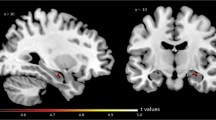
Abstract
Purpose
Aging is a major societal concern due to age-related functional losses. Synapses are crucial components of neural circuits, and synaptic density could be a sensitive biomarker to evaluate brain function. [11C]UCB-J is a positron emission tomography (PET) ligand targeting synaptic vesicle glycoprotein 2A (SV2A), which can be used to evaluate brain synaptic density in vivo.
Methods
We evaluated age-related changes in gray matter synaptic density, volume, and blood flow using [11C]UCB-J PET and magnetic resonance imaging (MRI) in a wide age range of 80 cognitive normal subjects (21–83 years old). Partial volume correction was applied to the PET data.
Results
Significant age-related decreases were found in 13, two, and nine brain regions for volume, synaptic density, and blood flow, respectively. The prefrontal cortex showed the largest volume decline (4.9% reduction per decade: RPD), while the synaptic density loss was largest in the caudate (3.6% RPD) and medial occipital cortex (3.4% RPD). The reductions in caudate are consistent with previous SV2A PET studies and likely reflect that caudate is the site of nerve terminals for multiple major tracts that undergo substantial age-related neurodegeneration. There was a non-significant negative relationship between volume and synaptic density reductions in 16 gray matter regions.
Conclusion
MRI and [11]C-UCB-J PET showed age-related decreases of gray matter volume, synaptic density, and blood flow; however, the regional patterns of the reductions in volume and SV2A binding were different. Those patterns suggest that MR-based measures of GM volume may not be directly representative of synaptic density.




Similar content being viewed by others
Reference
Farokhian F, Yang C, Beheshti I, Matsuda H, Wu S. Age-related gray and white matter changes in normal adult brains. Aging Dis. 2017;8:899–909. https://doi.org/10.14336/AD.2017.0502.
Weinberger DR, Radulescu E. Structural magnetic resonance imaging all over again. JAMA Psychiatry. 2021;78:11–2. https://doi.org/10.1001/jamapsychiatry.2020.1941.
Huttenlocher PR. Synaptic density in human frontal cortex - developmental changes and effects of aging. Brain Res. 1979;163:195–205. https://doi.org/10.1016/0006-8993(79)90349-4.
Terry RD, Katzman R. Life span and synapses: will there be a primary senile dementia? Neurobiol Aging. 2001;22:347–8. https://doi.org/10.1016/s0197-4580(00)00250-5. (discussion 53-4).
Rogalski EJ, Gefen T, Shi J, Samimi M, Bigio E, Weintraub S, et al. Youthful memory capacity in old brains: anatomic and genetic clues from the Northwestern SuperAging Project. J Cogn Neurosci. 2013;25:29–36. https://doi.org/10.1162/jocn_a_00300.
Nabulsi NB, Mercier J, Holden D, Carre S, Najafzadeh S, Vandergeten MC, et al. Synthesis and preclinical evaluation of 11C-UCB-J as a PET tracer for imaging the synaptic vesicle glycoprotein 2A in the brain. J Nucl Med. 2016;57:777–84. https://doi.org/10.2967/jnumed.115.168179.
Finnema SJ, Nabulsi NB, Eid T, Detyniecki K, Lin SF, Chen MK, et al. Imaging synaptic density in the living human brain. Sci Transl Med. 2016;8:348ra96. https://doi.org/10.1126/scitranslmed.aaf6667.
Cai Z, Li S, Matuskey D, Nabulsi N, Huang Y. PET imaging of synaptic density: a new tool for investigation of neuropsychiatric diseases. Neurosci Lett. 2019;691:44–50. https://doi.org/10.1016/j.neulet.2018.07.038.
Bartholome O, Van den Ackerveken P, Sanchez Gil J, de la Brassinne Bonardeaux O, Leprince P, Franzen R, et al. Puzzling out synaptic vesicle 2 family members functions. Front Mol Neurosci. 2017;10:148. https://doi.org/10.3389/fnmol.2017.00148.
Dunn AR, Stout KA, Ozawa M, Lohr KM, Hoffman CA, Bernstein AI, et al. Synaptic vesicle glycoprotein 2C (SV2C) modulates dopamine release and is disrupted in Parkinson disease. Proc Natl Acad Sci U S A. 2017;114:E2253–62. https://doi.org/10.1073/pnas.1616892114.
