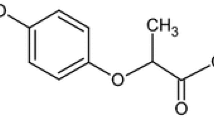
Abstract
Diphenyl ether herbicides, typical globally used herbicides, threaten the agricultural environment and the sensitive crops. The microbial degradation pathways of diphenyl ether herbicides are well studied, but the nitroreduction of diphenyl ether herbicides by purified enzymes is still unclear. Here, the gene dnrA, encoding a nitroreductase DnrA responsible for the reduction of nitro to amino groups, was identified from the strain Bacillus sp. Za. DnrA had a broad substrate spectrum, and the Km values of DnrA for different diphenyl ether herbicides were 20.67 μM (fomesafen), 23.64 μM (bifenox), 26.19 μM (fluoroglycofen), 28.24 μM (acifluorfen), and 36.32 μM (lactofen). DnrA also mitigated the growth inhibition effect on cucumber and sorghum through nitroreduction. Molecular docking revealed the mechanisms of the compounds fomesafen, bifenox, fluoroglycofen, lactofen, and acifluorfen with DnrA. Fomesafen showed higher affinities and lower binding energy values for DnrA, and residue Arg244 affected the affinity between diphenyl ether herbicides and DnrA. This research provides new genetic resources and insights into the microbial remediation of diphenyl ether herbicide-contaminated environments.
Key points
• Nitroreductase DnrA transforms the nitro group of diphenyl ether herbicides.
• Nitroreductase DnrA reduces the toxicity of diphenyl ether herbicides.
• The distance between Arg244 and the herbicides is related to catalytic efficiency.
Graphical abstract






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this manuscript.
References
Akiva E, Copp JN, Tokuriki N, Babbitt PC (2017) Evolutionary and molecular foundations of multiple contemporary functions of the nitroreductase superfamily. Proc Natl Acad Sci U S A 114(45):E9549–E9558. https://doi.org/10.1073/pnas.1706849114
Boddu RS, Perumal O, Divakar K (2021) Microbial nitroreductases: a versatile tool for biomedical and environmental applications. Biotechnol Appl Biochem 68(6):1518–1530. https://doi.org/10.1002/bab.2073
Carles L, Donnadieu LRC, Wawrzyniak A, Besse P, Batisson SEL (2021) Genomic analysis of the Bacillus megaterium Mes11: new insights into nitroreductase genes associated with the degradation of mesotrione. Int Biodeterior Biodegrad 162:105254. https://doi.org/10.1016/j.ibiod.2021.105254
Chen J, Rosen BP (2020) The Pseudomonas putida NfnB nitroreductase confers resistance to roxarsone. Sci Total Environ 748:141339. https://doi.org/10.1016/j.scitotenv.2020.141339
Chen ZJ, Qiao YX, Zhang N, Liu J, Yang H (2021) Insight into metabolism pathways of pesticide fomesafen in rice: reducing cropping and environmental risks. Environ Pollut 283:117128. https://doi.org/10.1016/j.envpol.2021.117128
Cui N, Wang S, Khorram MS, Fang H, Yu Y (2018) Microbial degradation of fomesafen and detoxification of fomesafen-contaminated soil by the newly isolated strain Bacillus sp FE-1 via a proposed biochemical degradation pathway. Sci Total Environ 616:1612–1619. https://doi.org/10.1016/j.scitotenv.2017.10.151
Dan HA, Dan LGM, Barroso ALL, Procopio SO, Oliveira RS Jr, Silva AG, Lima MDB, Feldkircher C (2010) Residual activity of herbicides used in soybean agriculture on grain sorghum crop succession. Planta Daninha 28:1087–1095. https://doi.org/10.1590/s0100-83582010000500016
Diao JL, Xu P, Wang P, Lu DH, Lu YL, Zhou ZQ (2010) Enantioselective degradation in sediment and aquatic toxicity to Daphnia magna of the herbicide lactofen enantiomers. J Agric Food Chem 58(4):2439–2445. https://doi.org/10.1021/jf9038327
