. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2018.06.048
Bennett S, Tafuro J, Duckett S et al (2022) Definition, prevalence, and clinical significance of mitral annular disjunction in different patient cohorts: a systematic review. Echocardiography. https://doi.org/10.1111/echo.15299
Lancellotti P, Pibarot P, Chambers J et al (2022) Multi-modality imaging assessment of native valvular regurgitation: an EACVI and ESC council of valvular heart disease position paper. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jeab253
Zoghbi WA, Adams D, Bonow RO et al (2017) Recommendations for noninvasive evaluation of native valvular regurgitation. J Am Soc Echocardiogr. https://doi.org/10.1016/j.echo.2017.01.007
Nordhues BD, Siontis KC, Scott CG et al (2016) Bileaflet mitral valve prolapse and risk of ventricular dysrhythmias and death. J Cardiovasc Electrophysiol. https://doi.org/10.1111/jce.12914
Avierinos J-F, Gersh BJ, Melton LJ et al (2002) Natural history of asymptomatic mitral valve prolapse in the community. Circulation. https://doi.org/10.1161/01.CIR.0000028933.34260.09
Hutchins GM, Moore GW, Skoog DK (1986) The association of floppy mitral valve with disjunction of the mitral annulus fibrosus. N Engl J Med. https://doi.org/10.1056/NEJM198602273140902
Eriksson MJ, Bitkover CY, Omran AS et al (2005) Mitral annular disjunction in advanced myxomatous mitral valve disease: echocardiographic detection and surgical correction. J Am Soc Echocardiogr. https://doi.org/10.1016/j.echo.2005.06.013
Carmo P, Andrade MJ, Aguiar C et al (2010) Mitral annular disjunction in myxomatous mitral valve disease: a relevant abnormality recognizable by transthoracic echocardiography. Cardiovasc Ultrasound. https://doi.org/10.1186/1476-7120-8-53
Perazzolo Marra M, Basso C, De Lazzari M et al (2016) Morphofunctional abnormalities of mitral annulus and arrhythmic mitral valve prolapse. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.116.005030
Muthukumar L, Rahman F, Jan MF et al (2017) The Pickelhaube sign. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2016.09.016
Slavich M, Palmisano A, Pannone L et al (2019) Hidden danger behind the prolapse. Circ Cardiovasc Imaging. https://doi.org/10.1161/CIRCIMAGING.119.009639
Huttin O, Pierre S, Venner C et al (2016) Interactions between mitral valve and left ventricle analysed by 2D speckle tracking in patients with mitral valve prolapse: one more piece to the puzzle. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jew075
Essayagh B, Sabbag A, Antoine C et al (2021) The mitral annular disjunction of mitral valve prolapse. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2021.04.029
Tong J, Yew M, Huang W, Yong QW (2022) The dance of death: cardiac arrest, mitral and tricuspid valve prolapses, and biannular disjunctions. CASE (Phila). https://doi.org/10.1016/j.case.2021.11.006
Tayal B, Delling FN, Malahfji M, Shah DJ (2021) Cardiac imaging for risk assessment of malignant ventricular arrhythmias in patients with mitral valve prolapse. Front Cardiovasc Med. https://doi.org/10.3389/fcvm.2021.574446
Figliozzi S, Georgiopoulos G, Lopes PM et al (2023) Myocardial fibrosis at cardiac MRI helps predict adverse clinical outcome in patients with mitral valve prolapse. Radiology. https://doi.org/10.1148/radiol.220454
Zugwitz D, Fung K, Aung N et al (2022) Mitral annular disjunction assessed using CMR imaging. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2022.07.015
Dejgaard LA, Skjølsvik ET, Lie ØH et al (2018) The mitral annulus disjunction arrhythmic syndrome. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2018.07.070
Faletra FF, Leo LA, Paiocchi VL et al (2022) Morphology of mitral annular disjunction in mitral valve prolapse. J Am Soc Echocardiogr. https://doi.org/10.1016/j.echo.2021.09.002
Mantegazza V, Volpato V, Gripari P et al (2021) Multimodality imaging assessment of mitral annular disjunction in mitral valve prolapse. Heart. https://doi.org/10.1136/heartjnl-2020-317330
Angelini A, Ho SY, Anderson RH et al (1988) A histological study of the atrioventricular junction in hearts with normal and prolapsed leaflets of the mitral valve. Heart. https://doi.org/10.1136/hrt.59.6.712
Wu S, Siegel RJ (2022) Mitral annular disjunction: a case series and review of the literature. Front Cardiovasc Med. https://doi.org/10.3389/fcvm.2022.976066