Chen MK, Mecca AP, Naganawa M, Finnema SJ, Toyonaga T, Lin SF, et al. Assessing synaptic density in Alzheimer disease with synaptic vesicle glycoprotein 2A positron emission tomograPHIC IMAGING. JAMA Neurol. 2018;75:1215–24. https://doi.org/10.1001/jamaneurol.2018.1836.
Finnema SJ, Nabulsi NB, Mercier J, Lin SF, Chen MK, Matuskey D, et al. Kinetic evaluation and test-retest reproducibility of [(11)C]UCB-J, a novel radioligand for positron emission tomography imaging of synaptic vesicle glycoprotein 2A in humans. J Cereb Blood Flow Metab. 2018;38:2041–52. https://doi.org/10.1177/0271678X17724947.
Holmes SE, Scheinost D, Finnema SJ, Naganawa M, Davis MT, DellaGioia N, et al. Lower synaptic density is associated with depression severity and network alterations. Nat Commun. 2019;10:1529. https://doi.org/10.1038/s41467-019-09562-7.
Finnema SJ, Toyonaga T, Detyniecki K, Chen MK, Dias M, Wang Q, et al. Reduced synaptic vesicle protein 2A binding in temporal lobe epilepsy: A [(11) C]UCB-J positron emission tomography study. Epilepsia. 2020;61:2183–93. https://doi.org/10.1111/epi.16653.
Matuskey D, Tinaz S, Wilcox KC, Naganawa M, Toyonaga T, Dias M, et al. Synaptic changes in Parkinson disease assessed with in vivo imaging. Ann Neurol. 2020;87:329–38. https://doi.org/10.1002/ana.25682.
Mecca AP, Chen MK, O’Dell RS, Naganawa M, Toyonaga T, Godek TA, et al. In vivo measurement of widespread synaptic loss in Alzheimer’s disease with SV2A PET. Alzheimers Dement. 2020;16:974–82. https://doi.org/10.1002/alz.12097.
D’Souza DC, Radhakrishnan R, Naganawa M, Ganesh S, Nabulsi N, Najafzadeh S, et al. Preliminary in vivo evidence of lower hippocampal synaptic density in cannabis use disorder. Mol Psychiatry. 2021;26:3192–200. https://doi.org/10.1038/s41380-020-00891-4.
Radhakrishnan R, Skosnik PD, Ranganathan M, Naganawa M, Toyonaga T, Finnema S, et al. In vivo evidence of lower synaptic vesicle density in schizophrenia. Mol Psychiatry. 2021;26:7690–8. https://doi.org/10.1038/s41380-021-01184-0.
Holmes SE, Finnema SJ, Naganawa M, DellaGioia N, Holden D, Fowles K, et al. Imaging the effect of ketamine on synaptic density (SV2A) in the living brain. Mol Psychiatry. 2022;27:2273–81. https://doi.org/10.1038/s41380-022-01465-2.
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–98. https://doi.org/10.1016/0022-3956(75)90026-6.
Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993;43:2412–4. https://doi.org/10.1212/wnl.43.11.2412-a.
Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. 1999;9:179–94. https://doi.org/10.1006/nimg.1998.0395.
Fischl B, Sereno MI, Tootell RB, Dale AM. High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum Brain Mapp. 1999;8:272–84. https://doi.org/10.1002/(sici)1097-0193(1999)8:4%3c272::aid-hbm10%3e3.0.co;2-4.
Fischl B, van der Kouwe A, Destrieux C, Halgren E, Segonne F, Salat DH, et al. Automatically parcellating the human cerebral cortex. Cereb Cortex. 2004;14:11–22. https://doi.org/10.1093/cercor/bhg087.
Rossano S, Toyonaga T, Finnema SJ, Naganawa M, Lu Y, Nabulsi N, et al. Assessment of a white matter reference region for (11)C-UCB-J PET quantification. J Cereb Blood Flow Metab. 2020;40:1890–901. https://doi.org/10.1177/0271678X19879230.
Carson RE, Barker WC, Liow JS, Johnson CA. Design of a motion-compensation OSEM list-mode algorithm for resolution-recovery reconstruction for the HRRT. IEEE Nucl Sci Symp conf Rec. 2003;5:3281–5.
Jin X, Mulnix T, Gallezot JD, Carson RE. Evaluation of motion correction methods in human brain PET imaging–a simulation study based on human motion data. Med Phys. 2013;40:102503. https://doi.org/10.1118/1.4819820.
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM. Fsl. Neuroimage. 2012;62:782–90. https://doi.org/10.1016/j.neuroimage.2011.09.015.