Dudev T, Lim C (2014) Competition among metal ions for protein binding sites: determinants of metal ion selectivity in proteins. Chem Rev 114(1):538–556. https://doi.org/10.1021/cr4004665
Feng ZZ, Li QF, Zhang J, Zhang J, Huang X, Lu P, Li SP (2012) Microbial degradation of fomesafen by a newly isolated strain Pseudomonas zeshuii BY-1 and the biochemical degradation pathway. J Agric Food Chem 60(29):7104–7110. https://doi.org/10.1021/jf3011307
Fu Y, Zhao J, Chen Z (2018) Insights into the molecular mechanisms of protein-ligand interactions by molecular docking and molecular dynamics simulation: a case of oligopeptide binding protein. Comput Math Methods Med 2018:3502514. https://doi.org/10.1155/2018/3502514
Gao W, Li XT, Ren D, Sun SS, Huo JQ, Wang YE, Chen L, Zhang JL (2019) Design and synthesis of N-phenyl phthalimides as potent protoporphyrinogen oxidase inhibitors. Molecules 24(23):4363. https://doi.org/10.3390/molecules24234363
Gao Y, Chen S, Hu M, Hu Q, Luo J, Li Y (2012) Purification and characterization of a novel chlorpyrifos hydrolase from Cladosporium cladosporioides Hu-01. PLoS ONE 7(6):e38137. https://doi.org/10.1371/journal.pone.0038137
Hao GF, Tan Y, Yu NX, Yang GF (2011) Structure-activity relationships of diphenyl-ether as protoporphyrinogen oxidase inhibitors: insights from computational simulations. J Comput-Aided Mol Des 25(3):213–222. https://doi.org/10.1007/s10822-011-9412-6
Hu T, Xiang Y, Chen Q, Shang N, Xu M, Huang X (2020) A novel esterase LanE from Edaphocola flava HME-24 and the enantioselective degradation mechanism of herbicide lactofen. Ecotoxicol Environ Saf 205:111141. https://doi.org/10.1016/j.ecoenv.2020.111141
Hu ZJ, Ma BY, Wolfson H, Nussinov R (2000) Conservation of polar residues as hot spots at protein interfaces. Proteins 39(4):331–342. https://doi.org/10.1002/(sici)1097-0134(20000601)39:4%3c331::Aid-prot60%3e3.3.Co;2-1
Huang Z, Mao X, Lv X, Sun G, Zhang H, Lu W, Liu Y, Li J, Du G, Liu L (2021) Engineering diacetylchitobiose deacetylase from Pyrococcus horikoshii towards an efficient glucosamine production. Bioresour Technol 334:125241. https://doi.org/10.1016/j.biortech.2021.125241
Janczy-Cempa E, Mazuryk O, Sirbu D, Chopin N, Zarnik M, Zastawna M, Colas C, Hiebel M-A, Suzenet F, Brindell M (2021) Nitro-pyrazinotriazapentalene scaffolds-nitroreductase quantification and in vitro fluorescence imaging of hypoxia. Sens Actuator B 346:130504. https://doi.org/10.1016/j.snb.2021.130504
Lei BF, Liu MY, Huang SQ, Tu SC (1994) Vibrio-harveyi NADPH-flavin oxidoreductase-cloning, sequencing and overexpression of the gene and purification and characterization of the cloned enzyme. J Bacteriol 176(12):3552–3558. https://doi.org/10.1128/jb.176.12.3552-3558.1994
Li Z, Gao J, Tian Y, Wang B, Xu J, Fu X, Han H, Wang L, Zhang W, Wang Y, Deng Y, Gong Z, Peng R, Yao Q (2022) ElNFS1, a nitroreductase gene from Enterobacter ludwigii, confers enhanced detoxification and phytoremediation of 4-nitrobenzaldehyde in rice. Environ Pollut 314:120292. https://doi.org/10.1016/j.envpol.2022.120292
Liang Y, Wei L, Hu J (2021) Residues and dietary intake risk assessments of clomazone, fomesafen, haloxyfop-methyl and its metabolite haloxyfop in spring soybean field ecosystem. Food Chem 360:129921. https://doi.org/10.1016/j.foodchem.2021.129921
Liang B, Zhao YK, Lu P, Li SP, Huang X (2010) Biotransformation of the diphenyl ether herbicide lactofen and purification of a lactofen esterase from Brevundimonas sp LY-2. J Agric Food Chem 58(17):9711–9715. https://doi.org/10.1021/jf101974y
Liang B, Lu P, Li H, Li R, Li S, Huang X (2009) Biodegradation of fomesafen by strain Lysinibacillus sp ZB-1 isolated from soil. Chemosphere 77(11):1614–1619. https://doi.org/10.1016/j.chemosphere.2009.09.033
