Abstract
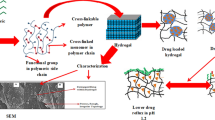
Purpose of the current research work was to synthesize gelatin based hydrogels for the controlled release of salbutamol sulphate. Free radical polymerization technique was used for the synthesis of hydrogels. Gelatin was cross-linked chemically with monomer methacrylic acid using methylene bisacrylamide as cross-linker while ammonium persulphate and sodium metabisulphite were used as initiators. All the formulations were evaluated through Fourier transform infrared spectroscopy, differential scanning calorimetry, thermo gravimetric analysis and scanning electron microscopy. Swelling properties of formulated hydrogels were also evaluated in both pH 7.4 and 1.2. For drug release study, USP peddle method was used and release study was carried out in both pH 7.4 and 1.2. Fourier transform infrared spectroscopy confirmed that drug salbutamol sulphate is compatible with the formulated system. Moreover, thermal stability was also confirmed by TG/DSC studies providing the fact that thermal stability of the formulated hydrogels is high as compared to individual content. Sol–gel fraction confirmed that gel content increased as the concentrations of the polymer, monomer and cross-inker were increased. All formulations showed profound gel fraction. Similarly, pH dependent swelling was observed, experiencing higher swelling at higher pH as compared to lower pH. Toxicity study was also conducted which endorsed the safety of the hydrogels as no toxic effect was observed on vital organs.







Similar content being viewed by others
References
Li Y et al (2013) Magnetic hydrogels and their potential biomedical applications. Adv Func Mater 23(6):660–672
Shah SA et al (2019) pH-responsive CAP-co-poly(methacrylic acid)-based hydrogel as an efficient platform for controlled gastrointestinal delivery: fabrication, characterization, in vitro and in vivo toxicity evaluation. Drug Deliv Transl Res 9(2):555–577
Ghorpade VS, Yadav AV, Dias RJ (2017) Citric acid crosslinked β-cyclodextrin/carboxymethylcellulose hydrogel films for controlled delivery of poorly soluble drugs. Carbohyd Polym 164:339–348
Dragan ES, Cocarta AI, Gierszewska M (2016) Designing novel macroporous composite hydrogels based on methacrylic acid copolymers and chitosan and in vitro assessment of lysozyme controlled delivery. Colloids Surf B 139:33–41
Deepa G et al (2012) Cross-linked acrylic hydrogel for the controlled delivery of hydrophobic drugs in cancer therapy. Int J Nanomed 7:4077–4088
Erol K et al (2019) Effect of immobilization on the activity of catalase carried by poly (HEMA-GMA) cryogels. Int J Biol Macromol 123:738–743
Köse K et al (2017) Affinity purification lipase from wheat germ: comparison of hydrophobic and metal chelation effect. Artif Cells Nanomed Biotechnol 45(3):574–583
Kim JK et al (2014) Natural and synthetic biomaterials for controlled drug delivery. Arch Pharmacal Res 37(1):60–68
Gupta P, Vermani K, Garg S (2002) Hydrogels: from controlled release to pH-responsive drug delivery. Drug Discov Today 7(10):569–579
Anirudhan TS, Mohan AM (2018) Novel pH sensitive dual drug loaded-gelatin methacrylate/methacrylic acid hydrogel for the controlled release of antibiotics. Int J Biol Macromol 110:167–178
Cheng N-C et al (2017) Sustained release of adipose-derived stem cells by thermosensitive chitosan/gelatin hydrogel for therapeutic angiogenesis. Acta Biomater 51:258–267
Casolaro M, Casolaro I, Lamponi S (2012) Stimuli-responsive hydrogels for controlled pilocarpine ocular delivery. Eur J Pharm Biopharm 80(3):553–561
Rey-Rico A, Madry H, Cucchiarini M (2016) Hydrogel-based controlled delivery systems for articular cartilage repair. Biomed Res Int 2016:1215263
Jing Z et al (2014) Preparation and adsorption properties of a novel superabsorbent based on multiwalled carbon nanotubes–xylan composite and poly (methacrylic acid) for methylene blue from aqueous solution. Polym Compos 35(8):1516–1528
Bilgin E et al (2018) Use of nicotinamide decorated polymeric cryogels as heavy metal sweeper. Environ Sci Pollut Res 25(27):27614–27627
Saidi, M., A. Dabbaghi, and S. Rahmani (2019) Swelling and drug delivery kinetics of click-synthesized hydrogels based on various combinations of PEG and star-shaped PCL: influence of network parameters on swelling and release behavior. Polymer Bulletin. 1–22.
