Abstract
Objective
The current study aimed to evaluate the clinical practice for hemodynamic tissue signature (HTS) method in IDH genotype prediction in three groups derived from high-grade gliomas.
Methods
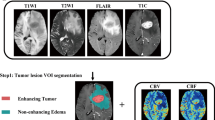
Preoperative MRI examinations of 44 patients with known grade and IDH genotype were assigned into three study groups: glioblastoma multiforme, grade III, and high-grade gliomas. Perfusion parameters were analyzed and were used to automatically draw the four reproducible habitats (high-angiogenic enhancing tumor habitats, low-angiogenic enhancing tumor habitats, infiltrated peripheral edema habitats, vasogenic peripheral edema habitats) related to vascular heterogeneity. These four habitats were then compared between inter-patient with IDH mutation and their wild-type counterparts at these three groups, respectively. The discriminating potential for HTS in assessing IDH mutation status prediction was assessed by ROC curves.
Results
Compared with IDH wild type, IDH mutation had significantly decreased relative cerebral blood volume (rCBV) at the high-angiogenic enhancing tumor habitats and low-angiogenic enhancing tumor habitats. ROC analysis revealed that the rCBVs in habitats had great ability to discriminate IDH mutation from their wild type in all groups. In addition, the Kaplan-Meier survival analysis yielded significant differences for the survival times observed from the populations dichotomized by low (< 4.31) and high (> 4.31) rCBV in the low-angiogenic enhancing tumor habitat.
Conclusions
The HTS method has been proven to have high prediction capabilities for IDH mutation status in high-grade glioma patients, providing a set of quantifiable habitats associated with tumor vascular heterogeneity.
Key Points
• The HTS method has a high accuracy for molecular stratification prediction for all subsets of HGG.
• The HTS method can give IDH mutation–related hemodynamic information of tumor-infiltrated and vasogenic edema.
• IDH-relevant rCBV difference in habitats will be a great prognosis factor in HGG.






Similar content being viewed by others
Abbreviations
- DSC-PWI:
-
Dynamic susceptibility contrast perfusion-weighted imaging
- FLAIR:
-
Fluid attenuation inversion recovery
- FLASH:
-
Fast low-angle shot
- FOV:
-
Field of view
- GBCA:
-
Gadolinium-based contrast agent
- GBM:
-
Glioblastoma multiforme
- HAT:
-
High-angiogenic enhancing tumor habitats
- HGG:
-
High-grade glioma
- HTS:
-
Hemodynamic tissue signatures
- IDH:
-
Isocitrate dehydrogenase
- IPE:
-
Infiltrated peripheral edema habitats
- LAT:
-
Low-angiogenic enhancing tumor habitats
- LGG:
-
Low-grade glioma
- NPV:
-
Negative predictive value
- PPV:
-
Positive predictive value
- RM-ANOVA:
-
Repeated measure analysis of variance
- rCBF:
-
Relative cerebral blood flow
- rCBV:
-
Relative cerebral blood volume
- ROC:
-
Receiver operating characteristic
- ROI:
-
Regions of interest
- SPM:
-
Statistical parametric mapping
- SPSS:
-
Statistical Package for the Social Sciences
- SVM:
-
Support vector machine
- T1-CE:
-
T1-weighted contrast-enhanced
- VASRI:
-
Visually AcceSAble Rembrandt Images
- VPE:
-
Vasogenic peripheral edema habitats
References
Ostrom QT, Gittleman H, Stetson L, Virk SM, Barnholtz-Sloan JS (2015) Epidemiology of gliomas. Cancer Treat Res 163:1–14
Ostrom QT, Gittleman H, Liao P et al (2017) CBTRUS Statistical Report: primary brain and other central nervous system tumors diagnosed in the United States in 2010-2014. Neuro Oncol 19:v1–v88