Erlandsson K, Buvat I, Pretorius PH, Thomas BA, Hutton BF. A review of partial volume correction techniques for emission tomography and their applications in neurology, cardiology and oncology. Phys Med Biol. 2012;57:R119-59. https://doi.org/10.1088/0031-9155/57/21/R119.
Lu Y, Toyonaga T, Naganawa M, Gallezot JD, Chen MK, Mecca AP, et al. Partial volume correction analysis for (11)C-UCB-J PET studies of Alzheimer’s disease. Neuroimage. 2021;238:118248. https://doi.org/10.1016/j.neuroimage.2021.118248.
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN, et al. Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab. 2007;27:1533–9. https://doi.org/10.1038/sj.jcbfm.9600493.
Finnema SJ, Rossano S, Naganawa M, Henry S, Gao H, Pracitto R, et al. A single-center, open-label positron emission tomography study to evaluate brivaracetam and levetiracetam synaptic vesicle glycoprotein 2A binding in healthy volunteers. Epilepsia. 2019;60:958–67. https://doi.org/10.1111/epi.14701.
Cunningham VJ, Rabiner EA, Slifstein M, Laruelle M, Gunn RN. Measuring drug occupancy in the absence of a reference region: the Lassen plot re-visited. J Cereb Blood Flow Metab. 2010;30:46–50. https://doi.org/10.1038/jcbfm.2009.190.
Hommel G. A stagewise rejective multiple test procedure based on a modified Bonferroni test. Biometrika. 1988;75:383–6. https://doi.org/10.1093/biomet/75.2.383.
Giorgio A, Santelli L, Tomassini V, Bosnell R, Smith S, De Stefano N, et al. Age-related changes in grey and white matter structure throughout adulthood. Neuroimage. 2010;51:943–51. https://doi.org/10.1016/j.neuroimage.2010.03.004.
Tisserand DJ, van Boxtel MP, Pruessner JC, Hofman P, Evans AC, Jolles J. A voxel-based morphometric study to determine individual differences in gray matter density associated with age and cognitive change over time. Cereb Cortex. 2004;14:966–73. https://doi.org/10.1093/cercor/bhh057.
Sowell ER, Thompson PM, Toga AW. Mapping changes in the human cortex throughout the span of life. Neuroscientist. 2004;10:372–92. https://doi.org/10.1177/1073858404263960.
Koole M, van Aalst J, Devrome M, Mertens N, Serdons K, Lacroix B, et al. Quantifying SV2A density and drug occupancy in the human brain using [(11)C]UCB-J PET imaging and subcortical white matter as reference tissue. Eur J Nucl Med Mol Imaging. 2019;46:396–406. https://doi.org/10.1007/s00259-018-4119-8.
Kijonka M, Borys D, Psiuk-Maksymowicz K, Gorczewski K, Wojcieszek P, Kossowski B, et al. Whole brain and cranial size adjustments in volumetric brain analyses of sex- and age-related trends. Front Neurosci. 2020;14:278. https://doi.org/10.3389/fnins.2020.00278.
Smart K, Liu H, Matuskey D, Chen MK, Torres K, Nabulsi N, et al. Binding of the synaptic vesicle radiotracer [(11)C]UCB-J is unchanged during functional brain activation using a visual stimulation task. J Cereb Blood Flow Metab. 2021;41:1067–79. https://doi.org/10.1177/0271678X20946198.
Mansur A, Rabiner EA, Comley RA, Lewis Y, Middleton LT, Huiban M, et al. Characterization of 3 PET tracers for quantification of mitochondrial and synaptic function in healthy human brain: (18)F-BCPP-EF, (11)C-SA-4503, and (11)C-UCB-J. J Nucl Med. 2020;61:96–103. https://doi.org/10.2967/jnumed.119.228080.
Michiels L, Delva A, van Aalst J, Ceccarini J, Vandenberghe W, Vandenbulcke M, et al. Synaptic density in healthy human aging is not influenced by age or sex: a (11)C-UCB-J PET study. Neuroimage. 2021;232:117877. https://doi.org/10.1016/j.neuroimage.2021.117877.
McGeer PL, McGeer EG, Suzuki JS. Aging and extrapyramidal function. Arch Neurol. 1977;34:33–5. https://doi.org/10.1001/archneur.1977.00500130053010.
Siddiqi Z, Kemper TL, Killiany R. Age-related neuronal loss from the substantia nigra-pars compacta and ventral tegmental area of the rhesus monkey. J Neuropathol Exp Neurol. 1999;58:959–71. https://doi.org/10.1097/00005072-199909000-00006.