McCain ML et al (2014) Micromolded gelatin hydrogels for extended culture of engineered cardiac tissues. Biomaterials 35(21):5462–5471
Jiang Y et al (2018) Preparation of cellulose nanofiber-reinforced gelatin hydrogel and optimization for 3D printing applications. BioResour 13(3):5909–5924
Xing Q et al (2014) Increasing mechanical strength of gelatin hydrogels by divalent metal ion removal. Sci Rep 4(1):4706
Bukhari SMH (2015) Synthesis and characterization of chemically cross-linked acrylic acid/gelatin hydrogels: effect of pH and composition on swelling and drug release. Int J Polym Sci 2015:187961
Dong Y et al (2017) Injectable and tunable gelatin hydrogels enhance stem cell retention and improve cutaneous wound healing. Adv Func Mater 27(24):1606619
Treesuppharat W et al (2017) Synthesis and characterization of bacterial cellulose and gelatin-based hydrogel composites for drug-delivery systems. Biotechnol Rep 15:84–91
Kalshetti PP et al (2012) Hydrogels as a drug delivery system and applications: a review. Int J Pharm Pharm Sci 4(1):1–7
Prasanth VV et al (2014) Effect of permeation enhancers in the mucoadhesive buccal patches of salbutamol sulphate for unidirectional buccal drug delivery. Res Pharm Sci 9(4):259–268
Kumar P et al (2015) Design and comparative in-vitro and in-vivo evaluation of starch-acrylate graft copolymer based salbutamol sulphate sustained release tablets. Asian J Pharm Sci 10(3):239–246
Mansur HS et al (2008) FTIR spectroscopy characterization of poly (vinyl alcohol) hydrogel with different hydrolysis degree and chemically crosslinked with glutaraldehyde. Mater Sci Eng C 28(4):539–548
Mundargi RC, Rangaswamy V, Aminabhavi TM (2011) Poly (N-vinylcaprolactam-co-methacrylic acid) hydrogel microparticles for oral insulin delivery. J Microencapsul 28(5):384–394
Köse K, Köse DA (2017) Removal of DDE by exploiting the alcoho-phobic interactions. Environ Sci Pollut Res 24(10):9187–9193
Jeong B, Kim SW, Bae YH (2002) Thermosensitive sol–gel reversible hydrogels. Adv Drug Deliv Rev 54(1):37–51
Khan S, Ranjha NM (2014) Effect of degree of cross-linking on swelling and on drug release of low viscous chitosan/poly (vinyl alcohol) hydrogels. Polym Bull 71(8):2133–2158
Kashif B et al (2017) Oxaliplatin-loaded crosslinked polymeric network of chondroitin sulfate-co-poly(methacrylic acid) for colorectal cancer: Its toxicological evaluation. J Appl Polym Sci 134(38):45312
Pachuau L, Sarkar S, Mazumder B (2008) Formulation and evaluation of matrix microspheres for simultaneous delivery of salbutamol sulphate and theophylline. Trop J Pharm Res 7(2):995–1002
Akin H, Hasirci N (1995) Preparation and characterization of crosslinked gelatin microspheres. J Appl Polym Sci 58(1):95–100
Ki CS et al (2005) Characterization of gelatin nanofiber prepared from gelatin–formic acid solution. Polymer 46(14):5094–5102
Rokhade AP et al (2006) Semi-interpenetrating polymer network microspheres of gelatin and sodium carboxymethyl cellulose for controlled release of ketorolac tromethamine. Carbohyd Polym 65(3):243–252
Panic VV, Velickovic SJ (2014) Removal of model cationic dye by adsorption onto poly (methacrylic acid)/zeolite hydrogel composites: kinetics, equilibrium study and image analysis. Sep Purif Technol 122:384–394