Meng L, Sun T, Li M, Saleem M, Zhang Q, Wang C (2019) Soil-applied biochar increases microbial diversity and wheat plant performance under herbicide fomesafen stress. Ecotoxicol Environ Saf 171:75–83. https://doi.org/10.1016/j.ecoenv.2018.12.065
Ni H, Wang F, Li N, Yao L, Dai C, He Q, He J, Hong Q (2016) Pendimethalin nitroreductase is responsible for the initial pendimethalin degradation step in Bacillus subtilis Y3. Appl Environ Microbiol 82(24):7052–7062. https://doi.org/10.1128/aem.01771-16
Ni H, Zeng SQ, Qin X, Sun XW, Zhang S, Zhao XY, Yu ZN, Li L (2015) Molecular docking and site-directed mutagenesis of a Bacillus thuringiensis chitinase to improve chitinolytic, synergistic lepidopteran-larvicidal and nematicidal activities. Int J Biol Sci 11(3):304–315. https://doi.org/10.7150/ijbs.10632
Peachey E, Doohan D, Koch T (2012) Selectivity of fomesafen based systems for preemergence weed control in cucurbit crops. Crop Prot 40:91–97. https://doi.org/10.1016/j.cropro.2012.04.003
Pitsawong W, Hoben JP, Miller AF (2014) Understanding the broad substrate repertoire of nitroreductase based on its kinetic mechanism. J Biol Chem 289(22):15203–15214. https://doi.org/10.1074/jbc.M113.547117
Preethi PS, Hariharan NM, Vickram S, Rameshpathy M, Manikandan S, Subbaiya R, Karmegam N, Yadav V, Ravindran B, Chang SW, Awasthi MK (2022) Advances in bioremediation of emerging contaminants from industrial wastewater by oxidoreductase enzymes. Bioresour Technol 359:127444. https://doi.org/10.1016/j.biortech.2022.127444
Rabemanolontsoa H, Saka S (2016) Various pretreatments of lignocellulosics. Bioresour Technol 199:83–91. https://doi.org/10.1016/j.biortech.2015.08.029
Santos-Martins D, Solis-Vasquez L, Tillack AF, Sanner MF, Koch A, Forli S (2021) Accelerating autodock4 with gpus and gradient-based local search. J Chem Theory Comput 17(2):1060–1073. https://doi.org/10.1021/acs.jctc.0c01006
Shang N, Chen L, Cheng M, Tian Y, Huang X (2022) Biodegradation of diphenyl ether herbicide lactofen by Bacillus sp. YS-1 and characterization of two initial degrading esterases. Sci Total Environ 806:151357. https://doi.org/10.1016/j.scitotenv.2021.151357
Sree BR, Sowjanya B, Divakar K (2019) Metagenomic bioprospecting of novel oxygen insensitive nitroreductase for degradation of nitro aromatic compounds. Int Biodeterior Biodegrad 143:104737. https://doi.org/10.1016/j.ibiod.2019.104737
Susha VS, Das TK, Nath CP, Pandey R, Paul S, Ghosh S (2018) Impacts of tillage and herbicide mixture on weed interference, agronomic productivity and profitability of a maize-wheat system in the north-western Indo-Gangetic Plains. Field Crops Res 219:180–191. https://doi.org/10.1016/j.fcr.2018.02.003
Taha M, Ismail NH, Khan A, Shah SAA, Anwar A, Halim SA, Fatmi MQ, Imran S, Rahim F, Khan KM (2015) Synthesis of novel derivatives of oxindole, their urease inhibition and molecular docking studies. Bioorg Med Chem Lett 25(16):3285–3289. https://doi.org/10.1016/j.bmcl.2015.05.069
Wang F, Yi X, Qu H, Chen L, Liu D, Wang P, Zhou Z (2017) Enantioselective accumulation, metabolism and phytoremediation of lactofen by aquatic macrophyte Lemna minor. Ecotoxicol Environ Saf 143:186–192. https://doi.org/10.1016/j.ecoenv.2017.04.051
Wu XH, Zhang Y, Du PQ, Xu J, Dong FS, Liu XG, Zheng YQ (2018) Impact of fomesafen on the soil microbial communities in soybean fields in Northeastern China. Ecotoxicol Environ Saf 148:169–176. https://doi.org/10.1016/j.ecoenv.2017.10.003
Xu Z, Ni H, Huang Y, Meng Y, Cao Z, Liao X, Zhang S, Guo X, Lu H (2020) Effect of fomesafen on the embryonic development of zebrafish. Chemosphere 259:127380. https://doi.org/10.1016/j.chemosphere.2020.127380