Ostrom QT, Gittleman H, Truitt G, Boscia A, Kruchko C, Barnholtz-Sloan JS (2018) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2011-2015. Neuro Oncol 20:iv1–iv86
Linkous AG, Yazlovitskaya EM (2011) Angiogenesis in glioblastoma multiforme: navigating the maze. Anticancer Agents Med Chem 11:712–718
De Bonis P, Lofrese G, Anile C, Pompucci A, Vigo V, Mangiola A (2013) Radioimmunotherapy for high-grade glioma. Immunotherapy 5:647–659
Rohle D, Popovici-Muller J, Palaskas N et al (2013) An inhibitor of mutant IDH1 delays growth and promotes differentiation of glioma cells. Science 340:626–630
Gomes DNS, Cucolicchio IC, Mattos MD et al (2017) Is there a correlation between overall survival and the mutation profile of IDH1 and TERT in high grade glioma? A Brazilian experience results. Int J Radiat Oncol 99:E76–E76
Parsons DW, Jones S, Zhang X et al (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321:1807–1812
Price SJ, Boonzaier NR, Lupson V, Larkin T (2014) Idh-1 mutated glioblastomas have a less invasive phenotype than Idh-1 wild type glioblastomas: a diffusion tensor imaging study. Neuro Oncol 16
Hartmann C, Hentschel B, Wick W et al (2010) Patients with IDH1 wild type anaplastic astrocytomas exhibit worse prognosis than IDH1-mutated glioblastomas, and IDH1 mutation status accounts for the unfavorable prognostic effect of higher age: implications for classification of gliomas. Acta Neuropathol 120:707–718
Deng L, Xiong P, Luo Y et al (2018) Association between IDH1/2 mutations and brain glioma grade. Oncol Lett 16:5405–5409
Lai A, Kharbanda S, Pope WB et al (2011) Evidence for sequenced molecular evolution of IDH1 mutant glioblastoma from a distinct cell of origin. J Clin Oncol 29:4482–4490
Dang L, White DW, Gross S et al (2010) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 465:966–966
Xu W, Yang H, Liu Y et al (2011) Oncometabolite 2-hydroxyglutarate is a competitive inhibitor of alpha-ketoglutarate-dependent dioxygenases. Cancer Cell 19:17–30
Flavahan WA, Drier Y, Liau BB et al (2016) Insulator dysfunction and oncogene activation in IDH mutant gliomas. Nature 529:110
Dang L, White DW, Gross S et al (2009) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462:739–744
Alves TR, Lima FR, Kahn SA et al (2011) Glioblastoma cells: a heterogeneous and fatal tumor interacting with the parenchyma. Life Sci 89:532–539
Popov S, Jury A, Laxton R et al (2013) IDH1-associated primary glioblastoma in young adults displays differential patterns of tumour and vascular morphology. Plos One 8
Pirozzi CJ, Yan H (2018) Improved grading of IDH-mutated astrocytic gliomas. Nat Rev Neurol 14:383–384
Zhang L, He LQ, Lugano R et al (2018) IDH mutation status is associated with distinct vascular gene expression signatures in lower-grade gliomas. Neuro Oncol 20:1505–1516
Ellingson BM (2015) Radiogenomics and imaging phenotypes in glioblastoma: novel observations and correlation with molecular characteristics. Curr Neurol Neurosci Rep 15
Chow D, Chang P, Weinberg BD, Bota DA, Grinband J, Filippi CG (2018) Imaging genetic heterogeneity in glioblastoma and other glial tumors: review of current methods and future directions. AJR Am J Roentgenol 210:30–38
Gatenby RA, Grove O, Gillies RJ (2013) Quantitative imaging in cancer evolution and ecology. Radiology 269:8–15
Swanton C (2012) Intratumor heterogeneity: evolution through space and time. Cancer Res 72:4875–4882
Fuster-Garcia E, Juan-Albarracin J, Garcia-Ferrando GA, Marti-Bonmati L, Aparici-Robles F, Garcia-Gomez JM (2018) Improving the estimation of prognosis for glioblastoma patients by MR based hemodynamic tissue signatures. NMR Biomed:31