Alonso-Nanclares L, Gonzalez-Soriano J, Rodriguez JR, DeFelipe J. Gender differences in human cortical synaptic density. Proc Natl Acad Sci U S A. 2008;105:14615–9. https://doi.org/10.1073/pnas.0803652105.
Esiri MM. Ageing and the brain. J Pathol. 2007;211:181–7. https://doi.org/10.1002/path.2089.
Fjell AM, Walhovd KB. Structural brain changes in aging: courses, causes and cognitive consequences. Rev Neurosci. 2010;21:187–221. https://doi.org/10.1515/revneuro.2010.21.3.187.
Bourgeois JP, Rakic P. Changes of synaptic density in the primary visual cortex of the macaque monkey from fetal to adult stage. J Neurosci. 1993;13:2801–20. https://doi.org/10.1523/JNEUROSCI.13-07-02801.1993.
VanGuilder HD, Yan H, Farley JA, Sonntag WE, Freeman WM. Aging alters the expression of neurotransmission-regulating proteins in the hippocampal synaptoproteome. J Neurochem. 2010;113:1577–88. https://doi.org/10.1111/j.1471-4159.2010.06719.x.
Varnas K, Stepanov V, Halldin C. Autoradiographic mapping of synaptic vesicle glycoprotein 2A in non-human primate and human brain. Synapse. 2020;74:e22157. https://doi.org/10.1002/syn.22157.
Lerman J, Strong HA, LeDez KM, Swartz J, Rieder MJ, Burrows FA. Effects of age on the serum concentration of alpha 1-acid glycoprotein and the binding of lidocaine in pediatric patients. Clin Pharmacol Ther. 1989;46:219–25. https://doi.org/10.1038/clpt.1989.129.
Grandison MK, Boudinot FD. Age-related changes in protein binding of drugs: implications for therapy. Clin Pharmacokinet. 2000;38:271–90. https://doi.org/10.2165/00003088-200038030-00005.
Resnick SM, Pham DL, Kraut MA, Zonderman AB, Davatzikos C. Longitudinal magnetic resonance imaging studies of older adults: a shrinking brain. J Neurosci. 2003;23:3295–301.
Smith CD, Chebrolu H, Wekstein DR, Schmitt FA, Markesbery WR. Age and gender effects on human brain anatomy: a voxel-based morphometric study in healthy elderly. Neurobiol Aging. 2007;28:1075–87. https://doi.org/10.1016/j.neurobiolaging.2006.05.018.
Pieperhoff P, Homke L, Schneider F, Habel U, Shah NJ, Zilles K, et al. Deformation field morphometry reveals age-related structural differences between the brains of adults up to 51 years. J Neurosci. 2008;28:828–42. https://doi.org/10.1523/JNEUROSCI.3732-07.2008.
Terribilli D, Schaufelberger MS, Duran FL, Zanetti MV, Curiati PK, Menezes PR, et al. Age-related gray matter volume changes in the brain during non-elderly adulthood. Neurobiol Aging. 2011;32:354–68. https://doi.org/10.1016/j.neurobiolaging.2009.02.008.
Salat DH, Buckner RL, Snyder AZ, Greve DN, Desikan RS, Busa E, et al. Thinning of the cerebral cortex in aging. Cereb Cortex. 2004;14:721–30. https://doi.org/10.1093/cercor/bhh032.
Chen JJ, Rosas HD, Salat DH. Age-associated reductions in cerebral blood flow are independent from regional atrophy. Neuroimage. 2011;55:468–78. https://doi.org/10.1016/j.neuroimage.2010.12.032.
Nugent S, Tremblay S, Chen KW, Ayutyanont N, Roontiva A, Castellano CA, et al. Brain glucose and acetoacetate metabolism: a comparison of young and older adults. Neurobiol Aging. 2014;35:1386–95. https://doi.org/10.1016/j.neurobiolaging.2013.11.027.
Shen X, Liu H, Hu Z, Hu H, Shi P. The relationship between cerebral glucose metabolism and age: report of a large brain PET data set. PLoS One. 2012;7:e51517. https://doi.org/10.1371/journal.pone.0051517.
Kim IJ, Kim SJ, Kim YK. Age- and sex-associated changes in cerebral glucose metabolism in normal healthy subjects: statistical parametric mapping analysis of F-18 fluorodeoxyglucose brain positron emission tomography. Acta Radiol. 2009;50:1169–74. https://doi.org/10.3109/02841850903258058.