Groeneveld SA, Kirkels FP, Cramer MJ et al (2022) Prevalence of mitral annulus disjunction and mitral valve prolapse in patients with idiopathic ventricular fibrillation. J Am Heart Assoc. https://doi.org/10.1161/JAHA.121.025364
Essayagh B, Iacuzio L, Civaia F et al (2019) Usefulness of 3-Tesla cardiac magnetic resonance to detect mitral annular disjunction in patients with mitral valve prolapse. Am J Cardiol. https://doi.org/10.1016/j.amjcard.2019.08.047
Basso C, Perazzolo Marra M (2018) Mitral annulus disjunction. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2018.07.069
Putnam AJ, Kebed K, Mor-Avi V et al (2020) Prevalence of mitral annular disjunction in patients with mitral valve prolapse and severe regurgitation. Int J Cardiovasc Imaging 36(7):1363–1370. https://doi.org/10.1007/s10554-020-01818-4
Kulkarni AA, Chudgar PD, Burkule NJ, Kamat NV (2022) Mitral annulus disjunction and arrhythmic mitral valve prolapse: emerging role of cardiac magnetic resonance imaging in the workup. Indian J Radiol Imaging 32(4):576–581. https://doi.org/10.1055/s-0042-1754357
Haugaa K (2021) Improving the imaging diagnosis of mitral annular disjunction. Heart. https://doi.org/10.1136/heartjnl-2020-317667
Romero Daza A, Chokshi A, Pardo P et al (2021) Mitral valve prolapse morphofunctional features by cardiovascular magnetic resonance: more than just a valvular disease. J Cardiovasc Magn Reson. https://doi.org/10.1186/s12968-021-00800-w
Aquaro GD, De Gori C, Grilli G et al (2023) Dark papillary muscles sign: a novel prognostic marker for cardiac magnetic resonance. Eur Radiol. https://doi.org/10.1007/s00330-023-09400-x
Dieterlen M-T, Klaeske K, Spampinato R et al (2023) Histopathological insights into mitral valve prolapse-induced fibrosis. Front Cardiovasc Med. https://doi.org/10.3389/fcvm.2023.1057986
Pradella S, Grazzini G, Miele V (2020) Mitral valve prolapse imaging: the role of tissue characterization. Quant Imaging Med Surg. https://doi.org/10.21037/qims-2020-25
Niu Z, Chan V, Mesana T, Ruel M (2016) The evolution of mitral valve prolapse: insights from the Framingham Heart Study. J Thorac Dis. https://doi.org/10.21037/jtd.2016.07.58
Thamman R, Gupta N, Doshi R et al (2020) Mitral annular disjunction: Wolf in sheep’s clothing? Echocardiography. https://doi.org/10.1111/echo.14898
Bui AH, Roujol S, Foppa M et al (2017) Diffuse myocardial fibrosis in patients with mitral valve prolapse and ventricular arrhythmia. Heart. https://doi.org/10.1136/heartjnl-2016-309303
Fulton BL, Liang JJ, Enriquez A et al (2018) Imaging characteristics of papillary muscle site of origin of ventricular arrhythmias in patients with mitral valve prolapse. J Cardiovasc Electrophysiol. https://doi.org/10.1111/jce.13374
Guglielmo M, Fusini L, Muscogiuri G et al (2021) T1 mapping and cardiac magnetic resonance feature tracking in mitral valve prolapse. Eur Radiol. https://doi.org/10.1007/s00330-020-07140-w
Hourdain J, Clavel MA, Deharo J-C et al (2018) Common phenotype in patients with mitral valve prolapse who experienced sudden cardiac death. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.118.033488
Kim AJ, Xu N, Umeyama K et al (2020) Deficiency of circulating monocytes ameliorates the progression of myxomatous valve degeneration in marfan syndrome. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.119.042391
Mewton N, Liu CY, Croisille P et al (2011) Assessment of myocardial fibrosis with cardiovascular magnetic resonance. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2010.11.013
Yue L, Xie J, Nattel S (2011) Molecular determinants of cardiac fibroblast electrical function and therapeutic implications for atrial fibrillation. Cardiovasc Res. https://doi.org/10.1093/cvr/cvq329
Sridhar S, Vandersickel N, Panfilov AV (2017) Effect of myocyte-fibroblast coupling on the onset of pathological dynamics in a model of ventricular tissue. Sci Rep. https://doi.org/10.1038/srep40985
Wynn TA, Ramalingam TR (2012) Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. https://doi.org/10.1038/nm.2807
Travers JG, Kamal FA, Robbins J et al (2016) Cardiac Fibrosis. Circ Res. https://doi.org/10.1161/CIRCRESAHA.115.306565
Piek A, de Boer RA, Silljé HHW (2016) The fibrosis-cell death axis in heart failure. Heart Fail Rev. https://doi.org/10.1007/s10741-016-9536-9
Nensa F, Bamberg F, Rischpler C et al (2018) Hybrid cardiac imaging using PET/MRI: a joint position statement by the European Society of Cardiovascular Radiology (ESCR) and the European Association of Nuclear Medicine (EANM). Eur Radiol. https://doi.org/10.1007/s00330-017-5008-4