Juan-Albarracin J, Fuster-Garcia E, Perez-Girbes A et al (2018) Glioblastoma: vascular habitats detected at preoperative dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging predict survival. Radiology 287:944–954
Welker K, Boxerman J, Kalnin A et al (2015) ASFNR recommendations for clinical performance of MR dynamic susceptibility contrast perfusion imaging of the brain. AJNR Am J Neuroradiol 36:E41–E51
Boxerman JL, Schmainda KM, Weisskoff RM (2006) Relative cerebral blood volume maps corrected for contrast agent extravasation significantly correlate with glioma tumor grade, whereas uncorrected maps do not. AJNR Am J Neuroradiol 27:859–867
Wu O, Ostergaard L, Weisskoff RM, Benner T, Rosen BR, Sorensen AG (2003) Tracer arrival timing-insensitive technique for estimating flow in MR perfusion-weighted imaging using singular value decomposition with a block-circulant deconvolution matrix. Magn Reson Med 50:164–174
Juan-Albarracin J, Fuster-Garcia E, Manjon JV et al (2015) Automated glioblastoma segmentation based on a multiparametric structured unsupervised classification. PLoS One 10:e0125143
Oei MTH, Meijer FJA, Mordang JJ et al (2018) Observer variability of reference tissue selection for relative cerebral blood volume measurements in glioma patients. Eur Radiol 28:3902–3911
Qi ST, Yu L, Li HZ et al (2014) Isocitrate dehydrogenase mutation is associated with tumor location and magnetic resonance imaging characteristics in astrocytic neoplasms. Oncol Lett 7:1895–1902
Jalbert LE, Elkhaled A, Phillips JJ et al (2017) Metabolic profiling of IDH mutation and malignant progression in infiltrating glioma. Sci Rep 7
Su CL, Jiang JJ, Zhang S et al (2019) Radiomics based on multicontrast MRI can precisely differentiate among glioma subtypes and predict tumour-proliferative behaviour. Eur Radiol 29:1986–1996
Zhou M, Hall L, Goldgof D et al (2014) Radiologically defined ecological dynamics and clinical outcomes in glioblastoma multiforme: preliminary results. Transl Oncol 7:5–13
Caulo M, Panara V, Tortora D et al (2014) Data-driven grading of brain gliomas: a multiparametric MR imaging study. Radiology 272:494–503
Schoenegger K, Oberndorfer S, Wuschitz B et al (2009) Peritumoral edema on MRI at initial diagnosis: an independent prognostic factor for glioblastoma? Eur J Neurol 16:874–878
Pope WB, Sayre J, Perlina A, Villablanca JP, Mischel PS, Cloughesy TF (2005) MR imaging correlates of survival in patients with high-grade gliomas. AJNR Am J Neuroradiol 26:2466–2474
Lemee JM, Clavreul A, Menei P (2015) Intratumoral heterogeneity in glioblastoma: don’t forget the peritumoral brain zone. Neuro Oncol 17:1322–1332
Das D, Yoon B, Golden L et al (2017) NIMG-37. Correlation of VASARI-based MRI phenotypes with MGMT and IDH status across glioma grades: a statistical analysis in 372 patients. Neuro Oncol 19:150–150
Artzi M, Bokstein F, Blumenthal DT et al (2014) Differentiation between vasogenic-edema versus tumor-infiltrative area in patients with glioblastoma during bevacizumab therapy: a longitudinal MRI study. Eur J Radiol 83:1250–1256
Min ZG, Niu C, Rana N, Ji HM, Zhang M (2013) Differentiation of pure vasogenic edema and tumor-infiltrated edema in patients with peritumoral edema by analyzing the relationship of axial and radial diffusivities on 3.0T MRI. Clin Neurol Neurosurg 115:1366–1370
Patel SH, Poisson LM, Brat DJ et al (2017) T2-FLAIR mismatch, an imaging biomarker for IDH and 1p/19q status in lower-grade gliomas: a TCGA/TCIA project. Clin Cancer Res 23:6078–6085