Knopman DS, Jack CR Jr, Wiste HJ, Lundt ES, Weigand SD, Vemuri P, et al. 18F-fluorodeoxyglucose positron emission tomography, aging, and apolipoprotein E genotype in cognitively normal persons. Neurobiol Aging. 2014;35:2096–106. https://doi.org/10.1016/j.neurobiolaging.2014.03.006.
Loessner A, Alavi A, Lewandrowski KU, Mozley D, Souder E, Gur RE. Regional cerebral function determined by FDG-PET in healthy volunteers: normal patterns and changes with age. J Nucl Med. 1995;36:1141–9.
Kalpouzos G, Chetelat G, Baron JC, Landeau B, Mevel K, Godeau C, et al. Voxel-based mapping of brain gray matter volume and glucose metabolism profiles in normal aging. Neurobiol Aging. 2009;30:112–24. https://doi.org/10.1016/j.neurobiolaging.2007.05.019.
Chen MK, Mecca AP, Naganawa M, Gallezot JD, Toyonaga T, Mondal J, et al. Comparison of [(11)C]UCB-J and [(18)F]FDG PET in Alzheimer’s disease: a tracer kinetic modeling study. J Cereb Blood Flow Metab. 2021;41:2395–409. https://doi.org/10.1177/0271678X211004312.
Karrer TM, Josef AK, Mata R, Morris ED, Samanez-Larkin GR. Reduced dopamine receptors and transporters but not synthesis capacity in normal aging adults: a meta-analysis. Neurobiol Aging. 2017;57:36–46. https://doi.org/10.1016/j.neurobiolaging.2017.05.006.
Matuskey D, Pittman B, Planeta-Wilson B, Walderhaug E, Henry S, Gallezot JD, et al. Age effects on serotonin receptor 1B as assessed by PET. J Nucl Med. 2012;53:1411–4. https://doi.org/10.2967/jnumed.112.103598.
Nord M, Cselenyi Z, Forsberg A, Rosenqvist G, Tiger M, Lundberg J, et al. Distinct regional age effects on [11C]AZ10419369 binding to 5-HT1B receptors in the human brain. Neuroimage. 2014;103:303–8. https://doi.org/10.1016/j.neuroimage.2014.09.040.
DuBois JM, Rousset OG, Rowley J, Porras-Betancourt M, Reader AJ, Labbe A, et al. Characterization of age/sex and the regional distribution of mGluR5 availability in the healthy human brain measured by high-resolution [(11)C]ABP688 PET. Eur J Nucl Med Mol Imaging. 2016;43:152–62. https://doi.org/10.1007/s00259-015-3167-6.
Mecca AP, Rogers K, Jacobs Z, McDonald JW, Michalak HR, DellaGioia N, et al. Effect of age on brain metabotropic glutamate receptor subtype 5 measured with [(18)F]FPEB PET. Neuroimage. 2021;238:118217. https://doi.org/10.1016/j.neuroimage.2021.118217.
Kato H, Shimosegawa E, Isohashi K, Kimura N, Kazui H, Hatazawa J. Distribution of cortical benzodiazepine receptor binding in right-handed healthy humans: a voxel-based statistical analysis of iodine 123 iomazenil SPECT with partial volume correction. AJNR Am J Neuroradiol. 2012;33:1458–63. https://doi.org/10.3174/ajnr.A3005.
Tobinaga M, Suzuki Y, Fujinaka H, Ozawa T, Nakajima T. Age-related increase in GABAA receptor distribution in the prefrontal cortex. J Clin Neurosci. 2019;64:106–10. https://doi.org/10.1016/j.jocn.2019.03.044.
Acknowledgements
We deeply thank all the staff of the Yale PET Center for their expert assistance.
Funding
This study was supported by grants UL1 TR000142 from the National Center for Advancing Translational Sciences (NCATS), and T32GM136651 from the National Institute of General Medical Sciences, components of the National Institutes of Health (NIH).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclaimer
The AQ contents of this article are solely the responsibility of the authors and do not necessarily represent the official view of NIH.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Toyonaga, T., Khattar, N., Wu, Y. et al. The regional pattern of age-related synaptic loss in the human brain differs from gray matter volume loss: in vivo PET measurement with [11C]UCB-J. Eur J Nucl Med Mol Imaging 51, 1012–1022 (2024). https://doi.org/10.1007/s00259-023-06487-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06487-8