Abstract
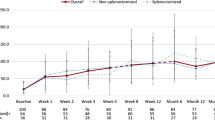
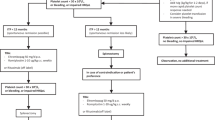
Primary immune thrombocytopenia (ITP) is an intriguing autoimmune disease characterized by autoantibodies against platelets and megakaryocytes. Clinical outcomes, response to treatment, and chronicity predictors were investigated. Patients with newly diagnosed primary ITP treated at a hematology referral center from 2008 to 2018 with complete medical and recent medication history were stratified by age as children < 16 years and adults > 16 years. Responses to treatment including steroids, splenectomy, rituximab, and eltrombopag were classified as response (R) and complete (CR). Factors for developing chronic ITP were determined by multiple regression with uni- and multivariate analysis. p < 0.05 was considered significant. A total of 175 patients were included, 52 children and 123 adults; women predominated with 57.7%. Response to first-line treatment in the whole cohort was 86.18%, CR 43.42% and R 42.76%. The initial response to steroids alone was 83.9% (n = 52/62), rituximab plus high-dose dexamethasone (HDD) 87.2% (n = 34/39), eltrombopag plus HDD 90.9% (n = 10/11), and children receiving IVIG alone 100% (n = 8/8); 9 children were under clinical observation and achieved spontaneous response; loss of response was documented in 15.21% children and 28.3% adults with a median time of 15.95 and 4.07 months respectively; 37.39% of adults and 30.76% of children progressed to a chronic course. Platelets ≥ 20 × 109/L and age ≥ 6 years were risk factors for chronic ITP in the univariate analysis in the adult and children groups, respectively. Clinical course and treatment outcomes for ITP are considerably heterogeneous. Higher platelet counts at diagnosis in adults and age ≥ 6 years in children were associated with an increased risk of chronicity.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Frederiksen H, Christiansen CF, Norgaard M (2012) Risk and prognosis of adult primary immune thrombocytopenia. Expert Rev Hematol 5:219–228. https://doi.org/10.1586/ehm.12.7
Grimaldi-Bensouda L, Nordon C, Michel M, Viallard JF, Adoue D, Magy-Bertrand N, Durand JM, Quittet P, Fain O, Bonnotte B, Morin AS, Morel N, Costedoat-Chalumeau N, Pan-Petesch B, Khellaf M, Perlat A, Sacre K, Lefrere F, Abenhaim L, Godeau B, Group for the PGRx-ITP Study (2016) Immune thrombocytopenia in adults: a prospective cohort study of clinical features and predictors of outcome. Haematologica 101:1039–1045. https://doi.org/10.3324/haematol.2016.146373
Heitink-Pollé KMJ, Nijsten J, Boonacker CWB, De Haas M, Bruin MCA (2014) Clinical and laboratory predictors of chronic immune thrombocytopenia in children: a systematic review and meta-analysis. Blood 124:3295–3307. https://doi.org/10.1182/blood-2014-04-570127
Rodeghiero F, Stasi R, Gernsheimer T, Michel M, Provan D, Arnold DM, Bussel JB, Cines DB, Chong BH, Cooper N, Godeau B, Lechner K, Mazzucconi MG, McMillan R, Sanz MA, Imbach P, Blanchette V, Kühne T, Ruggeri M, George JN (2009) Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood 113:2386–2393. https://doi.org/10.1182/blood-2008-07-162503
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ (2019) Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv 3:3780–3817. https://doi.org/10.1182/bloodadvances.2019000812
Swinkels M, Rijkers M, Voorberg J, Vidarsson G, Leebeek FWG, Jansen AJG (2018) Emerging concepts in immune thrombocytopenia. Front Immunol 9:880. https://doi.org/10.3389/fimmu.2018.00880
Harrington WJ, Sprague CC, Moore CV, Aulvin RC, Dubach R (1953) Immunologic mechanisms in idiopathic and neonatal thrombocytopenic purpura. Ann Intern Med 38:433–469. https://doi.org/10.1258/acb.2012.201227
Nugent D, McMillan R, Nichol JL, Slichter SJ (2009) Pathogenesis of chronic immune thrombocytopenia: increased platelet destruction and/or decreased platelet production. Br J Haematol 146:585–596. https://doi.org/10.1111/j.1365-2141.2009.07717.x
