Abstract
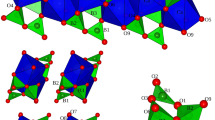
A synthetic \(\hbox {CaCO}_{3}\)–\(\hbox {SrCO}_{3}\) solid solution with composition \(\hbox {Ca}_{0.82}\hbox {Sr}_{0.18}\) \(\hbox {CO}_{3}\) was investigated by single-crystal X-ray diffraction in the pressure range between 0 and 22 GPa using different pressure-transmitting media. The samples were compressed in DACs using Ne up to \(\sim\)9 GPa and Ar up to \(\sim\)22 GPa. At ambient conditions, \(\hbox {Ca}_{0.82}\hbox {Sr}_{0.18}\) \(\hbox {CO}_{3}\) crystallizes in a monoclinic structure, isostructural to \(\hbox {CaCO}_{3}\)-II, Sr-calcite-II (Sr-CC-II), with space group \(P2_1/c\), 4 formula units per unit cell, Z, \(a = 6.4237(7)\) Å, \(b = 5.0176(1)\) Å, \(c = 8.1129(1)\) Å, \(\beta = 108.064(1)^\circ\) and \(V=248.60(1)\) Å\(^3\) (where the number in parenthesis is 1\(\sigma\) uncertainties on the last digit). At 1.72(5) GPa, a structural phase transition is observed to a new monoclinic structure, Sr-calcite-IIIc (Sr-CC-IIIc), with space group \(P2_1/m\) and \(Z=8\) (\(a~=~6.2683(2)\) Å, \(b = 9.9220(5)\) Å, \(c = 7.6574(6)\) Å, \(\beta = 103.856(6)^\circ\) and \(V = 462.39(5)\) Å\(^3\)), different from any pure \(\hbox {CaCO}_{3}\) polymorph. At 12 GPa, the sample transformed to a triclinic structure, Sr-calcite-IIIb (Sr-CC-IIIb), with space group \(P{\bar{1}}\) and \(Z=4\) ( \(a=6.059(5)\) Å, \(b=6.280(2)\) Å, \(c=6.331(2)\) Å, \(\alpha =95.20(3)^\circ\), \(\beta =108.89(5)^\circ\), \(\gamma =110.52(5)^\circ\) and \(V=207.7(2)\) Å\(^3\)), isostructural to end-member \(\hbox {CaCO}_{3}\)-IIIb. Finally, at 17 GPa, a transition is observed to Sr-calcite-VI (Sr-CC-VI), with space group \(P{\bar{1}}\) and \(Z=2\) (\(a=3.444(3)\) Å, \(b=4.985(4)\) Å, \(c=5.761(5)\) Å, \(\alpha =77.05(7)^\circ\), \(\beta =84.92(7)^\circ\), \(\gamma =89.00(7)^\circ\) and \(V=96.0(1)\) Å\(^3\)), isostructural to end-member \(\hbox {CaCO}_{3}\)-VI, which is preserved up to the maximum investigated pressure of 22 GPa. The results of this study show the effect of Sr/Ca cationic substitution on the high-pressure behavior and physical properties of a \(\hbox {CaCO}_{3}\)–\(\hbox {SrCO}_{3}\) solid solution. The phase evolution of \(\hbox {Ca}_{0.82}\hbox {Sr}_{0.18}\hbox {CO}_3\) and the crystallization of a new phase, Sr-CC-IIIc, different from the high-pressure polymorphs of end-member \(\hbox {CaCO}_{3}\), point to the importance of extending the study of carbonates to more complex systems than pure end-member compositions.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, L.P., upon reasonable request.
