Abstract
Purpose
Imaging assessment of abdominopelvic tumor burden is crucial for debulking surgery decision in ovarian cancer patients. This study aims to compare the efficiency of [68Ga]Ga-FAPI-04 FAPI PET and MRI-DWI in the preoperative evaluation and its potential impact to debulking surgery decision.
Methods
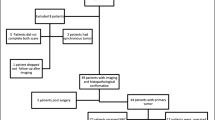
Thirty-six patients with suspected/confirmed ovarian cancer were enrolled and underwent integrated [68Ga]Ga-FAPI-04 PET/MRI. Nineteen patients (15 stage III–IV and 4 I–II stage) who underwent debulking surgery were involved in the diagnostic efficiency analysis. The images of [68Ga]Ga-FAPI-04 PET and MRI-DWI were visually analyzed respectively. Immunohistochemistry on FAP was performed in metastatic lesions to investigate the radiological missing of [68Ga]Ga-FAPI-04 PET as well as its different performance in primary debulking surgery (PDS) and interval debulking surgery (IDS) patients. Potential imaging impact on management was also studied in 35 confirmed ovarian cancer patients.
Results
[68Ga]Ga-FAPI-04 PET displayed higher sensitivity (76.8% vs.59.9%), higher accuracy (84.9% vs. 80.7%), and lower missing rate (23.2% vs. 40.1%) than MRI-DWI in detecting abdominopelvic metastasis. The diagnostic superiority of [68Ga]Ga-FAPI-04 PET is more obvious in PDS patients but diminished in IDS patients. [68Ga]Ga-FAPI-04 PET outperformed MRI-DWI in 70.8% abdominopelvic regions (17/24), which contained seven key regions that impact the resectability and surgical complexity. MRI-DWI hold advantage in the peritoneal surface of the bladder and the central tendon of the diaphragm. Of the contradictory judgments between the two modalities (14.9%), [68Ga]Ga-FAPI-04 PET correctly identified more lesions, particularly in PDS patients (73.8%). In addition, FAP expression was independent of lesion size and decreased in IDS patients. [68Ga]Ga-FAPI-04 PET changed 42% of surgical planning that was previously based on MRI-DWI.
Conclusion
[68Ga]Ga-FAPI-04 PET is more efficient in assisting debulking surgery in ovarian cancer patients than MRI-DWI. Integrated [68Ga]Ga-FAPI-04 PET/MR imaging is a potential method for planning debulking surgery in ovarian cancer patients.






Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. https://doi.org/10.3322/caac.21660.
Meyers MA. Distribution of intra-abdominal malignant seeding: dependency on dynamics of flow of ascitic fluid. Am J Roentgenol Radium Ther Nucl Med. 1973;119:198–206. https://doi.org/10.2214/ajr.119.1.198.
Armstrong DK, Alvarez RD, Bakkum-Gamez JN, Barroilhet L, Behbakht K, Berchuck A, et al. Ovarian cancer, Version 2.2020 NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2021;19:191–226. https://doi.org/10.6004/jnccn.2021.0007.
Chi DS, Eisenhauer EL, Zivanovic O, Sonoda Y, Abu-Rustum NR, Levine DA, et al. Improved progression-free and overall survival in advanced ovarian cancer as a result of a change in surgical paradigm. Gynecol Oncol. 2009;114:26–31. https://doi.org/10.1016/j.ygyno.2009.03.018.
Kehoe S, Hook J, Nankivell M, Jayson GC, Kitchener H, Lopes T, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015;386:249–57. https://doi.org/10.1016/s0140-6736(14)62223-6.
Vergote I, Tropé CG, Amant F, Kristensen GB, Ehlen T, Johnson N, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363:943–53. https://doi.org/10.1056/NEJMoa0908806.
Kyriazi S, Kaye SB, deSouza NM. Imaging ovarian cancer and peritoneal metastases–current and emerging techniques. Nat Rev Clin Oncol. 2010;7:381–93. https://doi.org/10.1038/nrclinonc.2010.47.
Rizzo S, Del Grande M, Manganaro L, Papadia A, Del Grande F. Imaging before cytoreductive surgery in advanced ovarian cancer patients. Int J Gynecol Cancer. 2020;30:133–8. https://doi.org/10.1136/ijgc-2019-000819.