Yin XJ, Zeng YJ, Chen J, Liu L, Gao ZZ (2022) Combined active pocket and hinge region engineering to develop an NADPH-dependent phenylglycine dehydrogenase. Bioorg Chem 120:105601. https://doi.org/10.1016/j.bioorg.2022.105601
Zang H, Wang H, Miao L, Cheng Y, Zhang Y, Liu Y, Sun S, Wang Y, Li C (2020) Carboxylesterase, a de-esterification enzyme, catalyzes the degradation of chlorimuron-ethyl in Rhodococcus erythropolis D310–1. J Hazard Mater 387:121684. https://doi.org/10.1016/j.jhazmat.2019.121684
Zenno S, Kobori T, Tanokura M, Saigo K (1998) Purification and characterization of NfrA1, a Bacillus subtilis nitro/flavin reductase capable of interacting with the bacterial luciferase. Biosci Biotechnol Biochem 62(10):1978–1987. https://doi.org/10.1271/bbb.62.1978
Zenno S, Koike H, Kumar AN, Jayaraman R, Tanokura M, Saigo K (1996) Biochemical characterization of NfsA, the Escherichia coli major nitroreductase exhibiting a high amino acid sequence homology to Frp, a Vibrio harveyi flavin oxidoreductase. J Bacteriol 178(15):4508–4514. https://doi.org/10.1128/jb.178.15.4508-4514.1996
Zhang J, Lu L, Chen F, Chen L, Yin J, Huang X (2018) Detoxification of diphenyl ether herbicide lactofen by Bacillus sp Za and enantioselective characteristics of an esterase gene lacE. J Hazard Mater 341:336–345. https://doi.org/10.1016/j.jhazmat.2017.07.064
Zhang J, Zhao M, Yu D, Yin J, Zhang H, Huang X (2017) Biochemical characterization of an enantioselective esterase from Brevundimonas sp LY-2. Microb Cell Fact 16:112. https://doi.org/10.1186/s12934-017-0727-4
Zhao H, Xu J, Dong F, Liu X, Wu Y, Wu X, Zheng Y (2016) Characterization of a novel oxyfluorfen-degrading bacterial strain Chryseobacterium aquifrigidense and its biochemical degradation pathway. Appl Microbiol Biotechnol 100(15):6837–6845. https://doi.org/10.1007/s00253-016-7504-x
Zhu X, Wang X, Li N, Wang Q, Liao C (2022) Bioelectrochemical system for dehalogenation: a review. Environ Pollut 293:118519. https://doi.org/10.1016/j.envpol.2021.118519
Zhou C, Chen TJ, Gu AD, Hu ZF, Li Y, Gong T, Chen JJ, Yang JL, Zhu P (2023) Combining protein and metabolic engineering to achieve green biosynthesis of 12 beta-O-Glc-PPD in Saccharomyces cerevisiae. Green Chem 25(4):1356–1367. https://doi.org/10.1039/d2gc04380g
Zuo Y, Yang SG, Luo YP, Tan Y, Hao GF, Wu QY, Xi Z, Yang GF (2013) Design and synthesis of 1-(benzothiazol-5-yl)-1H-1,2,4-triazol-5-ones as protoporphyrinogen oxidase inhibitors. Bioorg Med Chem 21(11):3245–3255. https://doi.org/10.1016/j.bmc.2013.03.056
Funding
This study was supported by the National Natural Science Fund of China (41977119), the Primary Research and Development Plan of Jiangsu Province (BE2020692), Jiangsu Agricultural Science and Technology Innovation Fund (JASTIF, CX (22) 3136), and the Natural Science Foundation of Henan Province (212300410215).
Author information
Authors and Affiliations
Contributions
YNT and LYL conceived and designed research, conducted experiments, and analyzed the data. YNT and GQZ drafted the manuscript. LYL and HZ contributed reagents and analytical tools. GQZ, HZ, and XH revised this manuscript. HZ and XH applied for funding and supervised research. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tian, Y., Zhao, G., Cheng, M. et al. A nitroreductase DnrA catalyzes the biotransformation of several diphenyl ether herbicides in Bacillus sp. Za. Appl Microbiol Biotechnol 107, 5269–5279 (2023). https://doi.org/10.1007/s00253-023-12647-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12647-5