References
Angel RJ, Bujak M, Zhao J, Gatta GD, Jacobsen SD (2007) Effective hydrostatic limits of pressure media for high-pressure crystallographic studies. J Appl Crystallogr 40(1):26–32. https://doi.org/10.1107/S0021889806045523
Angel RJ, Alvaro M, Gonzalez-Platas J (2014) Eosfit7c and a fortran module (library) for equation of state calculations. Z Kristallogr Cryst Mater 229(5):405–419. https://doi.org/10.1515/zkri-2013-1711
Antao SM, Hassan I, Mulder WH, Lee PL, Toby BH (2009) In situ study of the R-3c to R-3m orientational disorder in calcite. Phys Chem Miner 36(3):159–169. https://doi.org/10.1007/s00269-008-0266-y
Bayarjargal L, Fruhner C-J, Schrodt N, Winkler B (2018) \(\text{ CaCO}_{3}\) phase diagram studied with Raman spectroscopy at pressures up to 50 GPa and high temperatures and DFT modeling. Phys Earth Planet Inter 281:31–45. https://doi.org/10.1016/j.pepi.2018.05.002
Belkofsi R, Chahi G, Adjaoud O, Belabbas I (2021) On the impact of zero-point vibrations in calcium carbonate. Phase Transit 94(11):779–793. https://doi.org/10.1080/01411594.2021.1979973
Biedermann N, Winkler B, Speziale S, Reichmann H-J, Koch-Müller M (2017) Single-crystal elasticity of \(\text{ SrCO}_{3}\) by Brillouin spectroscopy. High Press Res 37(2):181–192. https://doi.org/10.1080/08957959.2017.1289193
Biedermann N, Bykova E, Morgenroth W, Efthimiopoulos I, Mueller J, Spiekermann G, Glazyrin K, Pakhomova A, Appel K, Wilke M (2020) Equation of state and high-pressure phase behaviour of \(\text{ SrCO}_{3}\). Eur J Mineral 32(6):575–586. https://doi.org/10.5194/ejm-32-575-2020
Boehler R (2006) New diamond cell for single-crystal X-ray diffraction. Rev Sci Instrum 77(11):115103. https://doi.org/10.1063/1.2372734
Brenker FE, Vollmer C, Vincze L, Vekemans B, Szymanski A, Janssens K, Szaloki I, Nasdala L, Joswig W, Kaminsky F (2007) Carbonates from the lower part of transition zone or even the lower mantle. Earth Planet Sci Lett 260(1–2):1–9. https://doi.org/10.1016/j.epsl.2007.02.038
Burton BP, Van de Walle A (2003) First-principles-based calculations of the \(\text{ CaCO}_{3}\)-\(\text{ MgCO}_{3}\) and \(\text{ CdCO}_{3}\)-\(\text{ MgCO}_{3}\) subsolidus phase diagrams. Phys Chem Miner 30(2):88–97. https://doi.org/10.1007/s00269-002-0294-y
Carlson WD (1980) The calcite-aragonite equilibrium: effects of SR substitution and anion orientational disorder. Am Mineral 65(11–12):1252–1262
Casey WH, Chal L, Navrotsky A, Rock PA (1996) Thermochemistry of mixing strontianite [\(\text{ SrCO}_{3}\)(s)] and aragonite [\(\text{ CaCO}_{3}\)(s)] to form Ca\(_x\) Sr\(_{1- x}\text{ CO}_{3}\) (s) solid solutions. Geochim Cosmochim Acta 60(6):933–940. https://doi.org/10.1016/0016-7037(96)00441-3
Chang LL (1965) Subsolidus phase relations in the systems \(\text{ BaCO}_{3}\)-\(\text{ SrCO}_{3}\), \(\text{ SrCO}_{3}\)-\(\text{ CaCO}_{3}\), and \(\text{ BaCO}_{3}\)-\(\text{ CaCO}_{3}\). J Geol 73(2):346–368. https://doi.org/10.1086/627065
Chang LL (1971) Subsolidus Phase Relations in the Aragonite- Type Carbonates: I. The System \(\text{ CaCO}_{3}\)-\(\text{ SrCO}_{3}\)-\(\text{ BaCO}_{3}\). Am Mineral J Earth Planet Mater 56(9–10):1660–1673
