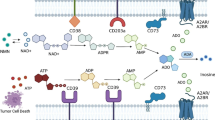
Abstract
The CD39-CD73–adenosinergic pathway converts adenosine triphosphate (ATP) to adenosine for inhibiting anti-tumor immune responses. Therefore, targeting CD73 to reinvigorate anti-tumor immunity is considered the novel cancer immunotherapy to eradicate tumor cells. To fully understand the critical role of CD39/CD73 in colon adenocarcinoma (COAD), this study aims to comprehensive investigate the prognostic significance of CD39 and CD73 in stage I–IV COAD. Our data demonstrated that CD73 staining strongly marked malignant epithelial cells and CD39 was highly expressed in stromal cells. Attractively, tumor CD73 expression was significantly associated with tumor stage and the risk of distant metastasis, which suggested CD73 was as an independent factor for colon adenocarcinoma patients in univariate COX analysis [HR = 1.465, 95%CI = 1.084–1.978, p = 0.013]; however, high stromal CD39 in COAD patients was more likely to have favorable survival outcome [HR = 1.458, p = 1.103–1.927, p = 0.008]. Notably, high CD73 expression in COAD patients showed poor response to adjuvant chemotherapy and high risk of distant metastasis. High CD73 expression was inversely associated with less infiltration of CD45+ and CD8+ immune cells. However, administration with anti-CD73 antibodies significantly increased the response to oxaliplatin (OXP). Blockade of CD73 signaling synergistically enhanced OXP-induced ATP release, which is a marker of immunogenic cell death (ICD), promotes dendritic cell maturation and immune cell infiltration. Moreover, the risk of colorectal cancer lung metastasis was also decreased. Taken together, the present study revealed tumor CD73 expression inhibited the recruitment of immune cells and correlated with a poor prognosis in COAD patients, especially patients received adjuvant chemotherapy. Targeting CD73 to markedly increased the therapeutic response to chemotherapy and inhibited lung metastasis. Therefore, tumor CD73 may be an independent prognostic factor as well as the potential of therapeutic target for immunotherapy to benefit colon adenocarcinoma patients.





Similar content being viewed by others
References
Huang CY, Chiang SF, Ke TW, Chen TW, You YS, Chen WT et al (2018) Clinical significance of programmed death 1 ligand-1 (CD274/PD-L1) and intra-tumoral CD8+ T-cell infiltration in stage II-III colorectal cancer. Sci Rep 8(1):15658. https://doi.org/10.1038/s41598-018-33927-5
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72(1):7–33. https://doi.org/10.3322/caac.21708
Adlard JW, Richman SD, Seymour MT, Quirke P (2002) Prediction of the response of colorectal cancer to systemic therapy. Lancet Oncol 3(2):75–82. https://doi.org/10.1016/s1470-2045(02)00648-4
McCleary NJ, Meyerhardt JA, Green E, Yothers G, de Gramont A, Van Cutsem E et al (2013) Impact of age on the efficacy of newer adjuvant therapies in patients with stage II/III colon cancer: findings from the ACCENT database. J Clin Oncol 31(20):2600–2606. https://doi.org/10.1200/JCO.2013.49.6638
Huang CY, Chiang SF, Ke TW, Chen TW, Lan YC, You YS et al (2018) Cytosolic high-mobility group box protein 1 (HMGB1) and/or PD-1+ TILs in the tumor microenvironment may be contributing prognostic biomarkers for patients with locally advanced rectal cancer who have undergone neoadjuvant chemoradiotherapy. Cancer Immunol Immunother 67(4):551–562. https://doi.org/10.1007/s00262-017-2109-5
Chen TW, Huang KC, Chiang SF, Chen WT, Ke TW, Chao KSC (2019) Prognostic relevance of programmed cell death-ligand 1 expression and CD8+ TILs in rectal cancer patients before and after neoadjuvant chemoradiotherapy. J Cancer Res Clin Oncol 145(4):1043–1053. https://doi.org/10.1007/s00432-019-02874-7
Wang J, Yuan R, Song W, Sun J, Liu D, Li Z (2017) PD-1, PD-L1 (B7–H1) and Tumor-site immune modulation therapy: the historical perspective. J Hematol Oncol 10(1):34. https://doi.org/10.1186/s13045-017-0403-5