Panic VV et al (2013) Poly(methacrylic acid) based hydrogels as sorbents for removal of cationic dye basic yellow 28: kinetics, equilibrium study and image analysis. Chem Eng J 217:192–204
Varghese JS, Chellappa N, Fathima NN (2014) Gelatin–carrageenan hydrogels: role of pore size distribution on drug delivery process. Colloids Surf B 113:346–351
Parvez S et al (2012) Preparation and characterization of artificial skin using chitosan and gelatin composites for potential biomedical application. Polym Bull 69(6):715–731
Dergunov SA et al (2005) Radiation synthesis and characterization of stimuli-sensitive chitosan–polyvinyl pyrrolidone hydrogels. Radiat Phys Chem 72(5):619–623
Ranjha NM et al (2010) Preparation and characterization of hybrid pH-sensitive hydrogels of chitosan-co-acrylic acid for controlled release of verapamil. J Mater Sci Mater Med 21(10):2805–2816
Van Vlierberghe S et al (2007) Porous gelatin hydrogels: 1. Cryog Form Struct Anal Biomacromolecules 8(2):331–337
Bukhari SMH (2015) Synthesis and characterization of chemically cross-linked acrylic acid/gelatin hydrogels: effect of pH and composition on swelling and drug release. Int J Polym Sci 2015:1–15
Gaballa HA et al (2013) Synthesis and characterization of physically crosslinked N-vinylcaprolactam, acrylic acid, methacrylic acid, and N, N-dimethylacrylamide hydrogels. J Polym Sci Part B Polym Phys 51(21):1555–1564
Junior CRF (2020) On the preparation and physicochemical properties of pH-responsive hydrogel nanocomposite based on poly(acid methacrylic)/laponite RDS. Mater Today Commun 23:100936
Lopes CM, Felisberti MI (2003) Mechanical behaviour and biocompatibility of poly (1-vinyl-2-pyrrolidinone)–gelatin IPN hydrogels. Biomaterials 24(7):1279–1284
Burugapalli K (2001) Interpenetrating polymer networks based on poly (acrylic acid) and gelatin. I: Swelling and thermal behavior. J Appl Polym Sci 82(1):217–227
Ramaraj B, Radhakrishnan G (1994) Interpenetrating hydrogel networks based on gelatin and polyacrylamide: synthesis, swelling, and drug release analysis. J Appl Polym Sci 52(7):837–846
Brazel CS, Peppas NA (1995) Synthesis and characterization of thermo-and chemomechanically responsive poly (n-isopropylacrylamide-co-methacrylic acid) hydrogels. Macromolecules 28(24):8016–8020
Chen S et al (2007) Structure and properties of the polyelectrolyte complex of chitosan with poly (methacrylic acid). Polym Int 56(10):1305–1312
Chen S et al (2005) Synthesis and swelling properties of pH-sensitive hydrogels based on chitosan and poly (methacrylic acid) semi-interpenetrating polymer network. J Appl Polym Sci 98(4):1720–1726
Brannon-Peppas L, Peppas NA (1990) Dynamic and equilibrium swelling behaviour of pH-sensitive hydrogels containing 2-hydroxyethyl methacrylate. Biomaterials 11(9):635–644
Suhag GS, Bhatnagar A, Singh H (2008) Poly (hydroxyethyl methacrylate)-based co-polymeric hydrogels for transdermal delivery of salbutamol sulphate. J Biomater Sci Polym Ed 19(9):1189–1200
Tyagi P et al (2011) Synthesis and characterization of poly (HEMA-MAA) hydrogel carrier for oral delivery of insulin. J Appl Polym Sci 122(3):2004–2012
Sadeghi M, Hosseinzadeh H (2010) Swelling behaviour of a novel protein-based super absorbent hydrogel composed of poly (methacrylic acid) and collagen. Asian J Chem 22(9):6734