Nam EJ, Yun MJ, Oh YT, Kim JW, Kim JH, Kim S, et al. Diagnosis and staging of primary ovarian cancer: correlation between PET/CT, Doppler US, and CT or MRI. Gynecol Oncol. 2010;116:389–94. https://doi.org/10.1016/j.ygyno.2009.10.059.
Castellucci P, Perrone AM, Picchio M, Ghi T, Farsad M, Nanni C, et al. Diagnostic accuracy of 18F-FDG PET/CT in characterizing ovarian lesions and staging ovarian cancer: correlation with transvaginal ultrasonography, computed tomography, and histology. Nucl Med Commun. 2007;28:589–95. https://doi.org/10.1097/MNM.0b013e3281afa256.
Kitajima K, Murakami K, Yamasaki E, Kaji Y, Fukasawa I, Inaba N, et al. Diagnostic accuracy of integrated FDG-PET/contrast-enhanced CT in staging ovarian cancer: comparison with enhanced CT. Eur J Nucl Med Mol Imaging. 2008;35:1912–20. https://doi.org/10.1007/s00259-008-0890-2.
Michielsen K, Vergote I, Op de Beeck K, Amant F, Leunen K, Moerman P, et al. Whole-body MRI with diffusion-weighted sequence for staging of patients with suspected ovarian cancer: a clinical feasibility study in comparison to CT and FDG-PET/CT. Eur Radiol. 2014;24:889–901. https://doi.org/10.1007/s00330-013-3083-8.
van ’t Sant I, Engbersen MP, Bhairosing PA, Lambregts DMJ, Beets-Tan RGH, van Driel WJ, et al. Diagnostic performance of imaging for the detection of peritoneal metastases: a meta-analysis. Eur Radiol. 2020;30:3101–12. https://doi.org/10.1007/s00330-019-06524-x.
Timmerman D, Planchamp F, Bourne T, Landolfo C, du Bois A, Chiva L, et al. ESGO/ISUOG/IOTA/ESGE Consensus Statement on pre-operative diagnosis of ovarian tumors. Int J Gynecol Cancer. 2021;31:961–82. https://doi.org/10.1136/ijgc-2021-002565.
Mikkelsen MS, Petersen LK, Blaakaer J, Marinovskij E, Rosenkilde M, Andersen G, et al. Assessment of peritoneal metastases with DW-MRI, CT, and FDG PET/CT before cytoreductive surgery for advanced stage epithelial ovarian cancer. Eur J Surg Oncol. 2021;47:2134–41. https://doi.org/10.1016/j.ejso.2021.03.239.
Dendl K, Koerber SA, Finck R, Mokoala KMG, Staudinger F, Schillings L, et al. (68)Ga-FAPI-PET/CT in patients with various gynecological malignancies. Eur J Nucl Med Mol Imaging. 2021;48:4089–100. https://doi.org/10.1007/s00259-021-05378-0.
Liu S, Feng Z, Xu X, Ge H, Ju X, Wu X, et al. Head-to-head comparison of [(18)F]-FDG and [(68) Ga]-DOTA-FAPI-04 PET/CT for radiological evaluation of platinum-sensitive recurrent ovarian cancer. Eur J Nucl Med Mol Imaging. 2023. https://doi.org/10.1007/s00259-022-06096-x.
Zheng W, Liu L, Feng Y, Wang L, Chen Y. Comparison of 68 Ga-FAPI-04 and fluorine-18-fluorodeoxyglucose PET/computed tomography in the detection of ovarian malignancies. Nucl Med Commun. 2023;44:194–203. https://doi.org/10.1097/mnm.0000000000001653.
Xi Y, Sun L, Che X, Huang X, Liu H, Wang Q, et al. A comparative study of [(68)Ga]Ga-FAPI-04 PET/MR and [(18)F]FDG PET/CT in the diagnostic accuracy and resectability prediction of ovarian cancer. Eur J Nucl Med Mol Imaging. 2023. https://doi.org/10.1007/s00259-023-06235-y.