Collins NC, Bebout GE, Angiboust S, Agard P, Scambelluri M, Crispini L, John T (2015) Subduction zone metamorphic pathway for deep carbon cycling: II. Evidence from HP/UHP metabasaltic rocks and ophicarbonates. Chem Geol 412:132–150. https://doi.org/10.1016/j.chemgeo.2015.06.012
CrysAlis PRO Software system (2018) Rigaku Oxford diffraction. Yarnton, UK
Dasgupta R, Hirschmann MM (2006) Melting in the Earth’s deep upper mantle caused by carbon dioxide. Nature 440(7084):659–662. https://doi.org/10.1038/nature04612
Dasgupta R, Hirschmann MM (2010) The deep carbon cycle and melting in Earth’s interior. Earth Planet Sci Lett 298(1–2):1–13. https://doi.org/10.1016/j.epsl.2010.06.039
Dewaele A, Rosa AD, Guignot N, Andrault D, Rodrigues JEF, Garbarino G (2021) Stability and equation of state of face-centered cubic and hexagonal close packed phases of argon under pressure. Sci Rep 11(1):15192. https://doi.org/10.1038/s41598-021-93995-y
Druzhbin D, Rashchenko SV, Shatskiy A, Crichton WA (2022) New high-pressure and high-temperature \(\text{ CaCO}_{3}\) polymorph. ACS Earth Space Chem 6(6):1506–1513. https://doi.org/10.1021/acsearthspacechem.2c00019
Dudnikova V, Urusov V, Eremin N (2015) Simulation of the local structure, mixing properties, and stability of \(\text{ Ca}_x\text{ Sr}_{1- x}\text{ CO}_{3}\) solid solutions by the interatomic potential method. Phys Solid State 57(6):1108–1113. https://doi.org/10.1134/S1063783415060104
Faiziev A, Iskandarov FS, Gafurov F (1998) Mineralogical and petrogenetic characteristics of carbonatites of the Dunkeldykskii alkalic massif (eastern Pamirs). Zapiski Vseross Mineral Obshchestva 127(3):54–57
Gavryushkin PN, Sagatov N, Belonoshko AB, Banaev MV, Litasov KD (2020) Disordered aragonite: the new high-pressure, high-temperature phase of \(\text{ CaCO}_{3}\). J Phys Chem C 124(48):26467–26473. https://doi.org/10.1021/acs.jpcc.0c08309
Gonzalez-Platas J, Alvaro M, Nestola F, Angel R (2016) Eos Fit7-GUI: a new graphical user interface for equation of state calculations, analyses and teaching. J Appl Crystallogr 49(4):1377–1382. https://doi.org/10.1107/S1600576716008050
Hatch DM, Merrill L (1981) Landau description of the calcite-\(\text{ CaCO}_{3}\) (II) phase transition. Phys Rev B 23(1):368. https://doi.org/10.1103/PhysRevB.23.368
Hirschmann MM (2018) Comparative deep Earth volatile cycles: the case for C recycling from exosphere/mantle fractionation of major (\(\text{ H}_2\text{ O }\), C, N) volatiles and from \(\text{ H}_2\text{ O }\)/Ce, \(\text{ CO}_2\)/Ba, and \(\text{ CO}_2\)/Nb exosphere ratios. Earth Planet Sci Lett 502:262–273. https://doi.org/10.1016/j.epsl.2018.08.023
Hou M, Zhang Q, Tao R, Liu H, Kono Y, Mao H-K, Yang W, Chen B, Fei Y (2019) Temperature-induced amorphization in \(\text{ CaCO}_{3}\) at high pressure and implications for recycled \(\text{ CaCO}_{3}\) in subduction zones. Nat Commun 10(1):1–8. https://doi.org/10.1038/s41467-019-09742-5
Ishizawa N (2014) Calcite V: a hundred-year-old mystery has been solved. Powder Diffr 29(S1):19–23. https://doi.org/10.1017/S0885715614000888