Wu S, Meng J, Yu Q, Li P, Fu S (2019) Radiomics-based machine learning methods for isocitrate dehydrogenase genotype prediction of diffuse gliomas. J Cancer Res Clin Oncol 145:543–550
Lu CF, Hsu FT, Hsieh KL et al (2018) Machine learning-based radiomics for molecular subtyping of gliomas. Clin Cancer Res 24:4429–4436
Arita H, Kinoshita M, Kawaguchi A et al (2018) Lesion location implemented magnetic resonance imaging radiomics for predicting IDH and TERT promoter mutations in grade II/III gliomas. Sci Rep:8
Hsieh KLC, Chen CY, Lo CM (2017) Radiomic model for predicting mutations in the isocitrate dehydrogenase gene in glioblastomas. Oncotarget 8:45888–45897
Levner I, Drabycz S, Roldan G, De Robles P, Cairncross JG, Mitchell R (2009) Predicting MGMT methylation status of glioblastomas from MRI texture. Med Image Comput Comput Assist Interv 12:522–530
Zhang X, Tian Q, Wang L et al (2018) Radiomics strategy for molecular subtype stratification of lower-grade glioma: detecting IDH and TP53 mutations based on multimodal MRI. J Magn Reson Imaging 48:916–926
Chang K, Bai HX, Zhou H et al (2018) Residual convolutional neural network for the determination of IDH status in low- and high-grade gliomas from MR imaging. Clin Cancer Res 24:1073–1081
Carrillo JA, Lai A, Nghiemphu PL et al (2012) Relationship between tumor enhancement, edema, IDH1 mutational status, MGMT promoter methylation, and survival in glioblastoma. AJNR Am J Neuroradiol 33:1349–1355
Smits M, van den Bent MJ (2017) Imaging correlates of adult glioma genotypes. Radiology 284:316–331
Law M, Young RJ, Babb JS et al (2008) Gliomas: predicting time to progression or survival with cerebral blood volume measurements at dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. Radiology 247:490–498
Hirai T, Murakami R, Nakamura H et al (2008) Prognostic value of perfusion MR imaging of high-grade astrocytomas: long-term follow-up study. AJNR Am J Neuroradiol 29:1505–1510
Jain R, Poisson L, Narang J et al (2013) Genomic mapping and survival prediction in glioblastoma: molecular subclassification strengthened by hemodynamic imaging biomarkers. Radiology 267:212–220
Hartmann C, Hentschel B, Simon M et al (2013) Long-term survival in primary glioblastoma with versus without isocitrate dehydrogenase mutations. Clin Cancer Res 19:5146–5157
Han K, Ren M, Wick W et al (2014) Progression-free survival as a surrogate endpoint for overall survival in glioblastoma: a literature-based meta-analysis from 91 trials. Neuro Oncol 16:696–706
Funding
This work was supported by the National Key R&D Program of China (2018YFC0115004) and grants from the Natural Science Foundation of China (81871421 and 81571660).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is W.G. Z., Ph.D, M.D., the Chief Director of Radiology Department of Army Medical Center.
Conflict of interest
One of the authors of this manuscript (Xiaoyue Zhou) is an employee of Siemens Healthineers. The remaining authors declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors (H.W.) has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• experimental
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1015 kb)
Rights and permissions
About this article
Cite this article
Wu, H., Tong, H., Du, X. et al. Vascular habitat analysis based on dynamic susceptibility contrast perfusion MRI predicts IDH mutation status and prognosis in high-grade gliomas. Eur Radiol 30, 3254–3265 (2020). https://doi.org/10.1007/s00330-020-06702-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-06702-2