Xu-Monette ZY, Zhang M, Li J, Young KH (2017) PD-1/PD-L1 blockade: have we found the key to unleash the antitumor immune response? Front Immunol 8:1597. https://doi.org/10.3389/fimmu.2017.01597
Beavis PA, Divisekera U, Paget C, Chow MT, John LB, Devaud C et al (2013) Blockade of A2A receptors potently suppresses the metastasis of CD73+ tumors. Proc Natl Acad Sci U S A 110(36):14711–14716. https://doi.org/10.1073/pnas.1308209110
Leclerc BG, Charlebois R, Chouinard G, Allard B, Pommey S, Saad F et al (2016) CD73 expression is an independent prognostic factor in prostate cancer. Clin Cancer Res 22(1):158–166. https://doi.org/10.1158/1078-0432.CCR-15-1181
Hatfield SM, Kjaergaard J, Lukashev D, Schreiber TH, Belikoff B, Abbott R et al (2015) Immunological mechanisms of the antitumor effects of supplemental oxygenation. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aaa1260
Antonioli L, Pacher P, Vizi ES, Hasko G (2013) CD39 and CD73 in immunity and inflammation. Trends Mol Med 19(6):355–367. https://doi.org/10.1016/j.molmed.2013.03.005
Synnestvedt K, Furuta GT, Comerford KM, Louis N, Karhausen J, Eltzschig HK et al (2002) Ecto-5’-nucleotidase (CD73) regulation by hypoxia-inducible factor-1 mediates permeability changes in intestinal epithelia. J Clin Invest 110(7):993–1002. https://doi.org/10.1172/JCI15337
Niemela J, Henttinen T, Yegutkin GG, Airas L, Kujari AM, Rajala P et al (2004) IFN-alpha induced adenosine production on the endothelium: a mechanism mediated by CD73 (ecto-5’-nucleotidase) up-regulation. J Immunol 172(3):1646–1653. https://doi.org/10.4049/jimmunol.172.3.1646
Burnstock G, Di Virgilio F (2013) Purinergic signalling and cancer. Purinergic Signal 9(4):491–540. https://doi.org/10.1007/s11302-013-9372-5
Loi S, Pommey S, Haibe-Kains B, Beavis PA, Darcy PK, Smyth MJ et al (2013) CD73 promotes anthracycline resistance and poor prognosis in triple negative breast cancer. Proc Natl Acad Sci U S A 110(27):11091–11096. https://doi.org/10.1073/pnas.1222251110
Zhang B, Song B, Wang X, Chang XS, Pang T, Zhang X et al (2015) The expression and clinical significance of CD73 molecule in human rectal adenocarcinoma. Tumour Biol 36(7):5459–5466. https://doi.org/10.1007/s13277-015-3212-x
Yegutkin GG, Marttila-Ichihara F, Karikoski M, Niemela J, Laurila JP, Elima K et al (2011) Altered purinergic signaling in CD73-deficient mice inhibits tumor progression. Eur J Immunol 41(5):1231–1241. https://doi.org/10.1002/eji.201041292
Perrot I, Michaud HA, Giraudon-Paoli M, Augier S, Docquier A, Gros L et al (2019) Blocking antibodies targeting the CD39/CD73 immunosuppressive pathway unleash immune responses in combination cancer therapies. Cell Rep 27(8):2411–2425. https://doi.org/10.1016/j.celrep.2019.04.091
Huang KC, Chiang SF, Chen TW, Chen WT, Yang PC, Ke TW et al (2020) Prognostic relevance of programmed cell death 1 ligand 2 (PDCD1LG2/PD-L2) in patients with advanced stage colon carcinoma treated with chemotherapy. Sci Rep 10(1):22330. https://doi.org/10.1038/s41598-020-79419-3
Lin TY, Fan CW, Maa MC, Leu TH (2015) Lipopolysaccharide-promoted proliferation of Caco-2 cells is mediated by c-Src induction and ERK activation. Biomedicine (Taipei) 5(1):5. https://doi.org/10.7603/s40681-015-0005-x
Wang X, Sheu JJ, Lai MT, Yin-Yi Chang C, Sheng X, Wei L et al (2018) RSF-1 overexpression determines cancer progression and drug resistance in cervical cancer. Biomedicine (Taipei) 8(1):4. https://doi.org/10.1051/bmdcn/2018080104
Uhlen M, Fagerberg L, Hallstrom BM, Lindskog C, Oksvold P, Mardinoglu A et al (2015) Proteomics tissue-based map of the human proteome. Science 347(6220):1260419. https://doi.org/10.1126/science.1260419
Uhlen M, Zhang C, Lee S, Sjostedt E, Fagerberg L, Bidkhori G et al (2017) A pathology atlas of the human cancer transcriptome. Science. https://doi.org/10.1126/science.aan2507
