Abstract
Background
Lip augmentation could be a possible cause of blindness following filler injections. This study evaluated the risk by simulating clinical scenarios of marginal injections to the upper and lower lips and then evaluated the risk of vascular injuries.
Methods
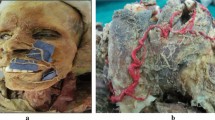
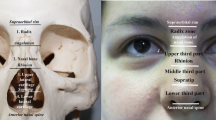
A 22G cannula was inserted bilaterally along the wet–dry junction of the upper and lower lip margins in fifteen cadavers, and then both lips were dissected to verify possible injuries to the superior and inferior labial arteries. The position of the labial arteries in the vermilion zone was documented to determine the appropriate injection technique.
Results
In the marginal injections to the lips, arterial injuries occurred at the medial segment of the vermilion zone of both the upper and lower lips, at the terminal part of the labial arteries or a distal branch. Considering arterial anatomy, the upper lip has a higher chance of arterial injury than the lower lip. The cannula should not be inserted in the submucosa as it is recommended to evert the vermilion because both the superior and inferior labial arteries are located in the submucosa of the medial and middle segments of the vermilion in all specimens.
Conclusion
Awareness of the possibility of vascular injury is necessary during injections of the medial segments of the vermilion of the lips. Vermilion border and marginal injections are recommended for safe and effective lip augmentation. Deep injection around the oral commissure and submucosal injection of the medial and middle segments of the vermilion zone are prohibited because of the high risk of arterial injury.
No Level Assigned
This journal requires that authors assign a level of evidence to each submission to which Evidence-Based Medicine rankings are applicable. This excludes Review Articles, Book Reviews, and manuscripts that concern Basic Science, Animal Studies, Cadaver Studies, and Experimental Studies. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266













Similar content being viewed by others
References
Klein AW (2006) Soft tissue augmentation 2006: filler fantasy. Dermatol Ther 19:129–133
Cartier H, Trevidic P, Rzany B, Sattler G, Kestemont P, Kerronuche N, Dhuin JC (2012) Perioral rejuvenation with a range of customized hyaluronic acid fillers: efficacy and safety over six months with a specific focus on the lips. J Drugs Dermatol 11:s17–s26
Grippaudo FR, Di Girolamo M, Mattei M, Pucci E, Grippaudo C (2014) Diagnosis and management of dermal filler complications in the perioral region. J Cosmet Laser Ther 16:246–252
San Miguel Moragas J, Reddy RR, Hernández Alfaro F, Mommaerts MY (1015) Systematic review of “filling” procedures for lip augmentation regarding types of material, outcomes and complications. J Craniomaxillofac Surg 43:883–906
Fischer TC, Sattler G, Gauglitz GG (2016) Hyaluron filler containing lidocaine on a CPM basis for lip augmentation: reports from practical experience. Facial Plast Surg 32:283–288
Fulton J, Caperton C, Weinkle S, Dewandre L (2012) Filler injections with the blunt-tip microcannula. J Drugs Dermatol 11:1098–1103
Philipp-Dormston WG, Hilton S, Nathan M (2014) A prospective, open-label, multicenter, observational, postmarket study of the use of a 15 mg/mL hyaluronic acid dermal filler in the lips. J Cosmet Dermatol 13:125–134
Smith SR, Lin X, Shamban A (2013) Small gel particle hyaluronic acid injection technique for lip augmentation. J Drugs Dermatol 12:764–769
Lanigan S (2011) An observational study of a 24 mg/mL hyaluronic acid with pre-incorporated lidocaine for lip definition and enhancement. J Cosmet Dermatol 10:11–14
Samuelson U, Fagrell D, Wetter A, Kuusk S, Hamilton L, Haglund P (2015) An open-label, multicenter, evaluator-blinded study to assess the efficacy and safety of a new hyaluronic acid-based gel product for lip enhancement. Dermatol Surg 41:1052–1059
Smith SR, Vander Ploeg HM, Sanstead M, Albright CD, Theisen MJ, Lin X (2015) Functional safety assessments used in a randomized controlled study of small gel particle hyaluronic acid for lip augmentation. Dermatol Surg 41:S137–S142
Dayan S, Bruce S, Kilmer S, Dover JS, Downie JB, Taylor SC, Skorupa A, Murphy DK (2015) Safety and effectiveness of the hyaluronic acid filler, HYC-24L, for lip and perioral augmentation. Dermatol Surg 41:S293–S301
Glogau RG, Bank D, Brandt F, Cox SE, Donofrio L, Dover J, Grekin S, Lawrence I, Lin X, Nestor M, Shamban A, Stewart D, Weiss R, Axford-Gatley RA, Theisen MJ, Smith S (2012) A randomized, evaluator-blinded, controlled study of the effectiveness and safety of small gel particle hyaluronic acid for lip augmentation. Dermatol Surg 38:1180–1192
Tansatit T, Apinuntrum P, Phetudom T (2014) A typical pattern of the labial arteries with implication for lip augmentation with injectable fillers. Aesthet Plast Surg 38:1083–1089
Lee SH, Gil YC, Choi YJ, Tansatit T, Kim HJ, Hu KS (2015) Topographic anatomy of the superior labial artery for dermal filler injection. Plast Reconstr Surg 135:445–450
Brandt F, Bassichis B, Bassichis M, O’Connell C, Lin X (2011) Safety and effectiveness of small and large gel-particle hyaluronic acid in the correction of perioral wrinkles. J Drugs Dermatol 10:982–987
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Tansatit, T., Apinuntrum, P. & Phetudom, T. Cadaveric Assessment of Lip Injections: Locating the Serious Threats. Aesth Plast Surg 41, 430–440 (2017). https://doi.org/10.1007/s00266-016-0755-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-016-0755-1