Ishizawa N, Setoguchi H, Yanagisawa K (2013) Structural evolution of calcite at high temperatures: phase V unveiled. Sci Rep 3:2832. https://doi.org/10.1038/srep02832
Kaminsky F (2012) Mineralogy of the lower mantle: a review of ‘super-deep’ mineral inclusions in diamond. Earth Sci Rev 110(1–4):127–147. https://doi.org/10.1016/j.earscirev.2011.10.005
Kaminsky F, Wirth R, Matsyuk S, Schreiber A, Thomas R (2009) Nyerereite and nahcolite inclusions in diamond: evidence for lower-mantle carbonatitic magmas. Mineral Mag 73(5):797–816. https://doi.org/10.1180/minmag.2009.073.5.797
Kelemen PB, Manning CE (2015) Reevaluating carbon fluxes in subduction zones, what goes down, mostly comes up. Proc Natl Acad Sci 112(30):3997–4006. https://doi.org/10.1073/pnas.1507889112
Kiseeva ES, Yaxley GM, Hermann J, Litasov KD, Rosenthal A, Kamenetsky VS (2012) An experimental study of carbonated eclogite at 3.5–5.5 GPa-implications for silicate and carbonate metasomatism in the cratonic mantle. J Petrol 53(4):727–759. https://doi.org/10.1093/petrology/egr078
Klotz S, Chervin J, Munsch P, Le Marchand G (2009) Hydrostatic limits of 11 pressure transmitting media. J Phys D Appl Phys 42(7):075413. https://doi.org/10.1088/0022-3727/42/7/075413
Koch-Müller M, Jahn S, Birkholz N, Ritter E, Schade U (2016) Phase transitions in the system \(\text{ CaCO}_{3}\) at high P and T determined by in situ vibrational spectroscopy in diamond anvil cells and first-principles simulations. Phys Chem Miner 43(8):545–561. https://doi.org/10.1007/s00269-016-0815-8
Lee C-T, Rudnick RL, McDonough WF, Horn I (2000) Petrologic and geochemical investigation of carbonates in peridotite xenoliths from northeastern Tanzania. Contrib Mineral Petrol 139(4):470–484. https://doi.org/10.1007/s004100000144
Liermann H-P, Konôpková Z, Morgenroth W, Glazyrin K, Bednarčik J, McBride E, Petitgirard S, Delitz J, Wendt M, Bican Y et al (2015) The extreme conditions beamline P02.2 and the extreme conditions science infrastructure at PETRA III. J Synchrotron Radiat 22(4):908–924. https://doi.org/10.1107/S1600577515005937
Litasov KD, Shatskiy A, Gavryushkin PN, Bekhtenova AE, Dorogokupets PI, Danilov BS, Higo Y, Akilbekov AT, Inerbaev TM (2017) PVT equation of state of \(\text{ CaCO}_{3}\) aragonite to 29 GPa and 1673 K: in situ X-ray diffraction study. Phys Earth Planet Inter 265:82–91. https://doi.org/10.1016/j.pepi.2017.02.006
Littlewood JL, Shaw S, Peacock CL, Bots P, Trivedi D, Burke IT (2017) Mechanism of enhanced strontium uptake into calcite via an amorphous calcium carbonate crystallization pathway. Cryst Growth Des 17(3):1214–1223. https://doi.org/10.1021/acs.cgd.6b01599
Logvinova AM, Wirth R, Fedorova EN, Sobolev NV (2008) Nanometre-sized mineral and fluid inclusions in cloudy Siberian diamonds: new insights on diamond formation. Eur J Mineral 20(3):317–331. https://doi.org/10.1127/0935-1221/2008/0020-1815
Logvinova A, Wirth R, Tomilenko A, Afanas’Ev V, Sobolev N (2011) The phase composition of crystal-fluid nanoinclusions in alluvial diamonds in the northeastern siberian platform. Russ Geol Geophys 52(11):1286–1297. https://doi.org/10.1016/j.rgg.2011.10.002