Hoadley KA, Yau C, Hinoue T, Wolf DM, Lazar AJ, Drill E et al (2018) Cell-of-origin patterns dominate the molecular classification of 10,000 tumors from 33 types of cancer. Cell 173(2):291–304. https://doi.org/10.1016/j.cell.2018.03.022
Huang KC, Lai CY, Hong WZ, Chang HY, Lin PC, Chiang SF et al (2022) A novel engineered AAV-based neoantigen vaccine in combination with radiotherapy eradicates tumors. Cancer Immunol Res. https://doi.org/10.1158/2326-6066.CIR-22-0318
Huang KC, Chiang SF, Chang HY, Chen WT, Yang PC, Chen TW et al (2022) Engineered sTRAIL-armed MSCs overcome STING deficiency to enhance the therapeutic efficacy of radiotherapy for immune checkpoint blockade. Cell Death Dis 13(7):610. https://doi.org/10.1038/s41419-022-05069-0
Chen TW, Hung WZ, Chiang SF, Chen WT, Ke TW, Liang JA et al (2022) Dual inhibition of TGFbeta signaling and CSF1/CSF1R reprograms tumor-infiltrating macrophages and improves response to chemotherapy via suppressing PD-L1. Cancer Lett. https://doi.org/10.1016/j.canlet.2022.215795
Huang KC, Chiang SF, Yang PC, Ke TW, Chen TW, Hu CH et al (2021) Immunogenic cell death by the novel topoisomerase i inhibitor tlc388 enhances the therapeutic efficacy of radiotherapy. Cancers (Basel). https://doi.org/10.3390/cancers13061218
Huang KC, Chiang SF, Chen WT, Chen TW, Hu CH, Yang PC et al (2020) Decitabine augments chemotherapy-induced PD-L1 upregulation for PD-L1 blockade in colorectal cancer. Cancers (Basel). https://doi.org/10.3390/cancers12020462
Wennerberg E, Spada S, Rudqvist NP, Lhuillier C, Gruber S, Chen Q et al (2020) CD73 blockade promotes dendritic cell infiltration of irradiated tumors and tumor rejection. Cancer Immunol Res 8(4):465–478. https://doi.org/10.1158/2326-6066.CIR-19-0449
Stagg J, Smyth MJ (2010) Extracellular adenosine triphosphate and adenosine in cancer. Oncogene 29(39):5346–5358. https://doi.org/10.1038/onc.2010.292
Messaoudi N, Cousineau I, Arslanian E, Henault D, Stephen D, Vandenbroucke-Menu F et al (2020) Prognostic value of CD73 expression in resected colorectal cancer liver metastasis. Oncoimmunology 9(1):1746138. https://doi.org/10.1080/2162402X.2020.1746138
Wu XR, He XS, Chen YF, Yuan RX, Zeng Y, Lian L et al (2012) High expression of CD73 as a poor prognostic biomarker in human colorectal cancer. J Surg Oncol 106(2):130–137. https://doi.org/10.1002/jso.23056
Ren ZH, Lin CZ, Cao W, Yang R, Lu W, Liu ZQ et al (2016) CD73 is associated with poor prognosis in HNSCC. Oncotarget 7(38):61690–61702. https://doi.org/10.18632/oncotarget.11435
Samanta D, Park Y, Ni X, Li H, Zahnow CA, Gabrielson E et al (2018) Chemotherapy induces enrichment of CD47(+)/CD73(+)/PDL1(+) immune evasive triple-negative breast cancer cells. Proc Natl Acad Sci U S A 115(6):E1239–E1248. https://doi.org/10.1073/pnas.1718197115
Kroemer G, Galluzzi L, Kepp O, Zitvogel L (2013) Immunogenic cell death in cancer therapy. Annu Rev Immunol 31:51–72. https://doi.org/10.1146/annurev-immunol-032712-100008
Beavis PA, Milenkovski N, Henderson MA, John LB, Allard B, Loi S et al (2015) Adenosine receptor 2A blockade increases the efficacy of anti-PD-1 through enhanced antitumor t-cell responses. Cancer Immunol Res 3(5):506–517. https://doi.org/10.1158/2326-6066.CIR-14-0211
Bhattarai S, Freundlieb M, Pippel J, Meyer A, Abdelrahman A, Fiene A et al (2015) alpha, beta-methylene-ADP (AOPCP) derivatives and analogues: development of potent and selective ecto-5’-nucleotidase (CD73) inhibitors. J Med Chem 58(15):6248–6263. https://doi.org/10.1021/acs.jmedchem.5b00802
Stagg J, Divisekera U, McLaughlin N, Sharkey J, Pommey S, Denoyer D et al (2010) Anti-CD73 antibody therapy inhibits breast tumor growth and metastasis. Proc Natl Acad Sci U S A 107(4):1547–1552. https://doi.org/10.1073/pnas.0908801107