Querleu D, Planchamp F, Chiva L, Fotopoulou C, Barton D, Cibula D, et al. European Society of Gynaecological Oncology (ESGO) guidelines for ovarian cancer surgery. Int J Gynecol Cancer. 2017;27:1534–42. https://doi.org/10.1097/igc.0000000000001041.
Dabir M, Novruzov E, Mattes-György K, Beu M, Dendl K, Antke C, et al. Distinguishing benign and malignant findings on [(68) Ga]-FAPI PET/CT based on quantitative SUV measurements. Mol Imaging Biol. 2023;25:324–33. https://doi.org/10.1007/s11307-022-01759-5.
Oliveira L, Horvat N, Andrieu PIC, Panizza PSB, Cerri GG, Viana PCC. Ovarian cancer staging: what the surgeon needs to know. Br J Radiol. 2021;94:20210091. https://doi.org/10.1259/bjr.20210091.
Mona CE, Benz MR, Hikmat F, Grogan TR, Lueckerath K, Razmaria A, et al. Correlation of (68)Ga-FAPi-46 PET biodistribution with FAP expression by immunohistochemistry in patients with solid cancers: interim analysis of a prospective translational exploratory study. J Nucl Med. 2022;63:1021–6. https://doi.org/10.2967/jnumed.121.262426.
Engbersen MP, Van Driel W, Lambregts D, Lahaye M. The role of CT, PET-CT, and MRI in ovarian cancer. Br J Radiol. 2021;94:20210117. https://doi.org/10.1259/bjr.20210117.
Roze JF, Hoogendam JP, van de Wetering FT, Spijker R, Verleye L, Vlayen J, et al. Positron emission tomography (PET) and magnetic resonance imaging (MRI) for assessing tumour resectability in advanced epithelial ovarian/fallopian tube/primary peritoneal cancer. Cochrane Database Syst Rev. 2018;10:Cd012567. https://doi.org/10.1002/14651858.CD012567.pub2.
Zhao L, Pang Y, Luo Z, Fu K, Yang T, Zhao L, et al. Role of [(68)Ga]Ga-DOTA-FAPI-04 PET/CT in the evaluation of peritoneal carcinomatosis and comparison with [(18)F]-FDG PET/CT. Eur J Nucl Med Mol Imaging. 2021;48:1944–55. https://doi.org/10.1007/s00259-020-05146-6.
Kuten J, Levine C, Shamni O, Pelles S, Wolf I, Lahat G, et al. Head-to-head comparison of [(68)Ga]Ga-FAPI-04 and [(18)F]-FDG PET/CT in evaluating the extent of disease in gastric adenocarcinoma. Eur J Nucl Med Mol Imaging. 2022;49:743–50. https://doi.org/10.1007/s00259-021-05494-x.
Totzeck M, Siebermair J, Rassaf T, Rischpler C. Cardiac fibroblast activation detected by positron emission tomography/computed tomography as a possible sign of cardiotoxicity. Eur Heart J. 2020;41:1060. https://doi.org/10.1093/eurheartj/ehz736.
Funding
This study was supported by the National Natural Science Foundation of China (Nos. 92259304, 81971646, 82122033, and 82202208).
Author information
Authors and Affiliations
Contributions
FK, XL, and JW: study concept and design. XL and FK: manuscript writing. YL and MW: patient enrollment and intraoperative record. JY and TH: tracer synthesized. GL: PET/MR scan. FK and XL: analysis of [68Ga]Ga-FAPI-04 PET imaging results. YT and ZQ: analysis of MRI-DWI imaging results. SL and XL: debulking surgery and surgical exploration. JW, XZ, ML, and TH: data analysis. Critical revision of the manuscript for important intellectual content: all authors.
Corresponding authors
Ethics declarations
Research involving human participants
All procedures performed in this study involving human participants were performed in accordance with the ethics standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethics standards.
Consent to participate
Informed consent was obtained from all participants.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiang Li, Xiaohui Lv, and Zhiyong Quan are first authors and contributed equally to this work.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Lv, X., Quan, Z. et al. Surgical evidence-based comparison of [68Ga]Ga-FAPI-04 PET and MRI-DWI for assisting debulking surgery in ovarian cancer patients. Eur J Nucl Med Mol Imaging 51, 1773–1785 (2024). https://doi.org/10.1007/s00259-023-06582-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06582-w