Logvinova AM, Shatskiy A, Wirth R, Tomilenko AA, Ugap’eva SS, Sobolev NV (2019) Carbonatite melt in type Ia gem diamond. Lithos 342:463–467. https://doi.org/10.1016/j.lithos.2019.06.010
Lucas-Girot A, Hernandez O, Oudadesse H (2007) Re-examination of the structural properties of solid solutions Sr\(_x\) Ca\(_{1- x}\text{ CO}_{3}\). Mater Res Bull 42(6):1061–1068. https://doi.org/10.1016/j.materresbull.2006.09.016
Marquardt H, Speziale S, Gleason A, Sinogeikin S, Kantor I, Prakapenka V (2013) Brillouin scattering and X-ray diffraction of solid argon to 65 GPa and 700 K: shear strength of argon at HP/HT. J Appl Phys 10(1063/1):4820578
Martirosyan NS, Efthimiopoulos I, Pennacchioni L, Wirth R, Jahn S, Koch-Müller M (2021) Effect of cationic substitution on the pressure-induced phase transitions in calcium carbonate. Am Mineral J Earth Planet Mater 106(4):549–558. https://doi.org/10.2138/am-2021-7547
Matsunuma S, Kagi H, Komatsu K, Maruyama K, Yoshino T (2014) Doping incompatible elements into calcite through amorphous calcium carbonate. Cryst Growth Des 14(11):5344–5348. https://doi.org/10.1021/cg500953h
Menadakis M, Maroulis G, Koutsoukos P (2007) A quantum chemical study of doped \(\text{ CaCO}_{3}\) (calcite). Comput Mater Sci 38(3):522–525. https://doi.org/10.1016/j.commatsci.2006.02.016
Merlini M, Hanfland M, Crichton W (2012) \(\text{ CaCO}_3\)-III and \(\text{ CaCO}_{3}\)-VI, high-pressure polymorphs of calcite: possible host structures for carbon in the Earth’s mantle. Earth Planet Sci Lett 333:265–271. https://doi.org/10.1016/j.epsl.2012.04.036
Merlini M, Crichton W, Chantel J, Guignard J, Poli S (2014) Evidence of interspersed co-existing \(\text{ CaCO}_3\)-III and \(\text{ CaCO}_{3}\)-IIIb structures in polycrystalline \(\text{ CaCO}_{3}\) at high pressure. Mineral Mag 78(2):225–233. https://doi.org/10.1180/minmag.2014.078.2.01
Merrill L, Bassett WA (1975) The crystal structure of \(\text{ CaCO}_3\) (II), a high-pressure metastable phase of calcium carbonate. Acta Crystallogr Sect B Struct Crystallogr Cryst Chem 31(2):343–349. https://doi.org/10.1107/S0567740875002774
Mirwald PW (1976) A differential thermal analysis study of the high-temperature polymorphism of calcite at high pressure. Contrib Mineral Petrol 59(1):33–40
Momma K, Izumi F (2011) VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J Appl Crystallogr 44(6):1272–1276. https://doi.org/10.1107/S0021889811038970
Müller WF, Franz G (2008) TEM-microstructures in omphacite and other minerals from eclogite near to a thrust zone; the Eclogite Zone Venediger nappe area, Tauern Window, Austria. Neues Jahrb Mineral Abh. https://doi.org/10.1127/0077-7757/2008/0099
Ono S, Kikegawa T, Ohishi Y, Tsuchiya J (2005) Post-aragonite phase transformation in \(\text{ CaCO}_3\) at 40 GPa. Am Mineral 90(4):667–671. https://doi.org/10.2138/am.2005.1610
Pal’Yanov YN, Sokol A, Borzdov YM, Khokhryakov A, Sobolev N (1999) Diamond formation from mantle carbonate fluids. Nature 400(6743):417–418. https://doi.org/10.1038/22678