Neunert C, Terrell DR, Arnold DM, Buchanan G, Cines DB, Cooper N, Cuker A, Despotovic JM, George JN, Grace RF, Kühne T, Kuter DJ, Lim W, McCrae KR, Pruitt B, Shimanek H, Vesely SK (2019) American Society of Hematology 2019 guidelines for immune thrombocytopenia. Blood Adv 3:3829–3866. https://doi.org/10.1182/bloodadvances.2019000966
Gómez-Almaguer D, Colunga-Pedraza PR, Gómez-De León A, Gutiérrez-Aguirre CH, Cantú-Rodríguez OG, Jaime-Pérez JC (2019) Eltrombopag, low-dose rituximab, and dexamethasone combination as frontline treatment of newly diagnosed immune thrombocytopaenia. Br J Haematol 184:288–290. https://doi.org/10.1111/bjh.15070
Gómez-Almaguer D, Tarín-Arzaga L, Moreno-Jaime B, Jaime-Pérez JC, Ceballos-López AA, Ruiz-Argüelles GJ, Ruiz-Delgado GJ, Cantú-Rodríguez OG, Gutiérrez-Aguirre CH, Sánchez-Cárdenas M (2013) High response rate to low-dose rituximab plus high-dose dexamethasone as frontline therapy in adult patients with primary immune thrombocytopenia. Eur J Haematol 90:494–500. https://doi.org/10.1111/ejh.12102
Gómez-Almaguer D, Herrera-Rojas MA, Jaime-Pérez JC, Gómez-de León A, Cantú-Rodríguez OG, Gutiérrez-Aguirre CH, Tarín-Arzaga L, Hernández-Reyes J, Ruiz-Arguelles GJ (2014) Eltrombopag and high-dose dexamethasone as frontline treatment of newly diagnosed immune thrombocytopenia in adults. Blood 123:3906–3908. https://doi.org/10.1182/blood-2014-01-549360
Gómez-Almaguer D (2018) Eltrombopag-based combination treatment for immune thrombocytopenia. Ther Adv Hematol 9:309–317. https://doi.org/10.1177/2040620718798798
Jaime-Pérez JC, Treviño-Reyna G, Aguilar-Calderón P, Cantú-Rodríguez OG, Marfil-Rivera LJ, Gómez-Almaguer D (2018) Contributions of a regional approach to document hematologic disease in Mexico: a 10-year experience in an open population. Hematology 23:803–809. https://doi.org/10.1080/10245332.2018.1498166
Mazzucconi MG, Fazi P, Bernasconi S, de Rossi G, Leone G, Gugliotta L, Vianelli N, Avvisati G, Rodeghiero F, Amendola A, Baronci C, Carbone C, Quattrin S, Fioritoni G, D'Alfonso G, Mandelli F, for the Gruppo Italiano Malattie EMatologiche dell'Adulto (GIMEMA) Thrombocytopenia Working Party (2007) Therapy with high-dose dexamethasone (HD-DXM) in previously untreated patients affected by idiopathic thrombocytopenic purpura: a GIMEMA experience. Blood 109:1401–1407. https://doi.org/10.1182/blood-2005-12-015222
Nomura S (2016) Advances in diagnosis and treatments for immune thrombocytopenia. Clin Med Insights Blood Disord 9:15–22. https://doi.org/10.4137/CMBD.S39643
Chang H, Tang TC, Hung YS, Li PL, Kuo MC, Wu JH, Wang PN (2018) Immune thrombocytopenia: effectiveness of frontline steroids and comparison of azathioprine, splenectomy, and rituximab as second-line treatment. Eur J Haematol 101:549–555. https://doi.org/10.1111/ejh.13144
Bussel JB, Eldor A, Kelton JG, Varon D, Brenner B, Gillis S, Angiolillo A, Kulkarni R, Abshire T, Kelleher J, the IGIV-C in ITP Study Group (2004) IGIV-C, a novel intravenous immunoglobulin: evaluation of safety, efficacy, mechanisms of action, and impact on quality of life. Thromb Haemost 91:771–778. https://doi.org/10.1160/TH03-10-0650
Arnold DM (2013) Positioning new treatments in the management of immune thrombocytopenia. Pediatr Blood Cancer 60:S19–S22. https://doi.org/10.1002/pbc.24341
Zimmer J (2019) Danazol as a second-line treatment for immune thrombocytopenic purpura. Eur J Haematol 102:97–98. https://doi.org/10.1111/ejh.13184
Zaja F, Battista ML, Pirrotta MT, Palmieri S, Montagna M, Vianelli N, Marin L, Cavallin M, Bocchia M, Defina M, Ippoliti M, Ferrara F, Patriarca F, Avanzini MA, Regazzi M, Baccarani M, Isola M, Soldano F, Fanin R (2008) Lower dose rituximab is active in adults patients with idiopathic thrombocytopenic purpura. Haematologica 93:930–933. https://doi.org/10.3324/haematol.12206