Zhang S et al (2014) Organic/inorganic superabsorbent hydrogels based on xylan and montmorillonite. J Nanomater 2014:2
Pourjavadi A, Harzandi AM, Hosseinzadeh H (2004) Modified carrageenan 3. Synthesis of a novel polysaccharide-based superabsorbent hydrogel via graft copolymerization of acrylic acid onto kappa-carrageenan in air. Eur Polym J 40(7):1363–1370
Pourjavadi A, Barzegar S, Mahdavinia GR (2006) MBA-crosslinked Na-Alg/CMC as a smart full-polysaccharide superabsorbent hydrogels. Carbohyd Polym 66(3):386–395
Sadeghi M, Hosseinzadeh H (2010) Synthesis and super-swelling behavior of a novel low salt-sensitive protein-based superabsorbent hydrogel: collagen-g-poly (AMPS). Turk J Chem 34(5):739–752
Zhang L-M et al (2005) A new class of starch-based hydrogels incorporating acrylamide and vinyl pyrrolidone: effects of reaction variables on water sorption behavior. J Bioact Compat Polym 20(5):491–501
Pourjavadi A, Hosseinzadeh H, Sadeghi M (2007) Synthesis, characterization and swelling behavior of gelatin-g-poly (sodium acrylate)/kaolin superabsorbent hydrogel composites. J Compos Mater 41(17):2057–2069
Sairam M et al (2006) Encapsulation efficiency and controlled release characteristics of crosslinked polyacrylamide particles. Int J Pharm 320(1):131–136
Lee PI (1985) Kinetics of drug release from hydrogel matrices. J Control Releas 2:277–288
Changez M et al (2003) The effect of composition of poly (acrylic acid)–gelatin hydrogel on gentamicin sulphate release: in vitro. Biomaterials 24(4):527–536
Narayani R, Rao KP (1994) Controlled release of anticancer drug methotrexate from biodegradable gelatin microspheres. J Microencapsul 11(1):69–77
Varshosaz J, Hajian M (2004) Characterization of drug release and diffusion mechanism through hydroxyethylmethacrylate/methacrylic acid pH-sensitive hydrogel. Drug Deliv 11(1):53–58
Garcıa D et al (2004) Timolol maleate release from pH-sensible poly (2-hydroxyethyl methacrylate-co-methacrylic acid) hydrogels. Eur Polymer J 40(8):1683–1690
Pourjavadi A, Barzegar S (2009) Smart pectin-based superabsorbent hydrogel as a matrix for ibuprofen as an oral non-steroidal anti-inflammatory drug delivery. Starch-Stärke 61(3–4):173–187
Khan KU, Akhtar N, Minhas MU (2020) Poloxamer-407-Co-Poly (2-Acrylamido-2-Methylpropane Sulfonic Acid) cross-linked nanogels for solubility enhancement of olanzapine: synthesis, characterization, and toxicity evaluation. AAPS PharmSciTech 21:1–15
Badshah SF (2020) Porous and highly responsive cross-linked β-cyclodextrin based nanomatrices for improvement in drug dissolution and absorption. Life Sci 2676:118931
Acknowledgements
The authors are thankful for the contribution of the Islamia University of Bahawalpur Pakistan for providing the finances and facilities for performing studies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rafique, N., Ahmad, M., Minhas, M.U. et al. Designing gelatin-based swellable hydrogels system for controlled delivery of salbutamol sulphate: characterization and toxicity evaluation. Polym. Bull. 79, 4535–4561 (2022). https://doi.org/10.1007/s00289-021-03629-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-03629-6