Pennacchioni L, Speziale S, Winkler B (2023) Elasticity of natural aragonite samples by Brillouin spectroscopy. Phys Chem Miner 50(3):22. https://doi.org/10.1007/s00269-023-01244-7
Petříček V, Dušek M, Palatinus L (2014) Crystallographic computing system JANA2006: general features. Z Kristallogr Cryst Mater 229(5):345–352. https://doi.org/10.1515/zkri-2014-1737
Pippinger T, Miletich R, Merlini M, Lotti P, Schouwink P, Yagi T, Crichton W, Hanfland M (2015) Puzzling calcite-III dimorphism: crystallography, high-pressure behavior, and pathway of single-crystal transitions. Phys Chem Miner 42(1):29–43. https://doi.org/10.1007/s00269-014-0696-7
Plank T, Manning CE (2019) Subducting carbon. Nature 574(7778):343–352. https://doi.org/10.1038/s41586-019-1643-z
Redfern SA, Angel RJ (1999) High-pressure behaviour and equation of state of calcite, \(\text{ CaCO}_{3}\). Contrib Mineral Petrol 134(1):102–106
Redfern S, Salje E, Navrotsky A (1989) High-temperature enthalpy at the orientational order-disorder transition in calcite: implications for the calcite/aragonite phase equilibrium. Contrib Mineral Petrol 101(4):479–484. https://doi.org/10.1007/BF00372220
Reeder R, Nakajima Y (1982) The nature of ordering and ordering defects in dolomite. Phys Chem Miner 8(1):29–35. https://doi.org/10.1007/BF00311160
Reeder R, Wenk H-R (1983) Structure refinements of some thermally disordered dolomites. Am Mineral 68(7–8):769–776
Reguir E (2001) Aspects of the mineralogy of the Murun alkaline complex, Yakutia, Russia. Ph.D. thesis
Saito A, Kagi H, Marugata S, Komatsu K, Enomoto D, Maruyama K, Kawano J (2020) Incorporation of incompatible Strontium and Barium ions into calcite (\(\text{ CaCO}_{3}\)) through amorphous calcium carbonate. Minerals 10(3):270. https://doi.org/10.3390/min10030270
Sheldrick GM (2015) Crystal structure refinement with SHELXL. Acta Crystallogr Sect C 71(1):3–8. https://doi.org/10.1107/S2053229614024218
Shen G, Wang Y, Dewaele A, Wu C, Fratanduono DE, Eggert J, Klotz S, Dziubek KF, Loubeyre P, Fat’yanov OV et al (2020) Toward an international practical pressure scale: a proposal for an IPPS ruby gauge (IPPS-Ruby2020). High Press Res 40(3):299–314. https://doi.org/10.1080/08957959.2020.1791107
Shimizu H, Tashiro H, Kume T, Sasaki S (2001) High-pressure elastic properties of solid argon to 70 GPa. Phys Rev Lett 86(20):4568. https://doi.org/10.1103/PhysRevLett.86.4568
Sleep NH (2009) Stagnant lid convection and carbonate metasomatism of the deep continental lithosphere. Geochem Geophys Geosyst. https://doi.org/10.1029/2009GC002702
Spahr D, König J, Bayarjargal L, Milman V, Persson MP, Winkler B (2021) Structural, physical, and thermodynamic properties of aragonitic \(\text{ Ca}_x\text{ Sr}_{1-x}\text{ CO}_{3}\) solid solutions. J Phys Chem C 125(31):17474–17481. https://doi.org/10.1021/acs.jpcc.1c04703
Stachel T, Harris JW (2009) Formation of diamond in the Earth’s mantle. J Phys Condens Matter 21(36):364206. https://doi.org/10.1088/0953-8984/21/36/364206
Stachel T, Harris JW, Brey GP, Joswig W (2000) Kankan diamonds (Guinea) II: lower mantle inclusion parageneses. Contrib Mineral Petrol 140(1):16–27. https://doi.org/10.1007/s004100000174