Newland AC, Sánchez-González B, Rejtő L, Egyed M, Romanyuk N, Godar M, Verschueren K, Gandini D, Ulrichts P, Beauchamp J, Dreier T, Ward ES, Michel M, Liebman HA, Haard H, Leupin N, Kuter DJ (2020) Phase 2 study of efgartigimod, a novel FcRn antagonist, in adult patients with primary immune thrombocytopenia. Am J Hematol 95:178–187. https://doi.org/10.1002/ajh.25680
Kühne T, Berchtold W, Michaels LA et al (2011) Newly diagnosed immune thrombocytopenia in children and adults: a comparative prospective observational registry of the intercontinental cooperative immune thrombocytopenia study group. Haematologica 96:1831–1837. https://doi.org/10.3324/haematol.2011.050799
Nobuko N, Hirokazu K, Akuta K et al (2020) Reevaluation of platelet function in chronic immune thrombocytopenia: impacts of platelet size, platelet-associated anti-αIIbβ3 antibodies and thrombopoietin receptor agonists. Br J Haematol 189:760–771. https://doi.org/10.1111/bjh.16439
Boulware R, Refaai MA (2020) Why do patients with immune thrombocytopenia (ITP) experience lower bleeding events despite thrombocytopenia? Thromb Res 187:154–158. https://doi.org/10.1016/j.thromres.2020.01.020
Cooper N, Bussel J (2006) The pathogenesis of immune thrombocytopaenic purpura. Br J Haematol 133:364–374. https://doi.org/10.1111/j.1365-2141.2006.06024.x
Cines DB, Blanchette VS (2002) Immune thrombocytopenic purpura. N Engl J Med 346:995–1008. https://doi.org/10.1056/NEJMra010501
Venkatesha SH, Durai M, Moudgil KD (2015) Epitope spreading in autoimmune diseases. In: Shoenfeld Y, Agmon-Levin N, Rose NR (eds) Infection and Autoimmunity, 2nd. edn. Academic press, Amsterdam, pp 45–68. https://doi.org/10.1016/B978-0-444-63269-2.00003-9\
Al-Samkari H, Rosovsky RP, Leaf RSK et al (2020) A modern reassessment of glycoprotein-specific direct platelet autoantibody testing in immune thrombocytopenia. Blood Adv 4:9–18. https://doi.org/10.1182/bloodadvances.2019000868
Lambert MP, Gernsheimer TB (2017) Clinical updates in adult immune thrombocytopenia. Blood 129:2829–2835. https://doi.org/10.1182/blood-2017-03-754119
Monzón Manzano E, Álvarez Román MT, Justo Sanz R, Fernández Bello I, Hernández D, Martín Salces M, Valor L, Rivas Pollmar I, Butta NV, Jiménez Yuste V (2020) Platelet and immune characteristics of immune thrombocytopaenia patients non-responsive to therapy reveal severe immune dysregulation. Br J Haematol 189:943–953. https://doi.org/10.1111/bjh.16459
Li J, Van Der Wal DE, Zhu G et al (2015) Desialylation is a mechanism of Fc-independent platelet clearance and a therapeutic target in immune thrombocytopenia. Nat Commun 6:7737. https://doi.org/10.1038/ncomms8737
Acknowledgments
We thank Sergio Lozano-Rodriguez, MD, for his critical review of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Lorena Salazar-Cavazos, Patrizia Aguilar-Calderón, and Raúl A. Jiménez-Castillo. The first draft of the manuscript was written by Eugenia M. Ramos-Dávila, David Gómez-Almaguer, and José Carlos Jaime-Pérez. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The protocol of the study was approved by the Ethics and Research Committee of the institution and is in full compliance with the principles of the Declaration of Helsinki as revised in 2013.
Consent to participate
The Ethics and Research Committee of the institution waved this consent due to its retrospective nature.
Consent for publication
The Ethics and Research Committee of the institution waved this consent due to its retrospective nature and anonymized information.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jaime-Pérez, J.C., Aguilar-Calderón, P., Jiménez-Castillo, R.A. et al. Treatment outcomes and chronicity predictors for primary immune thrombocytopenia: 10-year data from an academic center. Ann Hematol 99, 2513–2520 (2020). https://doi.org/10.1007/s00277-020-04257-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-020-04257-2