Nazir MS, Ismail TF, Reyes E et al (2018) Hybrid positron emission tomography–magnetic resonance of the heart: current state of the art and future applications. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jey090
Miller MA, Adams DH, Pandis D et al (2020) Hybrid positron emission tomography/magnetic resonance imaging in arrhythmic mitral valve prolapse. JAMA Cardiol. https://doi.org/10.1001/jamacardio.2020.1555
Miller MA, Devesa A, Robson PM et al (2023) Arrhythmic mitral valve prolapse with only mild or moderate mitral regurgitation. JACC Clin Electrophysiol. https://doi.org/10.1016/j.jacep.2023.04.011
Dweck MR, Abgral R, Trivieri MG et al (2018) Hybrid magnetic resonance imaging and positron emission tomography with fluorodeoxyglucose to diagnose active cardiac sarcoidosis. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2017.02.021
Feuchtner GM, Alkadhi H, Karlo C et al (2010) Cardiac CT angiography for the diagnosis of mitral valve prolapse: comparison with echocardiography <sup/>. Radiology. https://doi.org/10.1148/radiol.2541090393
Tsianaka T, Matziris I, Kobe A et al (2021) Mitral annular disjunction in patients with severe aortic stenosis: extent and reproducibility of measurements with computed tomography. Eur J Radiol Open. https://doi.org/10.1016/j.ejro.2021.100335
Toh H, Mori S, Izawa Y et al (2021) Prevalence and extent of mitral annular disjunction in structurally normal hearts: comprehensive 3D analysis using cardiac computed tomography. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jeab022
Koo HJ, Kang J-W, Oh SY et al (2019) Cardiac computed tomography for the localization of mitral valve prolapse: scallop-by-scallop comparisons with echocardiography and intraoperative findings. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jey139
Esposito A, Palmisano A, Barbera M et al (2019) Cardiac computed tomography in troponin-positive chest pain. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2018.08.013
Vignale D, Palmisano A, Colantoni C et al (2023) Toward a One-Stop Shop CT Protocol in Acute Chest Pain Syndrome. Radiology. https://doi.org/10.1148/radiol.220844
Palmisano A, Vignale D, Tadic M et al (2022) Myocardial late contrast enhancement CT in troponin-positive acute chest pain syndrome. Radiology. https://doi.org/10.1148/radiol.211288
Palmisano A, Vignale D, Benedetti G et al (2020) Late iodine enhancement cardiac computed tomography for detection of myocardial scars: impact of experience in the clinical practice. Radiol Med. https://doi.org/10.1007/s11547-019-01108-7
Piroli F, Boccellino A, Ingallina G et al (2023) Feasibility and reliability of comprehensive three-dimensional transoesophageal echocardiography screening process for transcatheter mitral valve replacement. Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jead015
Vignale D, Palmisano A, Gnasso C et al (2023) Extracellular volume fraction (ECV) derived from pre-operative computed tomography predicts prognosis in patients undergoing transcatheter aortic valve implantation (TAVI). Eur Heart J Cardiovasc Imaging. https://doi.org/10.1093/ehjci/jead040
Palmisano A, Vignale D, Peretto G et al (2021) Hybrid FDG-PET/MR or FDG-PET/CT to detect disease activity in patients with persisting arrhythmias after myocarditis. JACC Cardiovasc Imaging. https://doi.org/10.1016/j.jcmg.2020.03.009
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Antonio Esposito.
Conflict of interest
Marco Francone is European Radiology’s section editor of Cardiac in the Scientific editorial board. He has not taken part in the review or selection process of this article.
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Anna Palmisano is a member of the European Radiology Experimental scientific editorial board.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was not required for this study because it is a review article.
Ethical approval
Institutional Review Board approval was not required because it is a review article.
Study subjects or cohorts overlap
No original data are included in the review.
Methodology
• narrative review
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Esposito, A., Gatti, M., Trivieri, M.G. et al. Imaging for the assessment of the arrhythmogenic potential of mitral valve prolapse. Eur Radiol (2023). https://doi.org/10.1007/s00330-023-10413-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00330-023-10413-9