Suito K, Namba J, Horikawa T, Taniguchi Y, Sakurai N, Kobayashi M, Onodera A, Shimomura O, Kikegawa T (2001) Phase relations of \(\text{ CaCO}_3\) at high pressure and high temperature. Am Mineral 86(9):997–1002. https://doi.org/10.2138/am-2001-8-906
Ter Heege J, Renner J (2007) In situ impedance spectroscopy on pyrophyllite and \(\text{ CaCO}_{3}\) at high pressure and temperature: phase transformations and kinetics of atomistic transport. Phys Chem Miner 34(7):445–465. https://doi.org/10.1007/s00269-007-0162-x
Vinograd VL, Sluiter MH, Winkler B (2009) Subsolidus phase relations in the \(\text{ CaCO}_{3}\)-\(\text{ MgCO}_{3}\) system predicted from the excess enthalpies of supercell structures with single and double defects. Phys Rev B 79(10):104201. https://doi.org/10.1103/PhysRevB.79.104201
Wall F, Le Bas M, Srivastava R (1993) Calcite and carbocernaite exsolution and cotectic textures in a Sr, REE-rich carbonatite dyke from Rajasthan, India. Mineral Mag 57(388):495–513. https://doi.org/10.1180/minmag.1993.057.388.11
Wang A, Pasteris JD, Meyer HO, Dele-Duboi ML (1996) Magnesite-bearing inclusion assemblage in natural diamond. Earth Planet Sci Lett 141(1–4):293–306. https://doi.org/10.1016/0012-821X(96)00053-2
Wirth R (2009) Focused ion beam (FIB) combined with SEM and TEM: advanced analytical tools for studies of chemical composition, microstructure and crystal structure in geomaterials on a nanometre scale. Chem Geol 261(3–4):217–229. https://doi.org/10.1016/j.chemgeo.2008.05.019
Wondratschek H, Jeitschko W (1976) Twin domains and antiphase domains. Acta Crystallogr Sect A Cryst Phys Diffr Theor Gen Crystallogr 32(4):664–666. https://doi.org/10.1107/S056773947600137X
Yaxley GM, Crawford AJ, Green DH (1991) Evidence for carbonatite metasomatism in spinel peridotite xenoliths from western Victoria, Australia. Earth Planet Sci Lett 107(2):305–317. https://doi.org/10.1016/0012-821X(91)90078-V
Zhao C-S, Li H-P, Chen P-F, Jiang J-J (2019) Sound velocities across calcite phase transitions by Brillouin scattering spectroscopy. Am Mineral J Earth Planet Mater 104(3):418–424. https://doi.org/10.2138/am-2019-6682
Acknowledgements
The research presented in this study has been carried out within the scope of the DFG-funded German research unit DFG FOR 2125 ‘CarboPaT’ (WI1231, SP1216/7-1, BA4020, EF112/1-2, JA1469/11-2, KO1260/16-2). We would like to thank Nico Giordano for assistance in using beamline P02.2 (Petra-III/DESY), Anja Schreiber at GFZ for technical assistance on the FIB and Lkhamsuren Bayarjargal at Goethe University, Frankfurt Am Main, for technical assistance. We are indebted to Bjoern Winkler at Goethe University Frankfurt Am Main, Fabrizio Nestola and Martha Pamato at Universitá degli Studi di Padova for the fruitful discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pennacchioni, L., Martirosyan, N.S., Pakhomova, A. et al. Crystal structure and high-pressure phase behavior of a CaCO3–SrCO3 solid solution. Phys Chem Minerals 50, 29 (2023). https://doi.org/10.1007/s00269-023-01252-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00269-023-01252-7