Allard B, Pommey S, Smyth MJ, Stagg J (2013) Targeting CD73 enhances the antitumor activity of anti-PD-1 and anti-CTLA-4 mAbs. Clin Cancer Res 19(20):5626–5635. https://doi.org/10.1158/1078-0432.CCR-13-0545
Hay CM, Sult E, Huang Q, Mulgrew K, Fuhrmann SR, McGlinchey KA et al (2016) Targeting CD73 in the tumor microenvironment with MEDI9447. Oncoimmunology 5(8):e1208875. https://doi.org/10.1080/2162402X.2016.1208875
Vogt TJ, Gevensleben H, Dietrich J, Kristiansen G, Bootz F, Landsberg J et al (2018) Detailed analysis of adenosine A2a receptor (ADORA2A) and CD73 (5’-nucleotidase, ecto, NT5E) methylation and gene expression in head and neck squamous cell carcinoma patients. Oncoimmunology 7(8):e1452579. https://doi.org/10.1080/2162402X.2018.1452579
Chen Q, Pu N, Yin H, Zhang J, Zhao G, Lou W et al (2020) CD73 acts as a prognostic biomarker and promotes progression and immune escape in pancreatic cancer. J Cell Mol Med 24(15):8674–8686. https://doi.org/10.1111/jcmm.15500
Huang KC, Chiang SC, Ke TW, Chen TW, Hu CH, Yang PC et al (2021) DNMT1 constrains IFNβ-mediated anti-tumor immunity and PD-L1 expression to reduce the efficacy of radiotherapy and immunotherapy. OncoImmunology 10(1):1989790. https://doi.org/10.1080/2162402X.2021.1989790
Wang YJ, Fletcher R, Yu J, Zhang L (2018) Immunogenic effects of chemotherapy-induced tumor cell death. Genes Dis 5(3):194–203. https://doi.org/10.1016/j.gendis.2018.05.003
Young CNJ, Gorecki DC (2018) P2RX7 purinoceptor as a therapeutic target-the second coming? Front Chem 6:248. https://doi.org/10.3389/fchem.2018.00248
Tsukui H, Horie H, Koinuma K, Ohzawa H, Sakuma Y, Hosoya Y et al (2020) CD73 blockade enhances the local and abscopal effects of radiotherapy in a murine rectal cancer model. BMC Cancer 20(1):411. https://doi.org/10.1186/s12885-020-06893-3
Zhang B, Cheng B, Li FS, Ding JH, Feng YY, Zhuo GZ et al (2015) High expression of CD39/ENTPD1 in malignant epithelial cells of human rectal adenocarcinoma. Tumour Biol 36(12):9411–9419. https://doi.org/10.1007/s13277-015-3683-9
Acknowledgements
We are grateful for the tissue microarray support from the Translation Research Core, China Medical University Hospital. This study was supported in part by China Medical University Hospital (DMR-CELL-2103, DMR-CELL-2102 and DMR-110-045, Taiwan), the Ministry of Science and Technology (MOST110-2628-B-039-005 and MOST110-2314-B-039-032, Taiwan), and the Health and welfare surcharge on tobacco products, China Medical University Hospital Cancer Research Center of Excellence (MOHW110-TDU-B-212-144024, Taiwan). This study was partially based on clinical information from the China Medical University Hospital Cancer Registry. Experiments and data analysis were performed in part through the use of the Medical Research Core Facilities Center, Office of Research & Development at China medical University, Taichung, Taiwan, R.O.C.
Author information
Authors and Affiliations
Contributions
Data curation, YL and TC; Formal analysis, SC, WTC, KCH, JL and AS; Funding acquisition, KSCC and KCH; Investigation, WTC, TK and PY; Animal experiments and flow cytometry analysis, KCH, CC and WH; Project administration, KCH; Resources, WTC, TK and KSCC; Supervision, KSCC; Writing—original draft, KCH; Writing—review & editing, KSCC.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was reviewed and approved by the Internal Review Board (IRB) of China Medical University Hospital [Protocol number: CMUH105-REC2-073]. The method was carried out in accordance with the committee’s approved guidelines.
Informed consent
Informed consents were obtained from all participants in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, YS., Chiang, SF., Chen, CY. et al. Targeting CD73 increases therapeutic response to immunogenic chemotherapy by promoting dendritic cell maturation. Cancer Immunol Immunother 72, 2283–2297 (2023). https://doi.org/10.1007/s00262-023-03416-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03416-4